Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
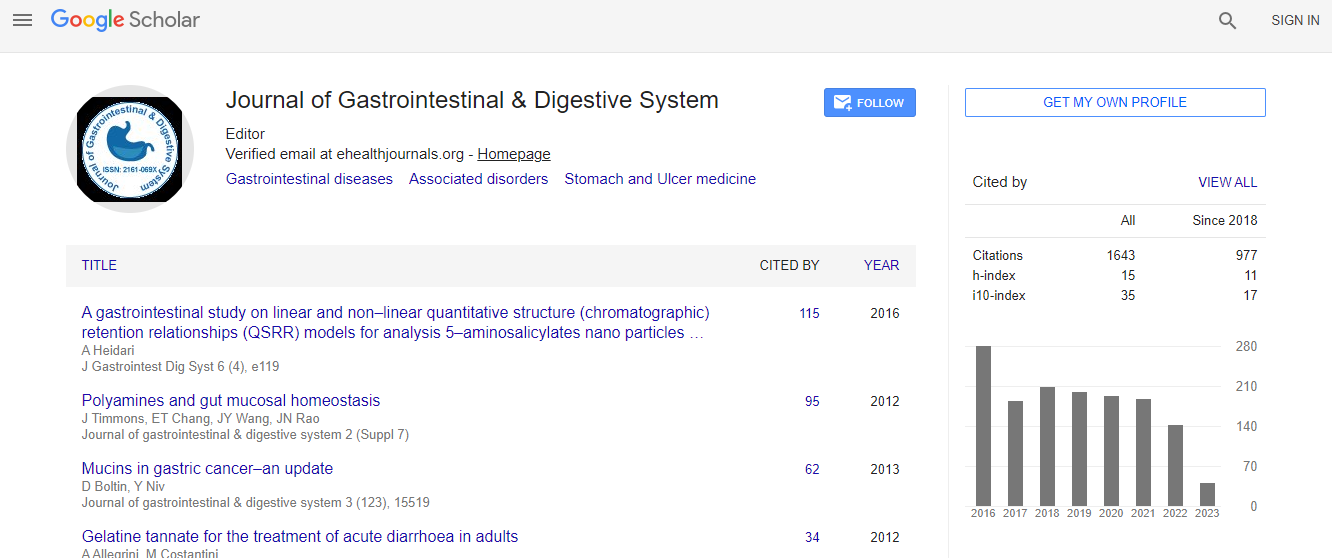
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Randomized clinical trial (RCT) of IFN-intolerant/ineligible (II) GT1 patients (TN or TE) including LDV/SOF failures with NS5A RAVs using SMV+SOF+/-RBV for 12/24 weeks
8th International Conference on Clinical Gastroenterology & Hepatology
Pockros P J, Skillin C, Box T and Agarwal R
Scripps Translational Science Institute, USA Scripps Clinic- Scripps Translational Science Institute, USA University of Utah, USA DIcahn School of Medicine at Mt. Sinai, USA
Posters & Accepted Abstracts: J Gastrointest Dig Syst
Abstract
Introduction: In a real-world database, retreatment of LDV/SOF failures with SMV+SOF +/- RBV for 12 or 24 weeks achieved 86% SVR [6/7], however, there was no randomization and it is unknown whether RBV was beneficial. A previous RCT of SMV+SOF for 12 weeks achieved 96% SVR [24/25] in patients with NS5A RAVs who had not failed prior DAA therapy. Methods: We initiated an RCT in Dec 2014 using SMV 150 mg QD+SOF 400 mg QD for 12 (non-cirrhotic) or 24 (cirrhotic) weeks in GT-1 II patients. Patients were randomized 1:1 to receive SMV+SOF+RBV 1,000-1,200 mg/d or -RBV and followed at 4 weeks intervals with HCV RNA, liver tests, PEs and AE monitoring. The protocol was amended in June 2015 to include patients who failed 12 weeks of LDV/SOF and had NS5A RAVs (28, 30, 31, 32, or 93). All patients were HIV- and HbsAg- and had compensated GT-1 HCV. We defined mITT as those patients who completed the trial and we used a 2-tailed t-test for RBV vs. no RBV groups. Results: 26 patients were screened and 24 (19 G1a and 5 G1b) were randomized from 3 US centers. 22 patients were non- Hispanic white (10M, 12F) with a mean age of 53 years. Patients qualified as II because of depression (11), prior intolerance (4), cardiomyopathy (1), and other (5). 3 patients had failed prior LDV/SOF therapy, and 8 were treatment experienced (TE) with PEG/RBV. 6/24 patients were F3-F4 and 4 of those received 24 weeks therapy. 4/15 GT1a patients had Q80K RAVs and all 3 patients who previously failed LDV/SOF had NS5A RAVs at Y93. Overall, 87.5% (21/24) of patients achieved SVR12 by ITT (mITT=91.3% [21/23]). 1 patient was LTFU, 2 relapsed and both had Q80K RAVs (1 previous LDV/SOF failure with 2 RAVs and 1 cirrhotic patient treated for 23 weeks). Of the 10 patients receiving RBV, 2 relapsed (SVR=80%). Of the 14 patients not receiving RBV, 0 relapsed, and 1 was LTFU (SVR=92.8%; p=NS). 2/3 LDV/SOF failures received RBV and 1 of these relapsed. Dose reduction or early D/C of RBV was necessary due to AEs in 4/10 and 1/10 patients. 2/10 patients receiving RBV had dose reductions due to a fall in Hgb>3 g/dl but no patient had Hgb<10 g/dl. AEs were all grades 1-2 and were nausea or other GI (13), fatigue (11), headache (5), and photosensitivity related to SMV (4). Fatigue and GI were more common in patients who received RBV. Conclusions: II patients treated with 12/24 weeks of SMV/SOF+/-RBV had an overall mITT 91.3% SVR12. Relapse was seen in half of the in patients with pre-existing Q80K RAVs (2/4). RBV did not improve SVR rates although it did increase AEs and required dose reduction in some patients. SMV+SOF+/-RBV was effective in 2/3 patients who failed LDV/SOF and had Y93 NS5A RAVs and may be considered as an alternative for these patients.Biography
Chase Skillin completed his bachelor’s degree at the age of 22 years from the University of California, Los Angeles. Since graduating, he has been a Clinical Trial Coordinator for Scripps Clinic in San Diego. His studies primarily focus on Hepatitis C, Cirrhosis, and Hepatocellular Carcinoma. He has contributed to several abstracts, textbook chapters, and presentations over the past year. Chase is currently applying to medical schools across the United States, and hopes to formally begin his medical career in the fall of 2017.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi