Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
Google Scholar citation report
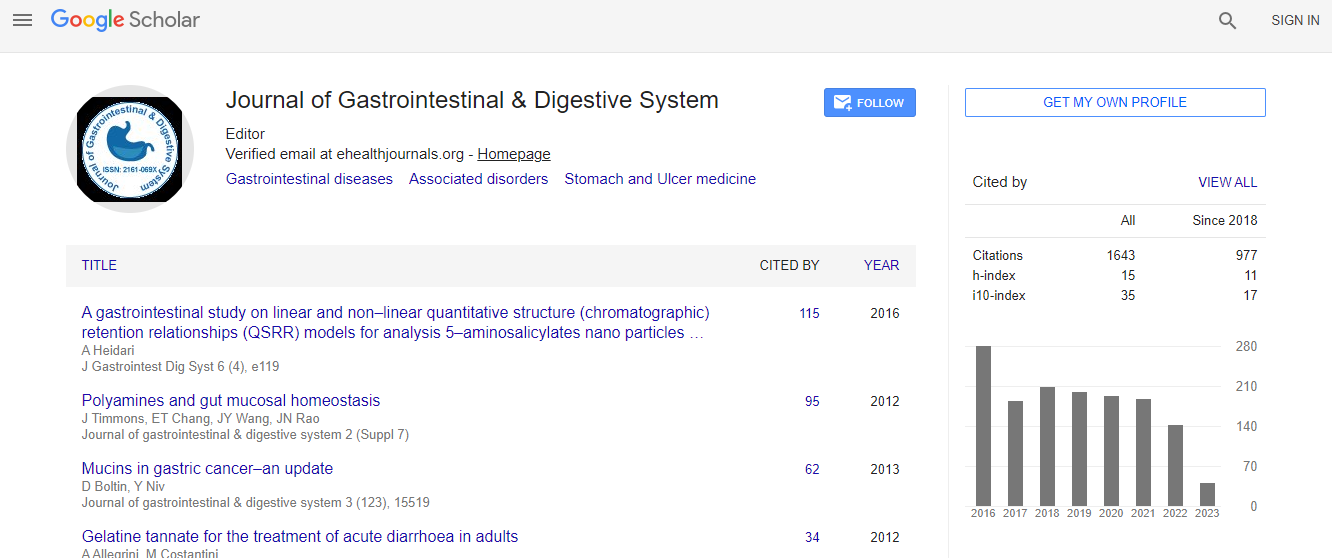
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Comparison of safety according to the timing of flumazenil administration in sedative endoscopy with midazolam: A single center, prospective, randomized and doubleblinded study
9th Euro Global Gastroenterology Conference
Hong Soon Jung
Catholic University of Korea, Korea
Posters & Accepted Abstracts: J Gastrointest Dig Syst
Abstract
Flumazenil is a benzodiazepine antagonist that blocks the central effects of benzodiazepines. Previous studies suggest that routine use of flumazenil after midazolam sedation for endoscopy shorten the recovery time, but there is no data on timing of flumazenil administration. In this study we assessed the timing of flumazenil in outpatients receiving intravenous sedation with midazolam. From 2012 August to 2015 July, 74 participants undergoing sedative diagnostic endoscopy with midazolam were prospectively included. They were randomized in a double-blind to receive either flumazenil immediately (immediately group) or 30 minutes later after the procedure (delayed group). Four hours after the sedative endoscopy each patient was interviewed by telephone to assess the primary end point, sleepiness and dizziness. 74 participants were analyzed, 37 with immediate administration of flumazenil and 37 with delayed administration. There were no significant differences between the two groups in age, sex, blood pressure, pulse rate, the average dose of midazolam, procedure duration, recovery time, previous history of endoscopy and satisfaction score from the patients. Also, there was no significant difference in sleepiness and dizziness between immediately group and delayed group. Sleepiness (immediately injected group=56.8%, delayed injected group=45.9%) and dizziness (immediately injected group=24.3% and delayed injected group=40.5%) was observed in almost half of the participants, in particular within two hours. In this prospective, randomized, double-blinded clinical trial showed that there is no significant difference in safety between two groups. Patient who received flumazenil should be monitored for 2 hours after its administration because of the potential for re-sedation.Biography
Hong Soon Jung has completed his MD from Catholic University of Korea and Post-doctoral studies from Catholic University Korea, College of Medicine. He is now working in the Division of Gastroenterology of Seoul St. Mary’s Hospital and Fellowship training.
Email: akaripio@naver.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi