Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
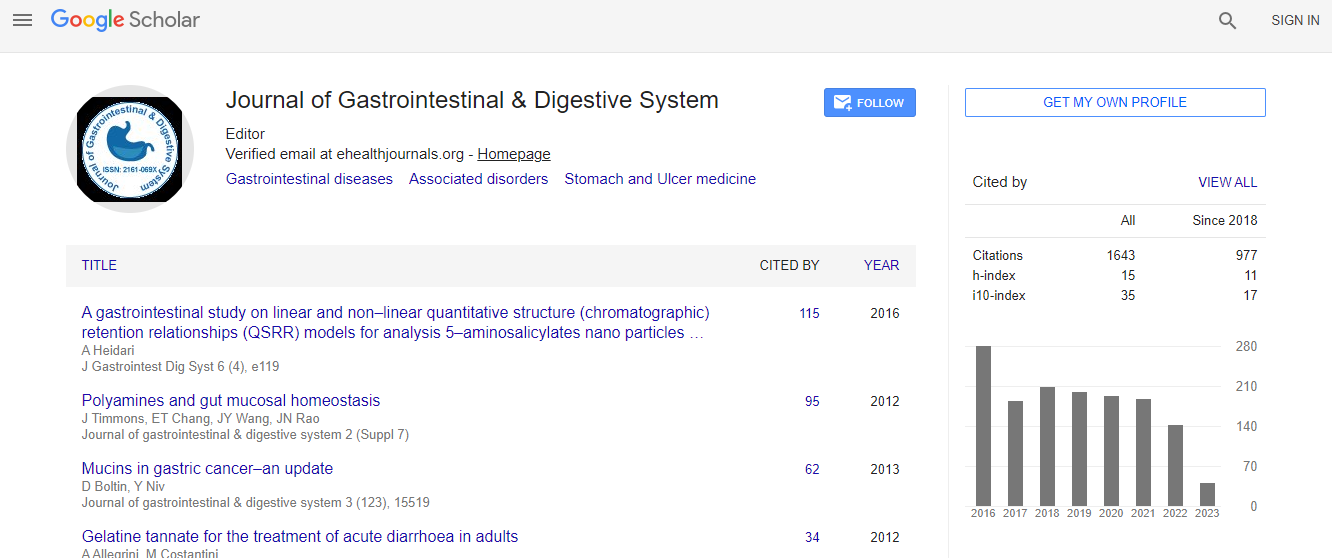
Google Scholar citation report
Citations : 2091
Journal of Gastrointestinal & Digestive System received 2091 citations as per Google Scholar report
Journal of Gastrointestinal & Digestive System peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Uncovering the role of planar cell polarity during intestinal morphogenesis
14th International Conference on Clinical Gastroenterology and Hepatology
Abilasha Rao-Bhatia, Sabrina Coquenlorge-Gallon, Wen-Chi Yin, Xiaoyun Zhang, Min Zhu, Chi-Chung Hui, Sevan Hopyan, Helen McNeill and Tae-Hee Kim
The Hospital for Sick Children, CanadaUniversity of Toronto, CanadaWashington University School of Medicine, USA
ScientificTracks Abstracts: J Gastrointest Dig Syst
Abstract
The mammalian intestine is lined with millions of finger-like projections, termed villi. These villi are critical for maximizing nutrient absorption, digesting food and serving as a barrier from the harsh luminal environment. As such, compromised villi can lead to serious diseases including malabsorption, short bowel syndrome, celiac, and others. Although villi are precisely patterned by a network of signaling pathways during embryogenesis, it remains unclear as to how these signals translate into distinct morphogenetic transformations. Previous studies attribute the formation of mesenchymal clusters distinguished by Hedgehog (Hh) activation, as critical for epithelial rearrangement into villi. However, the mechanisms of Hh-mediated clustering remain unknown. Our RNA-seq analyses coupled with GLI2 (Hh-transcriptional activator) ChIP-seq reveal that planar cell polarity (PCP) genes such as Fat4, Dchs1 and Vangl2 are putative direct targets of Hh in the gut mesenchyme. Notably, mice deleted and/or mutated for these genes exhibit severe villus fusions and fail to form mesenchymal clusters, demonstrating for the first time the importance of PCP in villification. Furthermore, genetic interaction studies reveal that the core PCP axis (Vangl2) acts in parallel to the atypical cadherin axis (Fat4, Dchs1) in maintaining PCP. Additionally, ongoing live-imaging of mutant villification ex vivo will uncover the types of mesenchymal cell behavior that is required for clustering and subsequent villus formation. Together, we introduce Hh-activated stromal PCP as novel mechanisms required for the morphogenetic events seen during villification.Biography
Abilasha Rao-Bhatia is currently a PhD candidate under the supervision of Dr Tae-Hee Kim, Scientist at The Hospital for Sick Children and Assistant Professor of the Molecular Genetics Department at the University of Toronto. She is part of an interdisciplinary team dedicated to understanding developmental and stem cell biology of the gastrointestinal system. Prior to this, she completed her undergraduate studies at the University of Waterloo with a degree in Honour’s Biology Cooperative studies. Her passion for biomedical research began here as a Co-op student in Dr John Dick’s laboratory studying the stem cell origins of acute myeloid leukemia relapse.
E-mail: abi.rao@mail.utoronto.ca

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi