Research Article Open Access
Likelihood of Smallpox Recurrence
Jennifer Gaudioso1, Tim Brooks2, Katsuhisa Furukawa3, Daniel O Lavanchy4, David Friedman5 and Erik D Heegaard6*
1International Biological Threat Reduction Program, Sandia National Laboratories, NM, US
2Novel & Dangerous Pathogens Department, Health Protection Agency, Centre for Emergency Preparedness & Response, Salisbury, UK
3Research Institute of Science and Technology for Society, Japan Science and Technology Agency, Tokyo, Japan
4Former member of the Global Alert and Response Team, World Health Organization, Geneva, Switzerland
5The Institute for National Security Studies, Tel Aviv, Israel
6Biosecurity Institute, Holte, Denmark
- *Corresponding Author:
- Erik D. Heegaard
Biosecurity Institute
Skovlybakken 13 2840 Holte, Denmark
Tel: +45 20 81 02 82
E-mail: heegaard@biosecurityinstitute.dk
Received Date: March 05, 2011; Accepted Date: May 04, 2011; Published Date: May 18, 2011
Citation: Gaudioso J, Brooks T, Furukawa K, Lavanchy DO, Friedman D et al. (2011) Likelihood of Smallpox Recurrence. J Bioterr Biodef 2:106. doi: 10.4172/2157-2526.1000106
Copyright: © 2011 Gaudioso J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Bioterrorism & Biodefense
Abstract
The 30 year anniversary of the eradication of smallpox was recently celebrated and represents a major achievement in international public health. However, the likelihood of re-introduction of eradicated diseases is expected to evolve with time, and warrants continued assessment. Using influence diagrams to structure the analysis, this paper seeks to systematically examine the various pathways that could lead to accidental or deliberate introduction of a novel or eradicated pathogen using smallpox as an example. The accidental reintroduction of smallpox may occur through three main pathways: Biosafety incident at a known repository, biosafety incident outside of a known repository, and environmental resurrection. The deliberate reintroduction can also be subdivided into three main pathways: Biosecurity incident at a known repository, illicit state biological weapons program, and synthesis using the tools of modern biotechnology. We conclude that the likelihood of recurrence of an eradicated agent, such as smallpox, is small, but ultimately unknown and expectedly increases with time primarily due to the rapid advancement of biotechnology.
Introduction
Infectious disease threats are a continuous challenge for society, and may originate from both naturally-occurring and deliberately introduced pathogens and toxins. Eradicated or novel pathogens pose a particular preparedness challenge for decision makers who are faced with the question of allocating scarce public resources to address diseases that are not a current threat. Of particular concern is the possible reintroduction of eradicated pathogens including 1918 Spanish flu (eradicated for 90 years), SARS (no natural outbreaks since 2003), and polio (soon-to-be eradicated). Smallpox virus is the most widely considered example, although the natural public health concerns of smallpox were eliminated through a global eradication program. Exhibiting unique non-proliferation security challenges, some experts remain concerned about the possibility that smallpox could reappear as a biological weapon [1].
The consequences of a smallpox outbreak have been documented throughout history [2], and more recently assessed in a modern globalized environment through a variety of epidemiological and mathematical models [3] and exercises [4]. In recognition of the possible serious devastation, many countries around the world are actively working to strengthen their bio preparedness in order to be able to respond. However, if there is no likelihood of introduction of a novel or eradicated pathogen, then, regardless of the severity of the consequences, there is no risk and, hence, no policy actions are required. Yet there is little to no structured assessment of the likelihood of inadvertent or deliberate reintroduction of smallpox virus or other novel or eradicated pathogens in the publicly available literature. To most appropriately allocate resources, decision makers need to understand both components of risk, i.e. the well-publicized consequences as well as the likelihood of recurrence (i.e. simply reintroduction by any accidental or deliberate means, not taking into account the probability of effective dissemination). If the likelihood of accidental or deliberate introduction of a novel or eradicated pathogen is nonzero, then policy makers must determine how to best manage risks. Both preventive and consequence mitigation measures can be considered in countering these risks. The publications and preparedness table top exercises for smallpox illustrate the range of consequence mitigation measures decision makers can consider. Recognizing what factors influence the likelihood of reoccurrence will yield crucial information about possible prevention control points and whether additional consequence mitigation measures are warranted.
This paper seeks to systematically examine the various pathways that could lead to accidental or deliberate introduction of a novel or eradicated pathogen. To focus the discussion, smallpox will be used as the example throughout the paper. However, the same analytical framework could be applied to other pathogens of concern. The accidental reintroduction of smallpox may occur through three main pathways: Biosafety incident at a known repository, biosafety incident outside of a known repository, and environmental resurrection (Table 1, Categories A-C). The deliberate reintroduction can also be subdivided into three main pathways: Biosecurity incident at a known repository, illicit state biological weapons program, and creation of the virus using the tools of modern biotechnology (Table 1, Categories D-F). Reliable data is not available to enable a probabilistic assessment of the likelihoods of the six main pathways. Thus, this paper utilizes influence diagrams to structure the analysis.
| Mode of introduction | Category | Mechanism | Conclusions of Pathway Analyses | ||
|---|---|---|---|---|---|
| Relative likelihood (low/medium/high) | Trend | ||||
| Accidental reintroduction | Biosafety incident at a known WHO repository | A | Laboratory acquired infection or accidental release from WHO repository (restricted to CDC and Vector) | Low | Decreasing |
| Biosafety incident outside of a known repository | B1 | Unknown, archived and untested/misdiagnosed clinical sample | Low | Decreasing | |
| B2 | Unknown, archived and tested, but unlabeled sample | Low | Decreasing | ||
| B3 | Rediscovered sample labeled as smallpox | Low | Decreasing | ||
| Environmental resurrection / mutation | C1 | Archived outside of the lab | Low | Decreasing | |
| C2 | Natural orthopox mutation into virus that can cause smallpox-like illness | Low | Constant | ||
| Deliberate reintroduction | Biosecurity incident: theft from a known repository | D1 | Insider theft | Medium | Constant |
| D2 | Outsider theft | Medium | Decreasing | ||
| D3 | Collusion | Medium | Constant | ||
| Biotechnology | E1 | De novo synthesis | Medium | Increasing | |
| E2 | Deliberate mutation of a related orthopox virus | Medium | Increasing | ||
| State illicit weapons program | F1 | State retained it post-eradication and post-consolidation of smallpox samples into 2 repositories | Low | Constant * | |
| F2 | State acquired it post eradication: accidental discovery, acquired from other state program, theft from repository, biotech | Low | Constant+ | ||
The table lists potential pathways for accidental or deliberate release of smallpox virus. See text for further details on mechanisms and associated relative likelihood.
* Security experts disagree as to how this may change over time, as discussed in the paper.
+ Most of these pathways are decreasing, but the biotech pathway is expected to increase with time.
Table 1: Main pathways for possible recurrence of smallpox.
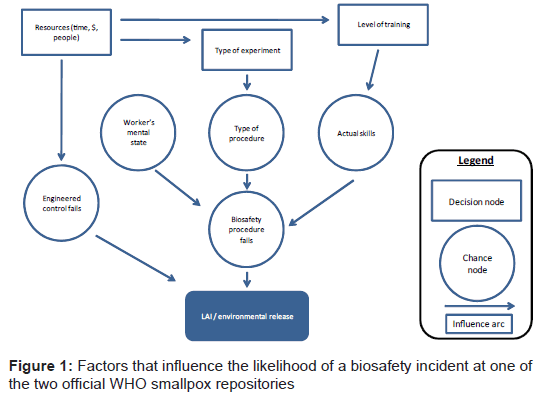
Influence diagrams (ID) [5] are graphical representations of the problem that are useful for illustrating the structure of the domain. IDs describe the relationships between the different elements; the IDs in this paper (see Figure 1 for an example) have decision nodes (represented by the open rectangles) and chance nodes (represented by the open circles). The relationships between the nodes are shown by the arcs. The outcomes are represented by the shaded rectangles with rounded corners. Elements can have causal links or be linked temporally. These diagrams can also help explore uncertain variables and the value of perfect and imperfect information on the problem. We conducted a comparative analysis of the relative likelihoods for these pathways and explored how key variables that influence likelihood may change over time. The IDs in this paper have been constructed with a level of detail to facilitate a comparative analysis of the likelihoods for the pathways; additional details could easily be added into these frameworks for a more detailed analysis of a particular pathway.
Accidental reintroduction pathways – Table 1, Categories A-C
The World Health Organization (WHO) declared smallpox eradicated in 1980. At the time of eradication, many laboratories still held samples of the virus. After several laboratory acquired infections (LAI) [6,7] and the inadvertent discovery of unregistered clinical smallpox samples, WHO established two official repositories - one at the Centers for Disease Control and Prevention (CDC) in the US and one that is now at VECTOR in Russia. This was done specifically to reduce the risk of reintroduction of smallpox from a biosafety incident by drastically reducing the number of laboratories having the virus. Biosafety incidents that could lead to reintroduction of smallpox can be subdivided into laboratory-acquired infections, where a worker handling the virus becomes exposed and subsequently infected, and a release from the laboratory into the environment through a breach of containment. Biosafety measures to mitigate against these situations are a combination of engineered and procedural controls [8,9]. Because of the potential consequences, smallpox virus is only handled in maximum containment (BSL4) at the two WHO repositories.
There are no known LAI of smallpox at either of the two repositories. However, there have been a few LAIs resulting from accidents with other high-risk pathogens in BSL4 settings [10-12]. Although possible, there is no evidence of an environmental release from a BSL4 having ever occurred, providing some evidence of how unlikely this event is. The ID for a biosafety incident (Figure 1) illustrates that factors that directly lead to a LAI or environmental release are procedural or equipment failure. For a given procedure, human factors (time pressures, skills, alertness/ confidence) influence the chance that there will be a procedural failure that could lead to an exposure and subsequent infection or breach of containment and environmental release. If key equipment fails, this can directly result in an environmental release or it can be a secondary cause. Redundancy in critical systems is put in place to specifically mitigate against this risk (e.g. double HEPA filters on exhaust). Certain procedures are inherently more risky than others, such as work with higher titers, animal studies, and aerosolization experiments. In a BSL4 setting, where workers are encased in a protective suit, there is a greater chance for accidental exposure if sharps are required for a procedure than if the protocol does not require syringes or other sharps. The ID highlights that proper execution of biosafety procedures is influenced by the skills of the worker carrying out the activity. Accordingly, maximum containment laboratories require workers to have significant work experience at lower levels of containment followed by a period of mentoring at BSL4 before being allowed to work independently. However, even highly skilled workers can be distracted and may occasionally make mistakes.
Presumably, the practice of biosafety improves with time. Architects and engineers have more sophisticated engineered controls to include in new buildings and facility managers have a better understanding of how to properly maintain those controls in their existing facilities. These factors reduce the risk of an inadvertent release due to a biosafety incident at a known repository.
However, complex systems for controlling biological risks may fail, through neglecting to properly maintain the equipment or bypassing systems to save time. A major outbreak of legionnaire's disease originated from the former [13], where a non-qualified individual cut maintenance to save money, for which she was eventually jailed. The recent incident involving an environmental release of Foot-and-Mouth Disease virus at a renowned research facility also highlighted problems involving poor maintenance, shared management responsibility, and non-validated procedures [14]. Inherent to these examples is the challenge of continually funding maintenance when there are no mishaps. As evidenced in the recent Spanair crash [15] or the 1999 criticality accident at the Tokai-Mura nuclear fuel cycle facility [16], users can become complacent towards and even bypass the checks required to ensure complex systems function properly.
The type of experiment also varies over time but it is not clear, if on average, the experiments are more or less risky. A greater emphasis on molecular techniques to characterize the virus would be expected to reduce the biosafety risk but an increased number of studies in nonhuman primates aimed at trying to understand the pathology of the disease would likely increase the biosafety risk.
Also, risks associated with other human factors, such as lack of training, information, understanding, and validation as well as negligence and inadvertence on the part of the laboratory workers may continue to exist. Therefore, despite the general improvements in laboratory biosafety and biosecurity, incidents of laboratory acquired infections may still be observed in developed countries [17]. While there have been no documented accidents with smallpox, other equally high-risk pathogens including Ebola and Marburg have infected workers in the US [12], Germany [18] and Russia [10,19] , the latter taking place at the Vector Institute, home to the Russian WHO smallpox repository. Corroborating these observations, virologist Heinz Feldmann commented on Ebola research "We have all the proper protocols in place, but we're still humans and we make mistakes. It's just a matter of time when the next incident will happen" [20].
We conclude that on balance, the likelihood of LAI and environmental releases of smallpox at WHO repositories have probably decreased due to improved biosafety involving more sophisticated engineered and procedural controls. In theory, the potential for additional improvements may eventually be exhausted, or at least a greater emphasis on biosafety will see diminishing returns.
Biosafety incident outside the known repositories– Table 1, Category B
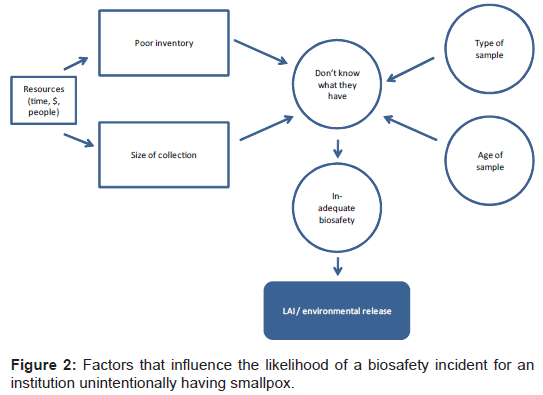
Although laboratories either destroyed or sent their known smallpox virus samples to the WHO repositories, it is possible that facilities may still unknowingly retain virus samples. Smallpox virus could be present in clinical samples that were taken to test for smallpox or other diseases while smallpox was a naturally occurring disease. Many institutions retain clinical samples for historical or research value. If clinical samples that were collected and tested for other reasons also contained smallpox virus, there could be a mismatch between the biosafety measures implemented and the pathogen (e.g. smallpox virus) being handled. The unintentional use of inadequate biosafety procedures could lead to a biosafety incident (Figure 2), resulting in an inadvertent reintroduction. The other plausible scenario for a biosafety incident outside of the known repositories is an unlabeled or mislabeled vial in a freezer at an institution or even the discovery of a labeled vial in a freezer that was forgotten. The likelihood of unknown samples containing smallpox virus is probably inversely correlated with the quality of the institution's inventory system. Accordingly, the age of a facility is of concern since inventory practices were perhaps less robust in the 1950's and 60's, when smallpox was still endemic.
We conclude that the risk of a biosafety incident beyond the known repositories should decrease with time. Although smallpox virus is relatively stable for a virus, samples stored under conditions not intended to preserve smallpox will inevitably degrade with time. The relevant collection of unknowns is also likely to decrease over time as facilities lose contents of freezers in power outages or reduce their collections as their missions change. At least none of the factors included in the ID can be expected to increase with time.
Samples from outside of a bioscience institution - Table 1, Category C1
There is also the possibility of finding samples of smallpox virus outside of a laboratory (Figure 3). Multiple experts have speculated about the possibility of isolating smallpox virus from cadavers in the permafrost [21]. In theory, the impacts of global warming and the urbanization under the global population expansion could potentially become contributing factors to facilitating the isolation of smallpox virus from cadavers buried in the Arctic permafrost in the future, although it expectedly carries a very low likelihood [22,23]. There may be other sources of archived samples. For example, a possible smallpox or vaccinia scab was discovered in a book in a library in Santa Fe in New Mexico [24], and virus has been reported originating from Indian skin samples stored at -20°C, although infectivity was low. Indeed, viable virus has been recovered from scabs stored at room temperature for more than a decade [25]. Storage conditions are a key variable in this pathway, thus the viability of these types of samples would clearly degrade over time.
Mutation of an orthopox virus - Table 1, Category C2
There are other orthopox viruses that could naturally mutate into a strain that could cause a smallpox-like illness. Alternately, a laboratory experiment with orthopox viruses could have unintended consequences and produce a virus that could cause a smallpox-like illness. The famous IL-4 experiment [26] is an obvious example of scientists not always fully understanding the potential consequences of their research ahead of time. The likelihood of a natural or inadvertent mutation (Figure 4) should correlate with the homology of the genome and / or the type of experiment being conducted. Many of the orthopox viruses have a high degree of homology with the smallpox virus (e.g. monkey pox virus has a 96.3% homology [27] but it is unclear how many mutations would be required to produce a strain that could cause similar clinical symptoms.
We conclude that while the likelihood of a natural mutation is negligible, constant over time, and influenced only by chance variables; the driving forces for selection of a mutant, however, are not constant. The increased proximity of man to monkeys in Africa as humans seek new places to live and starvation forces them to hunt bush meat increases the chance of exposure to new viruses including monkey pox. The number of exposures increases the probability of a mutation leading to a propagating disease appearing, exemplified by the fact that monkey pox is rather more infectious in Africa than in the US population. Finally, the likelihood of an unexpected experimental result producing a virus that could cause a smallpox-like illness may rise with time as more sophisticated experiments are undertaken by a broader set of scientists. This may be balanced by a growing understanding of the functioning of the genome, perhaps reducing the likelihood of unintended experimental consequences.
Deliberate reintroduction pathways – Table 1, Categories D-F
Biosecurity incident at a known repository - Table 1, Category D
As with any known valuable asset, it is feasible that someone could target the WHO repositories in an attempt to steal the virus (Figure 5). This may be especially pertinent for smallpox virus because repositories are the only known sources of the virus. Conceivably, the virus could be stolen by someone at the facility with access to the virus (an "Insider") or an external adversary that does not have authorized access (an "Outsider"). Laboratory biosecurity is the institutional and personal security measures designed to prevent the loss, theft, misuse, diversion or intentional release of pathogens and toxins [9]. Measures to reduce the risk of insider theft include limiting the number of insiders, access control, good material control and accountability programs, and personnel reliability programs [27,28]. Physical security measures are the main tool for mitigating against the outsider risk. As shown in the ID, the likelihood of an adversary attempting theft may also be influenced by their perception of the security of the smallpox virus holdings and the amount of information about the facilities and the collections that is publicly available.
Collusion is a theft scenario involving cooperation between an insider and an outsider; it is mitigated through a combination of laboratory biosecurity measures that would be put in place to address those adversary categories separately. One could also envision a scenario where an outside adversary attempts to buy the virus from someone with authorized access. Laboratory biosecurity measures to address the inside adversary also mitigate against this risk.
Like biosafety, laboratory biosecurity measures have improved with time [29]. However, no security measures are perfect. The US FBI has determined that the 2001 anthrax letters were perpetrated by a thoroughly vetted scientist that had authorized access to the material at work (i.e. an "Insider"). Beyond biology, there are a multitude of examples of traitors from both the US and the Soviet Union during the Cold War; these are all individuals that had been subject to extensive personnel reliability programs. The motivation of an Insider stealing smallpox virus is difficult to gauge objectively. The two repositories have "elaborate security measures" in place to prevent theft by outside adversaries [23].
However, there has been a rise of sophisticated yet loosely networked transnational terrorist groups that have shown an interest in bioterrorism [30]. This was emphasized in the recent report, World at Risk, by a US Congressionally-appointed commission [31], which also stated that we "should be less concerned that terrorists will become biologists and far more concerned that biologists will become terrorists". Therefore, we conclude that there is a medium but ultimately unknown likelihood of theft of smallpox virus from a WHO repository because the intent of an individual or terrorist can be impossible to discern.
Biotechnology – mutation or de novo synthesis - Table 1, Category E
The rapidly developing field of synthetic genomics offers great promise for both basic biological research and as a new and powerful tool for biotechnology. But as with virtually any new technology that promises significant benefits, synthetic genomics brings with those benefits a series of societal concerns and issues. Perhaps the most prominent concern raised is that the technology may end up being used to create dangerous pathogens by those with malicious intent and, accordingly, could be a source for the deliberate introduction of eradicated or novel pathogens (Figure 6). For example, The Nuclear Posture Review of the US Department of Defense released in April 2010 explicitly states: "Given the catastrophic potential of biological weapons and the rapid pace of bio-technology development, the United States reserves the right to make any adjustment in the assurance that may be warranted by the evolution and proliferation of the biological weapons threat and US capacities to counter that threat" [32].
Examples of recent advances include the synthesis of live infectious polio virus in 2002 [33], followed by Phi174 phage which was produced in just 2 weeks by the Venter Institute in 2003 [34]. Since, the synthesis of the 1918 influenza [35] and Marburg virus [36] were reported in 2005. In 2008 both SARS virus [37] and the bacteria M. genitalium [38] were produced, and the technique reported in the professional literature. While polio virus is a relatively simple and small virus with only 7,751 base pairs (bp), SARS virus contains approximately 30,000 bp and M. genitalium 582,970bp. The latest milestone, involving the creation of a bacterium controlled by a synthesized genome [39], demonstrates that not only are the technical barriers being pushed in terms of length of the genome and overall size, but also the complexity of the synthesized organisms is increasing and the technology is becoming easier to handle.
Turning again to smallpox virus as our indicative example, a full length sequence of the viral genome comprises about 186,000 bp, and the sequences of 45 distinct variola virus isolates have been made publicly available [40]. The 60 bp palindromic ends are not available for all 45 isolates, but have been determined for at least one highly pathogenic isolate [41]. Rather than synthesizing a full-length synthetic genome, deliberate mutation of a related orthopox virus could lead to the creation of a strain capable of causing a smallpox-like illness. Only a few thousand mutations out of the 186,000 bp genome would be needed to be introduced into closely related viruses such as readily accessible monkeypox, camelpox or gerbilpox [40]. Although these procedures have been declared illegal [42], it is unlikely that the appropriate legal framework is in place in all countries, and, even then, complete control of such activities is not feasible.
Also, there is an increasing concern that vaccinia virus used for producing smallpox vaccine could be used as a template for creation of variola virus. For example, vaccinia virus expresses a protein (vaccinia virus complement control protein [VCP]), which constitutes a vaccinia virulence factor. DNA comparison studies have revealed that the genomes of all variola strains encode a complement regulatory protein consisting of four "smallpox inhibitor of complement enzymes (SPICE)" which is the homologue of VCP [43]. According to Nariyoshi Shinomiya, professor of Japan's National Defense Medical College, Japan's smallpox vaccine is based on the LC16m8 Lister vaccinia strain, and there is an emerging concern that the introduction of SPICE into this vaccinia virus could potentially lead to the creation of a highly pathogenic strain of pox virus. Although it would not be a smallpox virus, it could possibly retain high pathogenicity as well [44].
In conclusion, the attempted synthesis of smallpox is not only illegal, but also highly controversial from both a security and scientific perspective. Distinguished scientists contend that given recent scientific advances the de novo synthesis constitutes a fairly straightforward enterprise in terms of skills, time and funding, while sceptics argue that the technology is presently not sophisticated enough to succeed [45]. Most agree, though, that biotechnology is rapidly evolving and it is hard to predict what might be technically feasible in a 5- to 10-year time frame [46].
State illicit biological weapons program - Table 1, Category F
There is a long history of interest, development, and usage of biological weapons by states underscoring that this is not merely of academic concern. Although the 1972 Biological Weapons Convention bans the development of biological weapons, this does not eliminate the chance that a state program could be interested in developing smallpox as a biological weapon through an illicit effort. States have multiple avenues under which they could obtain the virus for this purpose (Figure 7). Most simply, a state could have retained samples of the virus post-eradication, not destroying or turning over all of their specimens to the WHO repositories. This pathway would have required a state to have some motivation to go against the global norm and intentionally retain the virus. Gregory Koblentz has explored this possibility in detail [47]. This scenario can have a constant or possibly decreasing risk with time if states that may have retained the virus decide they no longer have a reason to maintain their specimens. The advancement of microbial forensics for law enforcement may possibly contribute to deterring those states from managing such illicit biological weapon program. Conversely, it is also possible for a state to deepen its interests in such illicit biological weapon program as an asymmetric tool to balance the military gap vis-a-vis its adversary.
If a state did not retain the virus, they could acquire it from the other pathways articulated in this paper. A state could accidentally discover the virus in a laboratory or non-laboratory setting within their territory. Alternatively, smallpox virus may be acquired from another state program that had retained the virus post-eradication or they could attempt to steal it from one of the WHO repositories. States could also utilize the latest biotechnology advances to create the virus. States have greater resources available to them than individual scientists or other non-state actors so a biotechnology pathway is more feasible for a state program. Of equal concern is the fact that motivation is not a constant, and depends on the regime in power at the time, the political and religious ideology of the regime and population, and the threat that they perceive from other states, thereby possibly influencing the accepted rules of engagement. Illustrating the latter point, war and blaming another state for "injustices" have historically been a powerful political force for unity. The ability of a tolerant state to become extreme is clearly shown in the whole of 20th century European history culminating in genocide in the Balkans a decade ago. A decreasing trend could easily change into a remilitarization trend in a limited time-frame.
We conclude that the likelihood of state illicit biological weapons programs having smallpox virus is constant or possibly decreasing for all possible routes except for biotechnology pathways. States are most able to take advantage of the latest scientific advances. Again, the unknown element is mainly whether a state program has the motivation to pursue acquisition of the smallpox virus through one of these pathways.
Overall trends
For the past three decades the main defense against smallpox, has been the extreme inaccessibility of the agent. However, the likelihood of an accidental or deliberate re-introduction of an eradicated agent is expected to evolve with time.
Certainly, the likelihood of a biosafety incident is tied to both the biosafety measures (generally improving with time), the type and level of research (increased sophistication), the number and location of research facilities (increasing with a global reach), as well as number and training of personnel. The viability of samples inadvertently retained outside of official repositories decrease with time, lowering the likelihood of re-introduction from these sources.
However, the likelihood of a biosecurity incident is also a dynamic balance between biosecurity safeguards and increased terrorist capability and interest in biological weapons. While historically of less concern, the deliberate mutagenesis or de novo synthesis will become increasingly important over time with the rapid advancement in biotechnology. Although research with pathogens not found in nature is important from a scientific, public, and animal health perspective, lower risk tolerance by society may require facilities that have these organisms to implement enhanced biosafety and biosecurity measures. This requires more attention to personnel reliability programs, types of experiments, and the numbers of laboratories and personnel working with eradicated pathogens to ensure that the likelihood of reintroduction is responsibly managed.
Conclusion
Current smallpox risk assessments are rather uniform in terms of the elements that constitute the consequence arm of the equation, and most conclude that any outbreak would constitute a potential major international security and public health challenge. The main, largely unknown, influencing factor on the overall risk is the availability of the agent. While the likelihood of acquisition (a necessary prerequisite to attack) is low, the advances of science to include the synthesis of eradicated, soon-to-be eradicated or inaccessible agents like 1918 influenza, polio virus and smallpox, respectively, is a main driver in influencing the likelihood of attack or accidental release, thus representing a game-changing event.
The difficulty in estimating the likelihood of the deliberate reintroduction pathways are very strongly determined by the motivation of an adversary and the technical challenges of de novo synthesis. We have not attempted a socio-political or technical analysis, both of which would probably only be valid for a limited time. Thus we concede that there could be disagreement with our conclusions. That said, countries preparedness for risks associated with eradicated and/or novel pathogens relies predominately on the unavailability of these pathogens. Through a systematic analysis of possible pathways of introduction, this paper has challenged this assumption. The likelihood of smallpox being reintroduced (accidently or deliberately) is unknown but non-zero and is increasing with time. With the possible advent of de-novo synthesis, the dangerous risk of recurrence becomes real and present, changing the overall risk assessment.
Prerequisites for utilizing the weapons potential of smallpox include overcoming both intentional and technical barriers. The unknown and changing nature of intent is partly tied to socioeconomic disadvantages that drive new political aspirations and encourage aggressive and often extreme measures, which could include legitimizing the use of bioweapons. In addition, given the many ways of obtaining dangerous pathogens and the number of additional steps required to use them as weapons, synthetic genomics has so far made a limited contribution to today's general bioterrorist threat. However, when looking at some of the most feared and potent biological agents, epitomized by smallpox, inaccessibility is seemingly the main defense against nefarious use, a barrier which is destined to crumble with the ongoing rapid advancement of the life sciences.
References
- Henderson DA, Inglesby TV, Bartlett JG, Ascher MS, Eitzen E, et al. (1999) Smallpox as a biological weapon: medical and public health management Working Group on Civilian Biodefense JAMA 281: 2127-2137.
- Fenner F, Henderson DA, Arita I, Jezek Z, Ladnyi ID (1988) Smallpox and its Eradication. World Health Organization.
- McKenzie FE (2004) Smallpox models as policy tools. Emerg Infect Dis 10: 2044-2047.
- Smith BT, Inglesby TV, Brimmer E, Borio L, Franco C, et al. (2005) Navigating the storm: report and recommendations from the Atlantic Storm exercise. Biosecur Bioterror 3: 256-267.
- Howard RA, Matheson JE (2005) Influence Diagrams. Decision Analysis 2: 127-143.
- Cox PJ, McCarthy K, Millar ELM (1974) Report of the Committee of Inquiry into the Smallpox Outbreak in London in March and April 1973. Her majesty's Stationery Office.
- (1978) Smallpox research after Birmingham. Lancet 2: 560.
- CDC/NIH (2007) Biosafety in Microbiological and Biomedical Laboratories 5th edition.
- World Health Organization (2004) Laboratory Biosafety Manual 3rd edition.
- (2004) Russian scientist dies in ebola accident at former weapons lab.
- (2010) Canadian-made Ebola vaccine used after German lab accident.
- Kortepeter MG, Martin JW, Rusnak JM, Cieslak TJ, Warfield KL, et al. (2008) Managing potential laboratory exposure to ebola virus by using a patient biocontainment care unit. Emerg Infect Dis 14: 881-887.
- Mitchell E, O'Mahony M, Watson JM, Lynch D, Joseph C, et al. (1990) Two outbreaks of Legionnaires' disease in Bolton Health District. Epidemiol Infect 104: 159-170.
- (2010) Final report on potential breaches of biosecurity at the Pirbright site 2007.
- (2010) Spanair MD-82 crash: Pilots twice failed to check flap setting.
- (2000) NRC Review of the Tokai-Mura criticality accident.
- Rusnak JM, Kortepeter MG, Hawley RJ, Anderson AO, Boudreau E, et al. (2004) Risk of occupationally acquired illnesses from biological threat agents in unvaccinated laboratory workers. Biosecur Bioterror 2: 281-293.
- (2010) Virologist in Isolation After Accident With Ebola.
- Nikiforov VV, Turovskii I, Kalinin PP, Akinfeeva LA, Katkova LR, et al. (1994) [A case of a laboratory infection with Marburg fever]. Zh Mikrobiol Epidemiol Immunobiol 104-106.
- (2010) New Ebola Drug 100 Percent Effective In Monkeys.
- L'vov DK, Zverev VV, Gintsburg AL, Marennikova SS, Pal'tsev MA (2008) [Smallpox is a dormant volcano]. Vopr Virusol 53: 4-8.
- (2010) Climate Threat: Thawing Tundra Releases Infected Corpses.
- Tucker J (2010) The Smallpox Destruction Debate: Could a Grand Bargain Settle the Issue?
- (2010) Century-old smallpox scabs discovered.
- Wolff HL, Croon JJ (1968) The survival of smallpox virus (variola minor) in natural circumstances, Bull World Health Organ 38: 492-493.
- Jackson R, Ramsay AG, Christensen C, Beaton S, Hall DF, et al. (2001) Expression of mouse interleukin-4 by a recombinant ectromelia virus suppresses cytolytic lymphocyte responses and overcomes genetic resistance to mousepox. J Virol 75: 1205-1210.
- Shchelkunov SN, Totmenin AV, Babkin IV, Safronov PF, Ryazankina OI, et al. (2001) Human monkeypox and smallpox viruses: genomic comparison. FEBS Lett 509: 66-70.
- Clevestig P (2009) Applied Biosecurity. Stockholm International Peace Research Institute Press.
- Salerno RM, Gaudioso JM (2007) Laboratory Biosecurity Handbook. CRC Press.
- Salerno RM, Hickok LT (2007) Strengthening bioterrorism prevention: global biological materials management. Biosecur Bioterror 5: 107-116.
- (2008) World at Risk. The Report of the Commission on the Prevention of WMD Proliferation and Terrorism.
- (2010) Nuclear Posture Review Report.
- Cello J, Paul AV, Wimmer E (2002) Chemical synthesis of poliovirus cDNA: generation of infectious virus in the absence of natural template. Science 297: 1016-1018.
- Smith HO, Hutchison CA III, Pfannkoch C, Venter JC (2003) Generating a synthetic genome by whole genome assembly: phiX174 bacteriophage from synthetic oligonucleotides. Proc Natl Acad Sci USA 100: 15440-15445.
- Tumpey TM, Basler CF, Aguilar PV, Zeng H, Solorzano A, et al. (2005) Characterization of the reconstructed 1918 Spanish influenza pandemic virus. Science 310: 77-80.
- Enterlein S, Volchkov V, Weik M, Kolesnikova L, Volchkova V, et al. (2006) Rescue of recombinant Marburg virus from cDNA is dependent on nucleocapsid protein VP30. J Virol 80: 1038-1043.
- Becker MM, Graham RL, Donaldson EF, RockxB, Sims AC, et al. (2008) Synthetic recombinant bat SARS-like coronavirus is infectious in cultured cells and in mice. Proc Natl Acad Sci USA 105: 19944-19949.
- Gibson DG, Benders GA, Andrews-Pfannkoch C, Denisova EA, Baden-Tillson H, et al. (2008) Complete chemical synthesis, assembly, and cloning of a Mycoplasma genitalium genome. Science 319: 1215-1220.
- Gibson DG, Glass J, Lartigue C, Noskov V, Chuang R, et al. (2010) Creation of a bacterial cell controlled by a chemically synthesized genome. Science 329: 52-56.
- Esposito JJ, Sammons SA, Frace AM, Osborne JD, Olsen-Rasmussen M, et al. (2006) Genome sequence diversity and clues to the evolution of variola (smallpox) virus. Science 313: 807-812.
- Massung RF, Liu LI, Qi J, Knight JC, Yuran TE, et al. (1994) Analysis of the complete genome of smallpox variola major virus strain Bangladesh-1975. Virology 201: 215-240.
- (2008) WHO recommendations concerning the distribution, handling and synthesis of variola virus DNA. Weekly Epidemiological Record 83: 393-400.
- Rosengard AM, Liu Y, Nie Z, Jimenez R (2002) Variola virus immune evasion design: expression of a highly efficient inhibitor of human complement. Proc Natl Acad Sci USA 99: 8808-8813.
- 44 (2010) Authors interview with professor Shinomiya. Personal Communication.
- VogelK (2008) Framing biosecurity: an alternative to the biotech revolution model?. Science and Public Policy 35: 45-54.
- Carlson R (2003) The Pace and Proliferation of Biological Technologies. Biosecur Bioterror 1: 203-214.
- Koblentz G (2003) Assessing the proliferation of smallpox.
Relevant Topics
- Anthrax Bioterrorism
- Bio surveilliance
- Biodefense
- Biohazards
- Biological Preparedness
- Biological Warfare
- Biological weapons
- Biorisk
- Bioterrorism
- Bioterrorism Agents
- Biothreat Agents
- Disease surveillance
- Emerging infectious disease
- Epidemiology of Breast Cancer
- Information Security
- Mass Prophylaxis
- Nuclear Terrorism
- Probabilistic risk assessment
- United States biological defense program
- Vaccines
Recommended Journals
Article Tools
Article Usage
- Total views: 19983
- [From(publication date):
April-2011 - Dec 18, 2025] - Breakdown by view type
- HTML page views : 15140
- PDF downloads : 4843