Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
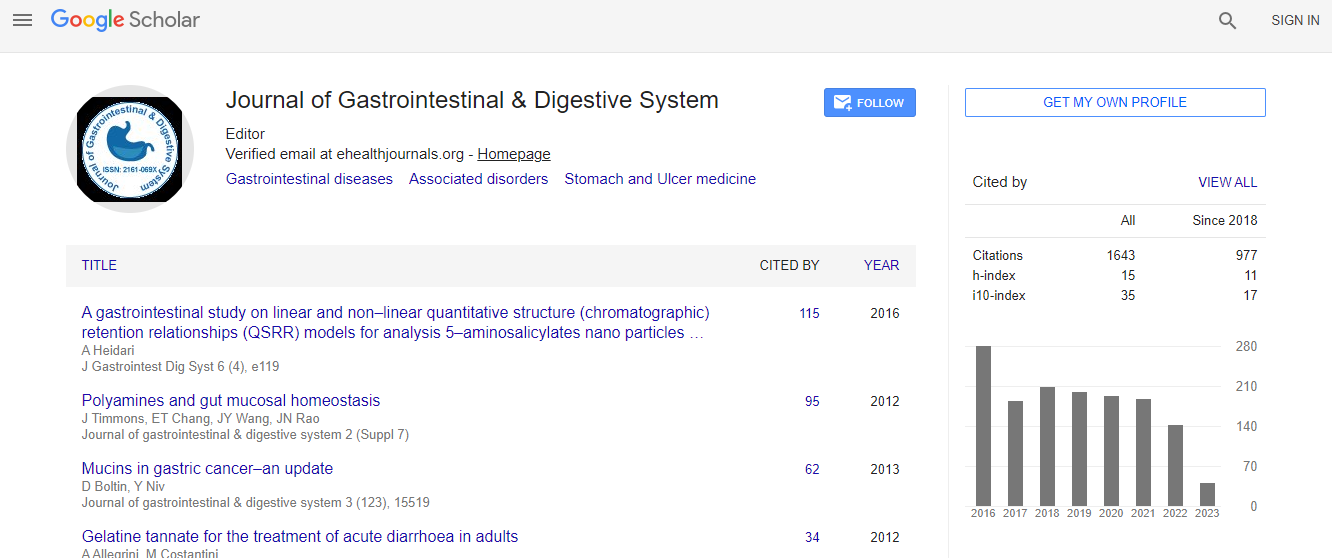
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Regulation of calcium-sensing receptor expression by vitamin D and proinflammatory cytokines in colorectal cancer
International Conference on Gastrointestinal Cancer and Therapeutics & 4th World Congress on Digestive & Metabolic Diseases & 26th Annual Congress on Cancer Science and Targeted Therapies
Irfete S Fetahu
Brigham and Womens Hospital, USA
ScientificTracks Abstracts: J Gastrointest Dig Syst
Abstract
Anti-proliferative effects of calcium in the colon are partially mediated by the calcium-sensing receptor (CaSR). The CaSR gene is located in the chromosomal region 3q13.3-21 and is constituted of 2 non-coding and 6 coding exons. The transcription of CaSR is under the control of promoters 1 and 2, which yield different transcripts containing either of the untranslated exons 1A or 1B. The expression of CaSR decreases during colorectal tumorigenesis and the underlying mechanisms regulating its expression are poorly understood. The CaSR promoters 1 and 2 harbor vitamin D elements responsive to 1,25-dihydroxyvitamin D3 (1,25-D3) and NF-kB, STAT, and SP1 binding sites accounting for responsiveness to proinflammatory cytokines. Vitamin D is known for its pro-apoptotic and anti-inflammatory effects in CRC. We have previously shown that high dietary doses of vitamin D prevented the formation of chemically-induced preneoplastic lesions in a mouse model. On contrary, increased production of proinflammatory cytokines, including TNFa and IL-6 has been reported in inflammatory bowel disease and CRC patients. The impact of 1,25-D3 and proinflammatory cytokines (IL-6, TNFa) on CaSR expression in colon cancer cells is not well known. This led us to hypothesize that they might regulate the expression of the CaSR. We performed a study where we analyzed the role of 1,25-D3, TNFa, and IL-6 on CaSR expression in Caco2/AQ, a well differentiated and Coga1A, a moderately differentiated colorectal cancer cell line. Over the time course of 48 hours, we observed upregulation of the CaSR expression in both cell lines. The well-differentiated Caco2/AQ cells responded with higher induction of the CaSR expression than Coga1A cell line. Moreover, we observed a reduction in the expression of the proliferation markers cyclin D1, minichromosome maintenance 2&7 (MCM2 and MCM7) in Caco2/AQ cells. These studies suggested that in addition to the direct role of vitamin D in chemoprevention, it can also upregulate the expression of the CaSR, thereby linking together the chemopreventive actions of vitamin D and calcium in colorectal tumors. In the less differentiated cell line Coga1A, TNFa had a pronounced effect in increasing the expression of the CaSR, indicating that CaSR might serve as a defense mechanism towards inflammatory stimuli. Additionally, in both cell lines, IL6 induced the expression of the CaSR. Interestingly, in the well-differentiated Caco2/AQ cells treated with 1,25-D3 counteracted the effects of IL6 and TNFa. This could be attributed probably to the anti-inflammatory role of the 1,25-D3. This study provides further evidence towards the protective role of the CaSR in colon cells against inflammation.Biography
Irfete S Fetahu received her PhD at the Medical University of Vienna (Austria) under the supervision of Prof Enikö Kallay as a Marie Curie Early Stage Researcher. During this time she was the recipient of several national and international awards, including fellowships from the European Association for Cancer Research and the Federation of European Biochemical Societies. She was a board member of the Young Scientist Association of the Medical University of Vienna. Following her PhD graduation, she started her postdoctoral fellowship at the Harvard Medical School (USA), where she is currently working in uncovering the aberrancies in the epigenomes of Alzheimer’s disease and melanoma. She is also a board member of the Harvard Medical Postdoc Association. She currently serves as an Ambassador of the European Association for Cancer Research.
E-mail: ifetahu@bwh.harvard.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi