Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
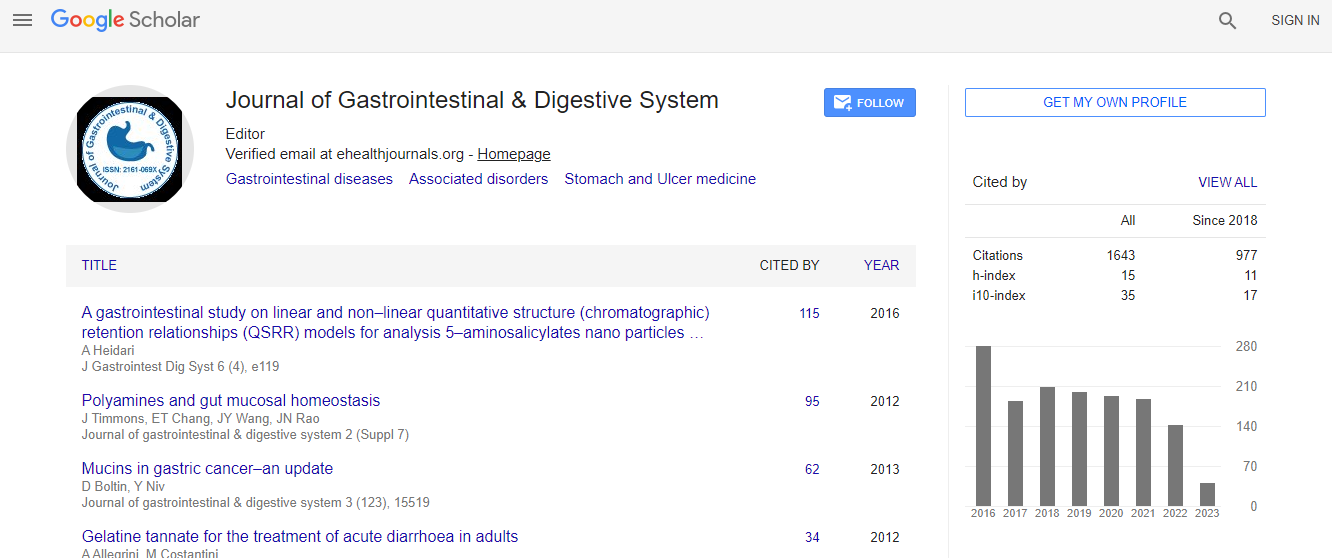
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Comparative molecular analysis of gastrointestinal adenocarcinomas yields novel therapeutic strategies
International Conference on Gastrointestinal Cancer and Therapeutics & 4th World Congress on Digestive & Metabolic Diseases & 26th Annual Congress on Cancer Science and Targeted Therapies
Nilay Sethi
Dana Farber Cancer Institute, USA
Keynote: J Gastrointest Dig Syst
Abstract
Combined, adenocarcinomas of the esophagus, stomach, colon, and rectum account for a devastating 1.4 million deaths each year worldwide. Conventional treatment approaches currently offer modest benefit, inspiring our effort to identify previously unrecognized biological mechanisms underlying the pathogenesis of gastroesophageal (GE) and colorectal (CR) cancers. To better understand these cancers, we analyzed 921 adenocarcinomas of the esophagus (n=79), stomach (n=383), colon (n=341) and rectum (n= 118) obtained from the fresh frozen tissue by The Cancer Genome Atlas Network using six molecular platforms. We uncovered five molecular subtypes that largely transcended anatomic boundaries including Epstein- Barr Virus (EBV) positive and Hypermutated tumors (HM), which further substratified into MSI and Hypermutated-SNV (HM-SNV). The remaining two groups were distinguished by presence or absence of extensive SCNAs. Chromosomal instability (CIN) tumors exhibited marked aneuploidy, a feature that was essentially absent in the Genome stable (GS) subtype. Evaluating the anatomic distribution revealed that HM tumors primarily occupied the central part of the GI tract in the distal stomach and proximal colon, whereas CIN tumors were more prevalent in the anatomic extremes, namely, the esophagus and distal colon/ rectum. Inspired by recent clinical advances in immunotherapy, we studied associations between our molecular groupings and key immune features. EBV+ were enriched for gene expression scores associated with CD8+ T-cells, M1-macrophages, and IFN-γ signatures. MSI tumors showed the next greatest IFN-γ signature and displayed diverse immune signatures depending on the tissue of origin of translational importance, an attenuation in HLA/antigen presentation and significant elevation in NK-cell gene expression was found in CR HM-SNV. Blockade of inhibitory signals or stimulation of activating cues can tip the balance in favor of cytotoxic endogenous NK activity, which may be a therapeutic option for patients with HM-SNV tumors. Overall, these findings further the rationale to develop innovative therapeutic strategies that recruit and reinvigorate the host immune system to battle cancer.Biography
Nilay Sethi earned his PhD from the Molecular Biology Department at Princeton University in 2010 and his MD at Rutgers Robert Wood Johnson Medical School in 2012. He completed his internal medicine training at the University of California San Francisco (UCSF) in 2014 and medical oncology fellowship at the Dana-Farber Cancer Institute in 2017. His work in cancer metastasis has led to publications, including a landmark study on Jagged1 in bone metastasis, and yielded numerous awards. He has now focused on better understanding the molecular mechanisms underlying gastrointestinal malignancies with the hope of translational advances that will improve outcomes in patients suffering from these devastating diseases.
E-mail: nilay_sethi@dfci.harvard.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi