Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
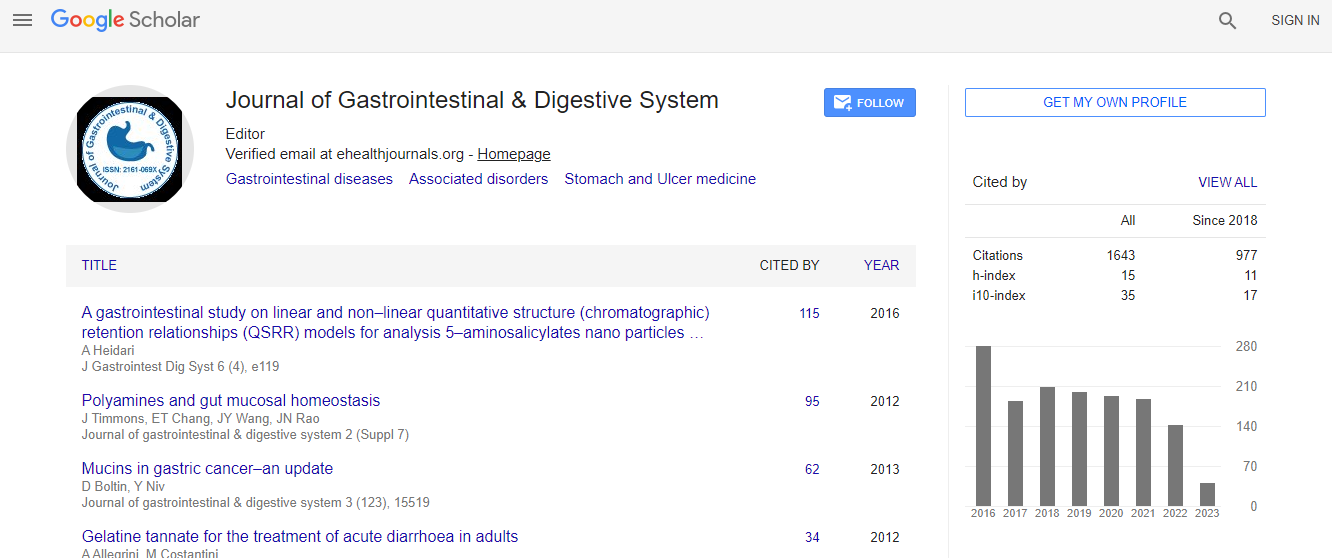
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
A new family of Cu(II) photoactive complexes: Enhanced DNA intercalation and fragmentation
International Conference on Gastrointestinal Cancer and Therapeutics & 4th World Congress on Digestive & Metabolic Diseases & 26th Annual Congress on Cancer Science and Targeted Therapies
Theodoros Mikroulis
National and Kapodistrian University of Athens. Greece
Posters & Accepted Abstracts: J Gastrointest Dig Syst
Abstract
Statement of the Problem: Treatment of aggressive and not accessible tumors, such as glioblastoma multiforme, is based on the synergistic result of surgical operation, radiotherapy and chemotherapy. However, these techniques come not only with serious side effects, but also with marginal improvement of the patient’s life expectancy.1 Photodynamic Therapy (PDT) has emerged as an attractive alternative to conventional cancer treatment. It induces destruction of cancer cells upon irradiation of specific photoactive compounds called photosensitizers (PS), usually by generating the highly reactive single-state oxygen 1O2.2-4 Transition metal complexes have been utilized in PDT exactly because they can promote such photochemical transformations, exhibit strong absorption in the UV-Vis region of the electromagnetic spectrum, and relatively long emission lifetimes of their triplet metal-to-ligand charge transfer state (3MLCT).5-6 The present study reports the synthesis of three novel Cu(II)-based complexes, bearing bidentate bipyridine-type ligands able to intercalate to the DNA strand and cleave it upon irradiation, resulting in apoptosis of the treated cells. Methodology & Theoretical Orientation: Three novel Cu(II)- based complexes were synthesized and characterized using mass spectroscopy, FT-IR and UV-Vis. The binding constants of these complexes with DNA were determined (before and after irradiation) by calculating the concentration of the complex at 50% quenching of DNA-bound ethidium bromide emission intensity. Agarose gel electrophoresis helped visualize the ability of these complexes to bind and cleave DNA upon irradiation. Findings: These novel complexes show high affinity for DNA, (calculated binding constants were one order of magnitude lower than that of ethidium bromide). When incubated in the dark, complexation with DNA resulted in only a small amount of fragmentation, which was substantially enhanced after irradiation, especially when the concentration of the complexes was increased to 200μM or higher. As a result, these complexes could potentially be used as photosensitizers in PDT.Biography
Theodoros Mikroulis is a chemist working at the interface of Organic and Medicinal Chemistry for the development of drugs and other biologically-related compounds against various diseases. He graduated from the Department of Chemistry of the National and Kapodistrian University of Athens and obtained a M.Sc. degree in Medicinal and Biological Chemistry at the University of Edinburgh. He is currently working towards his PhD on the synthesis of photoactive compounds that can potentially be used as anticancer agents, in the research group of Professor Georgios C. Vougioukalakis in the National and Kapodistrian University of Athens.
E-mail: theo.mik0@gmail.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi