Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
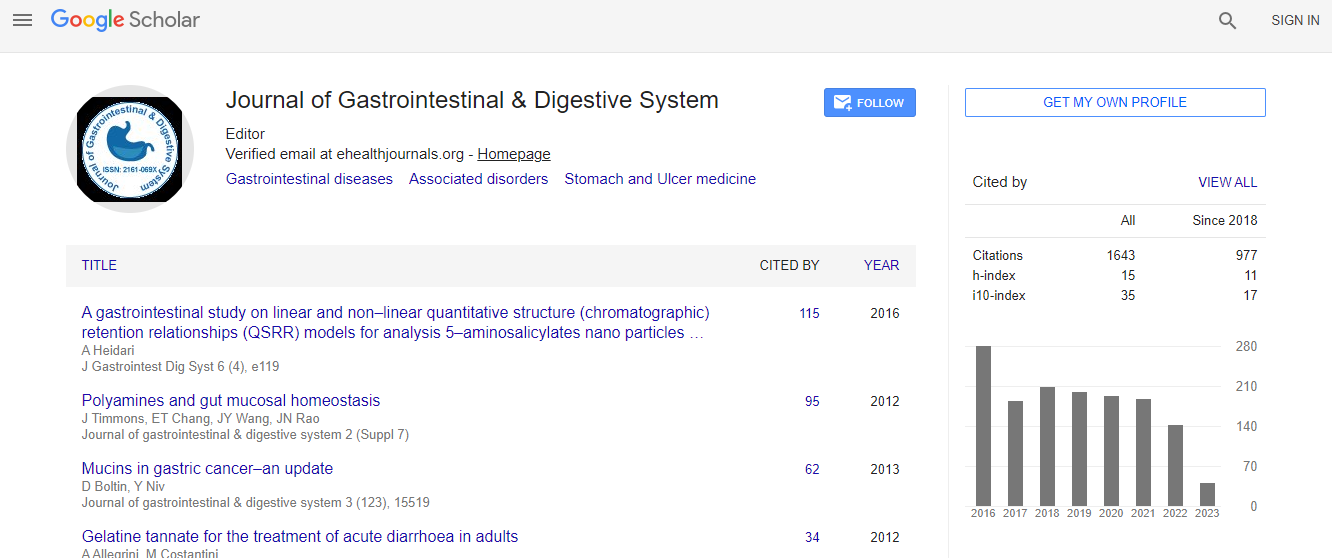
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Activation of Macrophage and Its Impact on structural defects of rat Liver in experimental Diabetes
9th Euro Global Gastroenterology Conference
Danilova I, Gette I, Abidov M
Ural Federal University, Russian Federation
Posters & Accepted Abstracts: J Gastrointest Dig Syst
Abstract
Statement of the Problem: The Morphofunctional state of the liver in diabetes mellitus should be assessed since all metabolic processes occur in liver; hepatocytes have the insulin-dependent mechanism of glucose metabolism and 40-60% of insulin is inactivated in liver. The structure of damaged liver tissue and mechanisms of hyperglycemia which cause the damage were described in many investigations. The assessment of various methods of damaged liver correction was performed. It is known that hepatic macrophages play key role in tissue injury and repair. However, its effects on regenerative processes in diabetic liver are not investigated enough. We previously set that the macrophage modulator 3-aminophthalhydrazid (3-APH) activated anti-inflammatory and antioxidant functions in monocyte-macrophage cells. The purpose of this study: is to evaluate the impact of macrophages activated by 3-APH on structural defects in diabetic rat liver. Methodology: A damaged liver tissue and hepatocyte reparation were studied in alloxan diabetic rats after modulation of macrophage activity by 3-APH during 30 and 60 days. Finding: The morphological study of diabetic liver tissue revealed degenerative changes in hepatocytes, inflammatory processes in the portal and intralobular stroma, microangiopathy and blood circulation disorders. Structural disorders of liver parenchyma increased at a prolongation of AD up to 60 days. It was found that the administration of 3-APH contributed to reduction of damage and restoration of normal liver histological structure due to activation of reparative processes. Conclusion: Thus, macrophage activation in diabetic liver causes increase in hepatocyte activity and compensation of structural damage in liver tissue. 3-APH is likely to be a promising drug for correction of liver tissue in diabetes due to its hepato protective effect. The study is supported by the grant from the Russian Science Foundation, the project ��? 16-15-00039. Figure 1: a - histological structure of intact rat liver tissue. b ��?diabetic liver tissue (30 days): 1- plethora in central venules, 2- lymphocytic infiltration of sinusoidal capillary, 3 - degeneration of hepatocytes. c ��? diabetic liver tissue (60 days): 1 - intralobular necrosis of hepatocytes, 2 - edema, vasodilatation, 3 - desquamation of endothelial of sinusoidal capillaries. d ��?diabetic liver tissue (60 days) treated by 3-APH: 1 - polyploid hepatocytes, 2 ��? dual-nucleus hepatocytes. Staining by hematoxylin-eosin. X400.Recent Publications
â�?¢ Lucchesi A.N. et al. (2013) Diabetes mellitus triggers oxidative stress in the liver of alloxan-treated rats: a mechanism for diabetic chronic liver disease. Acta Cirurgica Brasileira 28.7: 502-508.
â�?¢ Lucchesi A. N., Cassettari L. L., Spadella, C. T. (2015). Alloxan-induced diabetes causes morphological and ultrastructural changes in rat liver that resemble the natural history of chronic fatty liver disease in humans. Journal of diabetes research, 2015.
â�?¢ Holt M.P.,Cheng L., Ju C. (2008) Identification and characterization of infiltrating macrophages in acetaminopheninduced liver injury. Journal of leukocyte biology 84.6: 1410-1421.
â�?¢ Thomas J.A. et al. (2011) Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function. Hepatology 53.6: 2003-2015.
â�?¢ Welt K. et al. (2004) Ultrastructural, immunohistochemical and biochemical investigations of the rat liver exposed to experimental diabetes und acute hypoxia with and without application of Ginkgo extract. Experimental and Toxicologic Pathology 55.5: 331-345.
Biography
Irina Danilova, professor, director of the Fundamental Medicine department of the Ural Federal University (UFU), Yekaterinburg, Russia. She is an author of more than 220 scientific publications, “Macrophages Non-immunological Functions” monograph and 5 patents including an EU, USA patent. Irina Gette is a scientific associate of UFU, PhD. She has published 97 papers in reputed journals. Musa Abidov a professor, he is a creator of 3-aminophthalhydrazid. The scope of their research interests has impact on investigation of macrophages during reparative processes in tissue under various pathological states.
Email: ig-danilova@yandex.ru

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi