Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
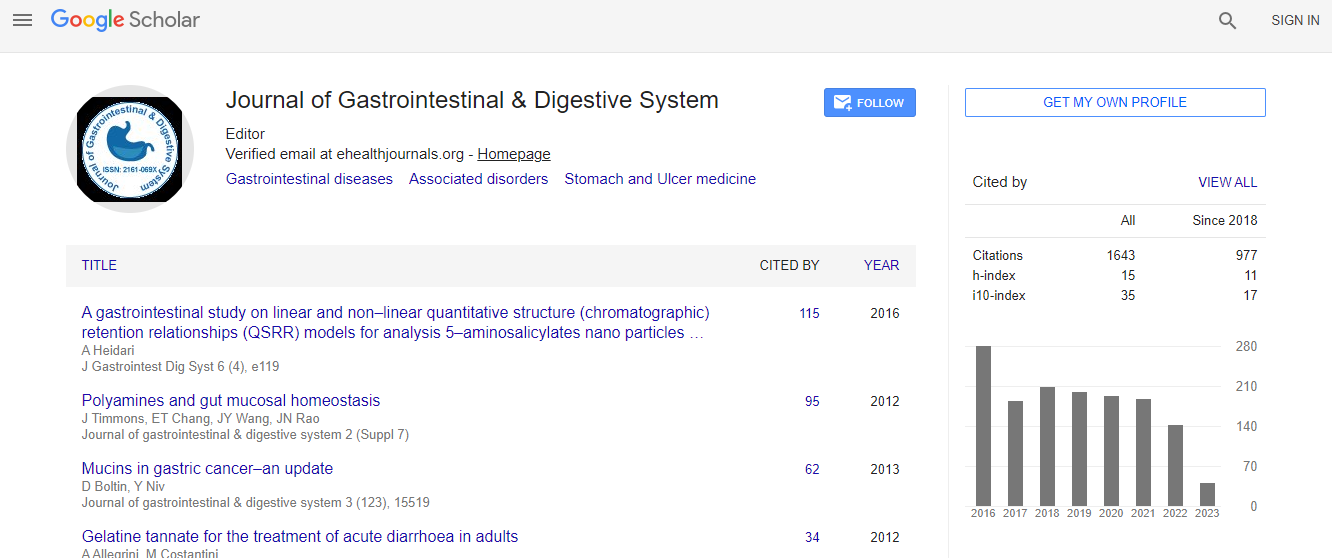
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
The importance of neuronal serotonin in colonic motor patterns
12th Euro-Global Gastroenterology Conference
Terence K Smith
University of Nevada, USA
Keynote: J Gastrointest Dig Syst
Abstract
5-hydroxytryptamine (5-HT, serotonin) has two sources in the gut, descending myenteric 5-HT interneurons (~5% of all 5-HT) and enterochromaffin (EC) cells (~95% of 5-HT). 5-HT is synthesized by the rate limiting enzyme tryptophan hydroxylase (TPH); TPH1 in EC cells and TPH2 in EC cells. In the colon, these two pools of 5-HT appear to be connected by intrinsic primary afferent neurons (IPANs) that have sensory endings in the mucosa and synapse with 5-HT interneurons. The prominent inhibitory effects of 5-HT1A/2B/3/4 and 7 receptor antagonists on colonic motility suggests that many of these receptors are on various cell types including glia that are largely activated by 5-HT interneurons. Major motility patterns in the colon involving 5-HT include tonic inhibition of the muscle layers interrupted by rhythmic peristaltic waves called colonic migrating motor complexes (CMMCs), and secretomotor activity and possibly blood flow. In large mammals (e.g. dog, pig and human colon) 5-HT neurons occur in both the myenteric plexus and the extensive Henle��?s plexus, that synapse with secretomotor neurons in Meissner��?s plexus. Henle��?s plexus also provides a motor innervation to submucosal pacemaker cells that generate slow waves. Some 5-HT neurons also project to prevertebral ganglia suggesting they are viscerofugal neurons that they can activate postganglionic sympathetic neurons, whose terminals surround 5-HT neurons. We propose that asynchronous firing in 5-HT neurons excite inhibitory motor neurons (IMNs) to generate tonic inhibition and suppress pacemaker activity. In contrast, 5-HT released from EC cells activates IPANs that synchronizes 5-HT neurons. Synchronized firing of 5-HT neurons likely activates glial cells, which release PGE2, that switch off IMNs and remove tonic inhibition. Synchronized 5-HT neurons also likely generate a slow excitatory postsynaptic potential (sEPSP) in IPANS by activating 5-HT7 receptors that generate CMMCs that in turn excite excitatory motor neurons and pacemaker cells.Biography
Terence Smith is Professor in the department of Physiology and Cell Physiology at the University of Nevada-Reno, USA, where he is the Director of the Dynamic Imaging Core. He received his PhD in Neuropharmacology/Electrophysiology from Monash University, Victoria, Australia under Professors Mollie Holman and David Hirst who first classified enteric neurons electrically. His interests have focused on how the enteric nervous system regulates gut pacemakers, and more recently optogenetics to study gut motility.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi