Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
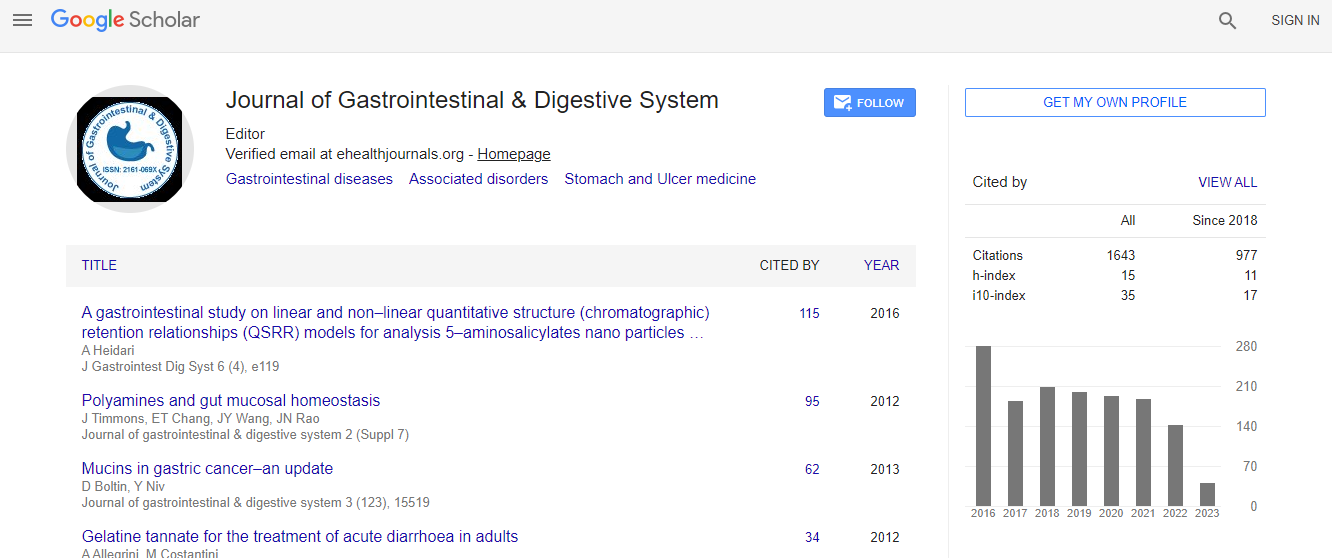
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Genetic and small-molecule modulation of STAT3 in mouse models of inflammatory bowel disease
14th Euro-Global Gastroenterology Conference
Prema Robinson
The University of Texas MD Anderson Cancer Center, USA
Keynote: J Gastrointest Dig Syst
Abstract
Background & Aims: Ulcerative colitis (UC) and Crohn’s disease (CD) are Inflammatory bowel diseases (IBD) of unclear etiology that cause substantial morbidity and predispose to Colorectal-cancer (CRC). There are two isoforms of STAT3-α and β; STAT3α is pro-inflammatory and anti-apoptotic, while STAT3β has opposing-effects on STAT3α. We determined the contribution of STAT3 to UC and CD pathogenesis by comparing disease severity caused by dextran sodium sulfate (DSS; UC model) or 2, 4, 6-trinitrobenzenesulfonic acid (TNBS; CD model) in mice expressing only STAT3α (Δβ/Δβ) and in wild-type (WT) mice treated with TTI-101, a small-molecule inhibitor of both isoforms of STAT3.Methods: Seven days following administration of DSS in drinking water or two days following a single intra-rectal administration of TNBS, Δβ/Δβ mice, cage-control (+/+) mice and WT mice given TTI-101 or vehicle were examined for IBD manifestations; their colons were evaluated for apoptosis of CD4+ T cells, levels of STAT3 activation, IL-17A protein expression and transcriptome alternations.
Results: IBD manifestations were increased in Δβ/Δβ transgenic vs. cage-control WT mice and were accompanied by decreased apoptosis of colonic CD4+ T cells. Complementing and extending these results, TTI-101 treatment of WT mice prevented IBD, markedly increased apoptosis of colonic CD4+ T cells, reduced colon levels of IL17A-producing cells and down-modulated STAT3-gene targets involved in inflammation, apoptosis-resistance and colorectal-cancer metastases.
Conclusion: STAT3, especially in CD4+ T cells, contributes to the pathogenesis of UC and CD and its targeting may provide a novel approach to disease treatment.
Biography
Prema Robinson is an Associate Professor since 2015 in the Department of Infectious Diseases, Infection Control and Employee Health, Division of Internal Medicine in The University of Texas MD Anderson Cancer Center, USA.
E-mail: PRobinson1@mdanderson.org

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi