Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
Google Scholar citation report
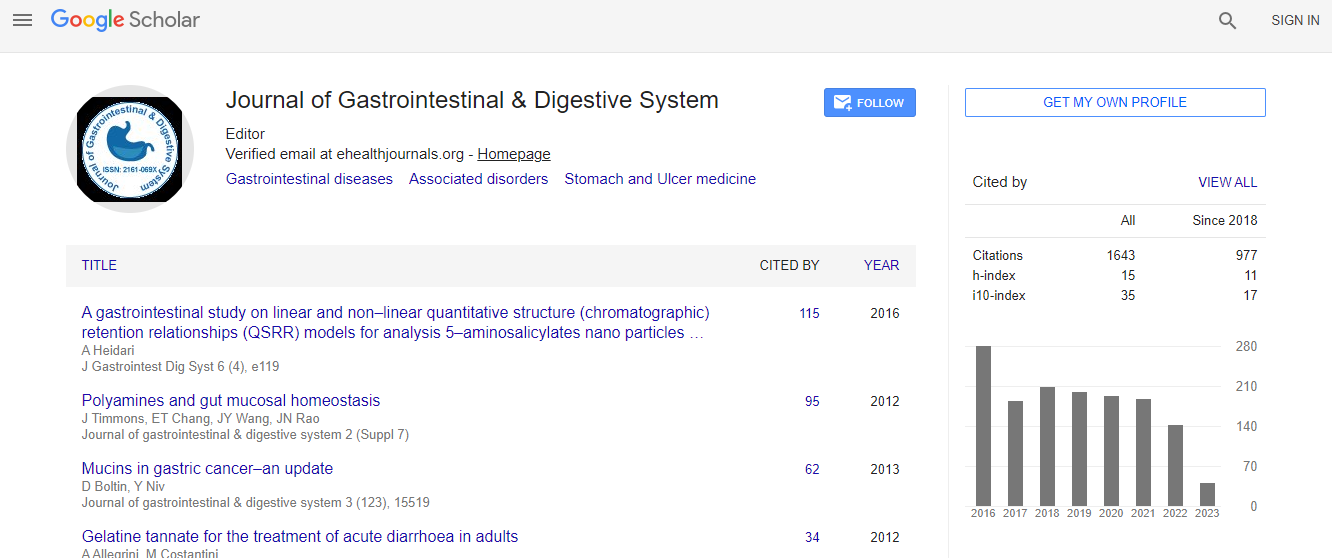
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Subsequent clinical exocrine pancreatic insufficiency, a disease requiring recognition and therapy
11th Global Gastroenterologists Meeting
David H Van Thiel
Advanced Liver and Gastrointestinal Disease Center, USA Stronger Hospital of Cook County, USA
Keynote: J Gastrointest Dig Syst
Abstract
Introduction: The criteria utilize for the identification of the diagnoses of exocrine pancreatic insufficiency (EPI) was established in the late 1950s and early 1960s. The possible criteria was the finding of high-grade steatorrhea equal to or greater than 20 g of fecal fat per day collected over a 72 hour. While the patient was ingesting a diet with a minimum of 100 g of fat daily. It was reported that this criterion was met when more than 90% of the function of the exocrine pancreas was lost. This requirement for the diagnoses of exocrine pancreatic insufficiency is difficult to achieve because the test is cumbersome and poorly accepted by patients, physicians and laboratory personnel. A multitude of alternative tests have been developed, but none of them has achieved universal acceptance and/or availability. The detection and quantification of exocrine pancreatic enzymes in stool has been evaluated for this purpose, but all of the enzymes with the exception of pancreatic elastase are destroyed during the transit through the intestine, making them unacceptable as an diagnostic tool. Pancreatic elastase measured in stool is greater than 200 ng/g stool and normal individuals. Those with less than 200 mg greater than 100 ng per male have subclinical pancreatic insufficiency. Those with less than 100 ng per male have overt pancreatic insufficiency as defined by the original definition for the detection of pancreatic insufficiency. Aim: Identify an individuals with subclinical exocrine pancreatic insufficiency having stool he less based determination was less than 201 greater than 100 and evaluate the response to treatment with pancreatic enzyme supplementation. Methods: #1 Consecutive patients seen in outpatient gastroenterology clinic were evaluated. #2 Stool elastase was measured in all enabling the segregation of the subjects into 3 distinct groups consisting of normal individuals( elastase >200, those with minimal to moderate pancreatic insufficiency less than 200 but greater than 100 ng elastase/gram of stool and those with overt pancreatic insufficiency having less than 100 ng/g pancreatic elastase in stool. #3 symptoms of abdominal pain, cramps, bloating, gas, flatulence, diarrhea, large, bulky stools, foul smelling stools, I requirement for double flushing, were each evaluated on a 0-10 scale@each clinic visit over a 6 month period. #4 6 laboratory parameters consisting of hemoglobin, BUN, creatinine, albumin, levels of vitamin A, vitamin D, and vitamin E were recorded and each clinic visit. Results: Symptoms in most patients and groups to ON 3 improved with time and continued utilization of pancreatic enzyme replacement. The response occurred earlier in those with mild to moderate pancreatic insufficiency as compared to those with overt pancreatic insufficiency and no improvement was noted in those with normal levels of pancreatic elastase in stool. Conclusions: #1 individuals with unexplained abdominal symptoms should be evaluated utilizing a stool elastase determination of the presence of pancreatic insufficiency. #2 individuals with pancreatic elastase in stool less than the lower limit of normal, but greater than 100 ng per gram stool have pancreatic disease that responds to therapy achieving a symptom response pattern, similar to that of normal individuals having a normal stool elastase determination greater than 200 ng/g stool. Those and groups 3 with overt pancreatic insufficiency response lower to a degree less than that observed by those in group to with subclinical pancreatic insufficiency. Summary: Subclinical exocrine pancreatic insufficiency response to therapy consisting of esophagitis pancreatic enzyme supplementation and represents the clinical disease prosess that can and should be treated.Biography
David Van Thiel is a Gastroenterologist in Berwyn, Illinois. He is affiliated with multiple hospitals in the area, including Rush Oak Park Hospital and Rush University Medical Center. He completed his Medical degree from David Geffen School of Medicine at UCLA and has been in practice for 39 years. He is one of the 21 doctors at Rush Oak Park Hospital and one of 25 doctors at Rush University Medical Center who has specialization in Gastroenterology. He completed his Graduation from University of California at Los Angeles. He has obtained board certification from the member board for Internal Medicine and Hepatology.
Email: dvanthiel@dr.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi