Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
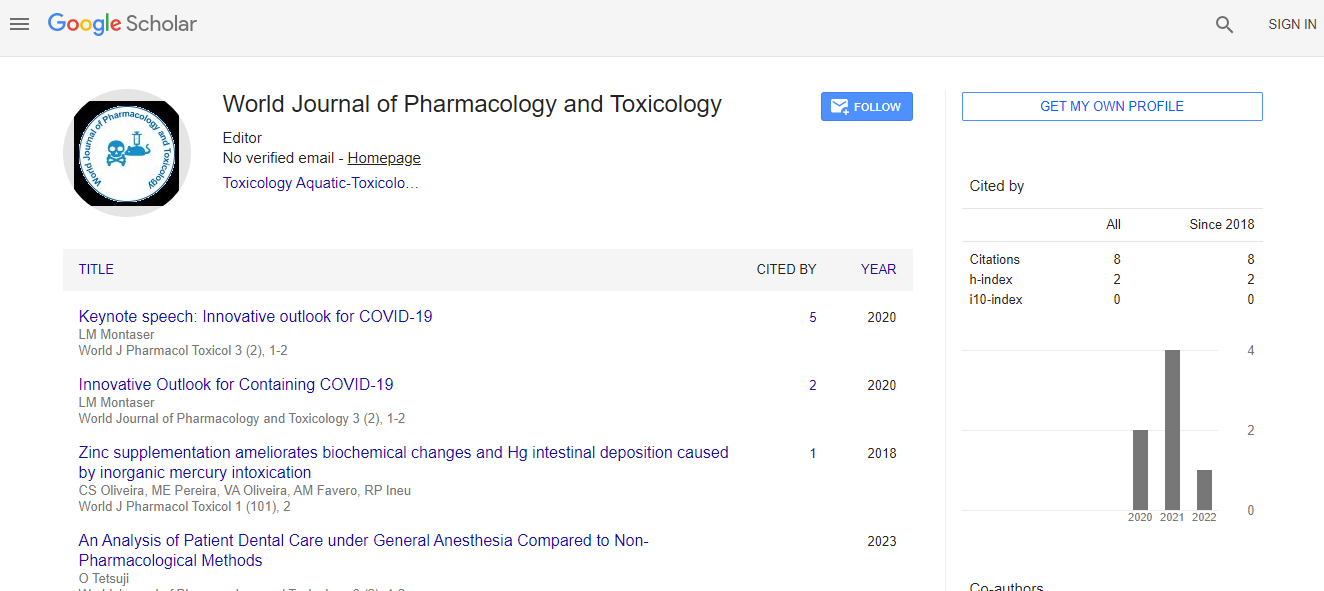
Google Scholar citation report
Citations : 8
World Journal of Pharmacology and Toxicology received 8 citations as per Google Scholar report
Indexed In
- Google Scholar
- ICMJE
Useful Links
Share This Page
Varun Ahuja

Varun Ahuja
Lupin Limited
India
Biography
Dr. Varun Ahuja (M.V.Sc., Ph.D) is a Scientist and American Board certified toxicologist (DABT) working in Drug Safety Assessment Department, Novel Drug Discovery & Development, Lupin Ltd., India. He accomplished his Master’s degree in Veterinary Pharmacology & Toxicology from G.B.Pant University, Pantnagar, India. He received doctoral degree from Department of Veterinary Medicine, Freie University, Berlin, Germany. Varun pursued post-doctoral work from Charité Medical University, Berlin. Later, he worked as a Study Director, Toxicology, at BSL Bioservice Scientific Laboratories, Munich, Germany. During his research career, he received fellowships from various prestigious Indian (ICAR-Indian Council of Agricultural Research) and German (DAAD-German Academic Exchange Services) organizations. In a scientific career spanning more than 15 years, Varun has got experience working in various fields such as pharmacokinetics, in vitro and in vivo toxicity testing, safety pharmacology, risk assessment and drug discovery and development. Presently, he is interested in working in the field of preclinical safety assessment of drugs and establishing new techniques/models for the same. He has about 75 publications in the field of biomedical sciences. He has also presented papers/delivered talks in various meetings in India/Europe/USA. Varun has extensive experience with nonclinical safety testing of drugs from various therapeutic classes.
Research Interest
Preclinical Toxicity testing; Safety Pharmacology; Drug Discovery and Development; Risk Assessment

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi