Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
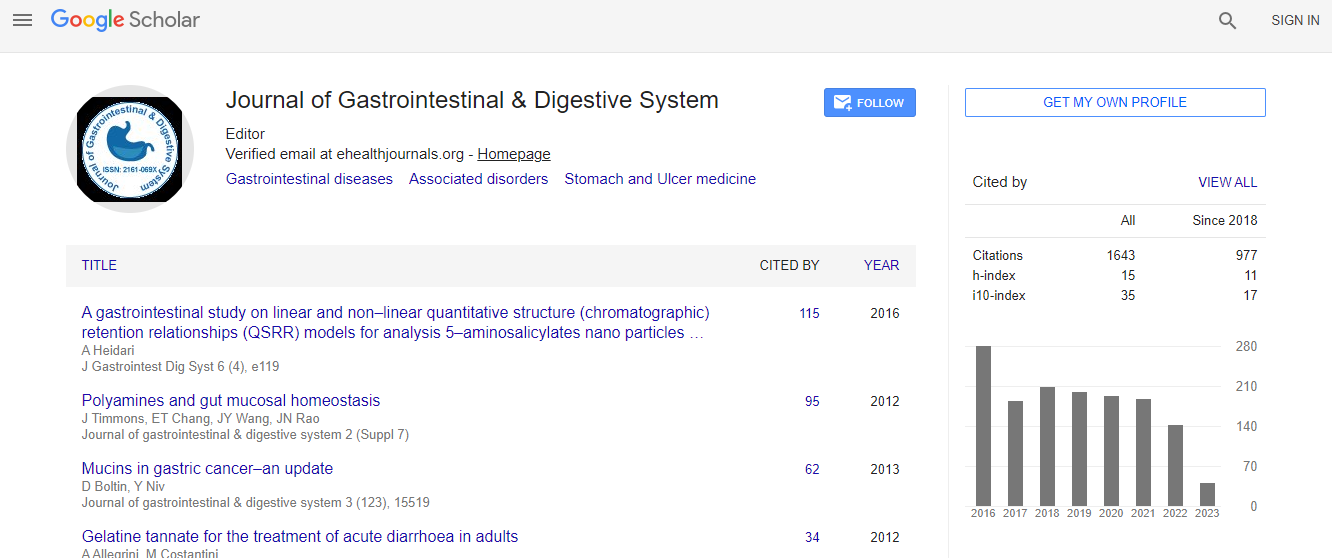
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Serum-derived bovine immunoglobulin in the management of chronic gastrointestinal disease
International Conference and Exhibition on Gastrointestinal Therapeutics
Larry I Good
1Good Pharmaceutical Development Company, USA 2South Nassau Communities Hospital, USA
ScientificTracks Abstracts: J Gastrointest Dig Syst
Abstract
Serum-derived Bovine Immunoglobulin (SBI) possesses the entire immune experience of cows. Oral administrations of SBI prepared from the serum of lots of 3000 cows at the time of slaughter has been demonstrated to be effective in the management of HIV enteropathy, diarrhea predominant irritable bowel syndrome, ulcerative colitis, Crohn��?s Disease, pouchitis and C. difficle colitis. Data will be presented of clinical outcomes from randomized, double blind trials and observational studies. Mechanism of action of this product includes binding of pro-inflammatory intra-luminal bacterial degradation products, pro-inflammatory cytokines, bacterial endotoxins and tightening of tight junctions between intestinal epithelial cells. The excellent safety profile and GRAS status of this medical food product will be discussed.Biography
Larry I Good has been a practicing Gastroenterologist since 1978. He has graduated Colgate University Magna Cum Laude in 1969 and received his MD with Alpha Omega Alpha status from the Medical University of South Carolina in 1973. He was a fellow trained in Gastroenterology from 1976-78 at the University of Pennsylvania. He has served as the Director of Liver Diseases at Nassau County Medical Center and was for many years Chief, Department of Medicine at South Nassau Communities Hospital. He is an Assistant Clinical Professor of Medicine at SUNY Stony Brook. He was the Chief Medical Officer at Ritter Pharmaceuticals in Los Angeles and his current clinical research activities involve the microbiome, inflammatory bowel disease, irritable bowel syndrome and the application of orally administered gamma globulin to patients with acute and chronic gastro-intestinal disorders. He has Founded Good Pharmaceutical Development Co., LLC, in 2014. He was recently appointed CEO of Compassionate Care Center of New York. He was appointed as an Editor of BMC Gastroenterology, a prestigious on line peer reviewed medical journal. He is a Member of the Speakers Bureau for Entera Health, Abbvie and Cubist Pharmaceuticals.
Email: good1b@optonline.net

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi