Commentary Open Access
A “Chaotic” Approach to the Treatment of Advanced Cancer
Sarah Adelaide Crawford*
Cancer Biology Research Laboratory, Southern Connecticut State University, CT 06515, USA
- *Corresponding Author:
- Sarah Adelaide Crawford
Professor, Cancer Biology Research Laboratory
Southern Connecticut State University, CT 06515, USA
Tel: +1-203-392-6215
E-mail: crawfords2@southernct.edu
Received date: July 21, 2017; Accepted date: August 01, 2017; Published date: August 07, 2017
Citation: Crawford SA (2017) A “Chaotic” Approach to the Treatment of Advanced Cancer. J Tradit Med Clin Natur 6:232.
Copyright: © 2017 Crawford SA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Traditional Medicine & Clinical Naturopathy
Milestones in Cancer Discovery and their Clinical Therapeutic Applications
Not so many years ago the origins of cancer were a mystery. Of course, even the ancients were aware of its manifestations, as the archaeological record shows. But the cause, the how and why it happens, have remained elusive until very recently. The dawn of the 20th century led to discoveries by Rous, Ellerman and Bang on the viral etiology of some avian cancers, suggesting a possible viral connection with human cancers. At mid-century, the groundbreaking work by Nowell and Hungerford identified the “Philadelphia” chromosome as a ubiquitous component of Chronic Myeloid Leukemia (CML) suggesting a genetic cause. As the 20th century closed, all the pieces began to come together as studies of the Rous Sarcoma Virus (RSV) identified src as a cellular gene absconded by an avian Retrovirus and mutated into a cancer-causing gene. Alas, to quote Shakespeare, “the fault is not in the stars, but in ourselves” a conclusion about cancer made obvious as researchers began to unearth a seeming avalanche of mutated human genes implicated in many types of malignancy. The products of these genes were shown to comprise growth promoting oncogenes and tumor suppressor genes whose roles in cell cycle regulation were abrogated as a consequence of mutation, leading directly to malignant cell transformation. Even the viral theory of cancer was validated as some viruses were shown to encode gene products that interacted with cellular growth control genes to disrupt cellular growth control mechanisms. All the pieces of this long mysterious puzzle began to come together in an intellectually satisfying manner. Even more importantly, these discoveries led to the development of a targeted therapeutic that interferes with the abnormal abl oncogene signaling activated by the translocation in CML that produces the Philadelphia chromosome! It is a beautiful story, the historical convergence of theoretical paradigms in a therapeutically meaningful way. The extraordinary success of Gleevec and its relatives in the treatment of CML, formerly a death sentence, became the paradigm for the development of many other targeted therapeutics, including Rituxan for the treatment of hematologic malignancies associated with CD20 up-regulation, and many, many other targeted drugs, far too numerous to cite here. Many successful results have been obtained using this approach, which is an extraordinary validation of the significance of a century of inquiry on the origins of cancer.
Problems with Current Therapeutic Paradigm: Reductionism
Here is where I would like to end with, “and they all lived happily ever after”, but this ending, of course, is reserved for fairytales, and current therapeutic approaches to cancer treatment and management are, for many patients, no fairytale. Unfortunately, despite the extraordinary success rates in the treatment of certain cancers such as the abovementioned CML, certain childhood leukemias, and others, many of the most common cancers are often not only treatment refractory, but are the subject of considerable uncertainty with respect to the most useful treatment approaches. One current approach to addressing this problem involves the concept of “personalized” cancer treatment, involving a detailed study of the genetic origins of individual patient tumors as a guide to identifying the best combination of targeted therapeutic approaches most likely to produce disease remission. Even this approach is frequently unsuccessful in achieving long-term disease remission. This therapeutic paradigm is based on the reductionist viewpoint, that targeting specific mutational dysfunctions linked to the malignant phenotype will inflict a lethal cytotoxic effect. There are several problems with this approach, particularly with respect to cancers with complex and advanced aetiology.
Therapy Resistance
One of the oft-cited reasons for therapy failure in personalized medicine as well as in conventional chemotherapy/radiation therapies is the phenomenon of drug resistance. The tumor, as a microcosm of natural selection, evolves the capacity to resist the cytotoxic effects of chemotherapy and radiation due to the survival of a few genetically resistant cells that ultimately become the predominant tumor cell population, which ultimately leads to therapy failure. Moreover, this type of resistance mechanism has also been shown to be responsible for the failure of much gene-targeted therapeutics to establish durable treatment responses. This phenomenon is believed to be a major factor in recurrent disease and has led to the development of combinatorial treatment approaches in an attempt to hit multiple targets simultaneously to decrease the likelihood that small resistant tumor populations will survive.
Cancer: Is the whole greater than the sum of its parts?
There is also another possible cause for chemotherapy/radiation and targeted therapy failure, that is, the systemic causes and effects of cancer that resemble a system in chaos, that may not respond to gene targeting by simply turning off the dysregulated motor that drives cancer cell growth. A system in chaos may have lost direct connection with the primary driver events that originally set the system in motion, and, therefore, may be susceptible only to disruptions that are directly involved in sustaining the altered state of dysregulated homeostasis. The reductionist paradigm that underscores many current approaches to cancer therapy relies on a linear model of cause and effect relationships between abnormal cell gene expression and growth regulation. However, the linear model is a poor representation of cancer progression, which follows non-linear kinetics where the lines between cause and effect are far more loosely associated [1].
If this is the case, then perhaps the reductionist approach is not adequate to derail advanced systemic cancers that have lost the ability to connect with their primary genetic origins due to the establishment of a complex net of interactions at the cellular, multicellular and systemic levels that drive the chaotic patterns of dysregulated homeostasis that define many human cancers. Is this a viable model of cancer? Can Chaos Theory account for the perturbations of cell growth that define the cancer phenotype? And, if so, where is the evidence and how should we think about the treatment and prevention of cancer in a chaotic paradigm?
Chromosomes and Chaos
Intriguing research by Davoli et al. [2] has shown that the apparently random distribution of chromosomal aneuploidies seen in many cancers is actually the result of patterns directly implicated in oncogene and tumor suppressor gene dys-regulation. These researchers found that these seemingly random chromosomal aneuploidies are part of a global cellular response to abnormal epigenetic/environmental factors that select for this abnormal phenotype. The apparent random chromosomal content seen in many malignancies has now been shown to be a patterned genetic response to selective forces in the tumor microenvironment favoring proliferation and survival, resulting in a microcosm of convergent evolution (Figure 1). All of this suggests that the so-called disease progression factors associated with many later stage cancers may actually be responses to an abnormal environment that selects for altered (malignant) phenotypic behavior. In this conception of cancer, gene targeted therapeutic approaches may have a relatively minor effect on stemming the tide of disease progression. As long as the tumor microenvironment and systemic factors remain unchanged, the malignancy will continue to evolve in patterns that support survival and proliferation in this abnormal milieu.
Figure 1: A comparison of a normal human karyotype (a) and a karyotype from an aneuploid tumor (b). What appear to be random increases in chromosome number are now attributed to selection for amplification of oncogenes or deletion of tumor suppressor genes found on different chromosomes. Image: Joanne Davidson, Mira Grigorova and Paul Edwards/University of Cambridge.
Evidence for chaos in cancer
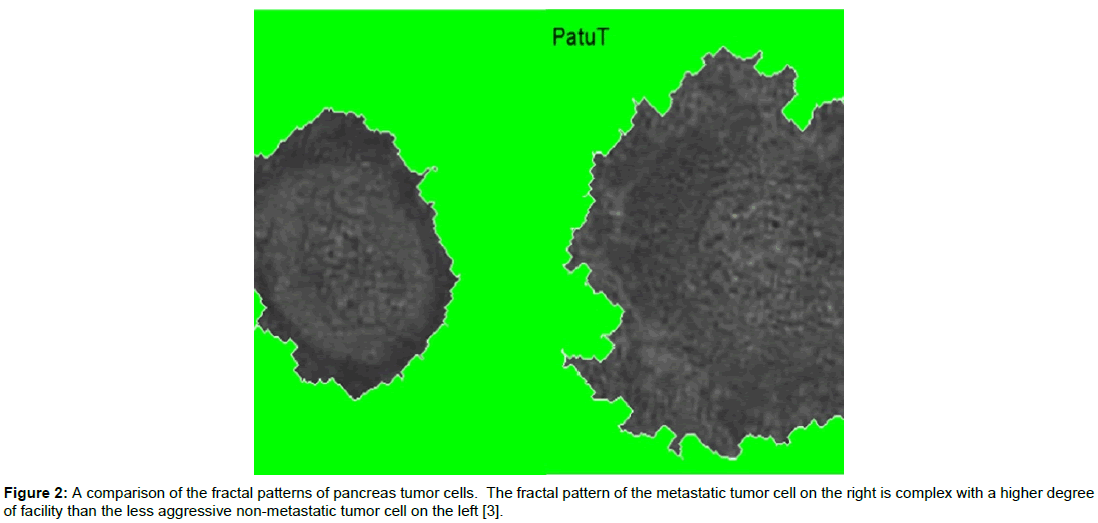
So- in the world of cancer chaos, there may be driving forces that determine the patterns of gene dys-regulation consistent with phenotypic fitness. The analogy between this biological system and systems in chaos is compelling, as both involve the repetition of self, the production of fractal structures, and even the existence of attractors. An emerging principle of biophysics is the concept that cells are complex adaptable systems that display fractal behavior and non-linear growth patterns. Repeating recognizable structures that form in different scales as fractals can be seen in the morphological growth patterns of many solid tumors. Research by Klein et al. [3], on Reflection Interference Contrast Microscopy (RICM) of tumor cells by fractal analysis of cell adhesion patterns and surface contour geometry of human pancreatic tumor cells has shown a reproducible (to 97% accuracy) correlation between fractal dimension parameters and tumor stage and grade (Figure 2). This ability to quantitate the status of disease progression using calculations of fractal dimension patterns underscores the relationship between solid tumors and fractal patterns defined by Chaos Theory. In this context, the dys-regulated gene expression pattern of the tumor can be seen to produce a state of growth imbalance and disease progression that produces increased fractal dimensional complexity of both the contours and adhesion patterns of the tumor surfaces.
Figure 2: A comparison of the fractal patterns of pancreas tumor cells. The fractal pattern of the metastatic tumor cell on the right is complex with a higher degree of facility than the less aggressive non-metastatic tumor cell on the left [3].
What about the return to previous starting points of form called the “strange attractors”? The “immortalized” repetition of self is the hallmark of the cancer cell. A developing tumor can be modeled as a self-organizing system where higher order levels of complexity are the product of interactions between the evolving system (the tumor) and the context (systemic environment) in which it develops. Genetic dys-regulation, inflammation, oncogenic virus infection and other proximate initiators of malignant cell transformation comprise stressors that can be seen on the biophysical level to induce instability in this complex system. If equilibrium is not restored, the dys-regulated growth patterns resulting from tumor initiation mechanisms will move this system to what is termed in Chaos Theory a “critical choice” between alternative pathways that may involve multiple possibilities. It is at this point that the system becomes unpredictable. The system itself, when examined closely, (in this case, the tumor) displays what appears to be random changes (for example, degrees of localized invasion versus metastatic expansion) that are difficult to predict or control. However, due to the deterministic boundaries of this representative chaotic system, its overall behavior follows a discrete and finite boundary circumscribed by the interactions between the boundary-the systemand the chaotic center-the tumor- in the form of strange attractors that construct the finite set of patterns to which the system may evolve. The direction that is ultimately followed past the critical point results in what is called a “second order change” (McClure, 1998) where behavior becomes increasingly unpredictable and even explosive. This can be seen in the later stages of tumor progression as systemic equilibrium unravels. The fractal dimensions of the tumor in its later stages represent the product of a higher order level of self-organization that results in increased complexity displayed in the form of disease progression and systemic metastasis. The instability of the evolving system is influenced by the strange attractors to produce adaptations that facilitate survival (tumor progression) or result in death.
The Cancer Paradox: Deterministic Unpredictability
The chaotic system, as defined by Poincare, incorporates random unpredictability with determinism, a seeming paradox that that can be explained by defining randomness confined within specific parameters as in Poincare’s confinement of mathematical variables to a specific area or box. By analogy with tumorigenic mechanisms, tumor cell growth occurs in chaotic patterns with a random unpredictability encased within the deterministic box of the systemic micro/macroenvironment (Figure 3). As a consequence of this defined association between randomness and determinism, mediated by strange attractors, chaotic variability contains an intrinsic predictability. As applied to cancer, the intrinsic predictability involves the certainty that, in the absence of surgical/medical intervention, many cancers will progress to fatal disease. Moreover, the random parameters of tumor growth and spread that are the result of small changes in growth conditions means that therapeutic intervention in many cases cannot be expected to produce consistent, predictable results. Apparent evidence for this is the fact that therapeutic outcome for many later stage cancers may vary widely from one patient to another, even when the malignancies are of the same origin and approximate stage of disease and the treatment protocols are the same.
Can we Identify Attractors Critical to Defining the Limits of the Deterministic Box?
The downside of Chaos Theory as it applies to biological systems involves the concept that perturbations of non-linear systems that result from small changes in initial conditions generate unpredictable outcomes because one can never completely define the initial conditions. Yet, if one can identify the attractors that define the limits or possible trajectories of a biological system, it may be possible to more describe more accurately the initial state and the probabilistic fate potential associated with small system perturbations. One may define the system as a deterministic box in which the systemic environment (or micro-environment) defines the limits of the cellular system via bioenergetic attractors whose effects are manifest in microevolutionary fitness, i.e., survival. The attractor states are dynamic and are the result of the dynamic interaction between redox regulation that may be systemically driven and intracellular bioenergetic mechanisms that define the energy state of the cell.
In defining cancer and chaos, can we identify the initial system perturbation?
A large body of research supports the notion that cell-Extracellular Matrix (ECM) detachment is a critical initiating event in solid tumor formation. In cell culture systems, the usual result is cell death or anoikis; however, an alternative trajectory may be achieved if the detached cells acquire the capacity for anchorage independent survival, a hallmark of malignancy (Figure 4). Thus, ECM detachment can be a primary initiator of transformation; it is driven by mitotic/oxidative stress. A non-linear model of tumorigenesis predicts large scale effects. The possibilities include cell death, reattachment/renewal or survival/transformation. Some of the steps leading to the latter include: glycolytic metabolic shift, reactive oxygen species (ROS) mediated mitochondrial damage and the induction of pro-inflammatory and pro-survival signals resulting from oxidative stress.
A New Therapeutic Paradigm: Targeting the Deterministic Box
What is the deterministic box that defines the limits of cancer progression? Dalgleish [1] proposed that immune system variables play a very important role in defining the systemic deterministic environment. There is considerable supporting evidence for this view, beginning with the correlation between immunosuppression and cancer incidence rates. However, there are additional systemic factors that may be equally important, including chronic inflammation and mitochondrial dysfunction/ROS levels associated with aging. In fact, immune system functions are intimately linked to REDOX metabolism. It is well-known that radiation and some chemotherapy drugs exert their anti-cancer effects via oxidative stress. In addition, it has been argued that anti-oxidants may actually decrease their beneficial therapeutic effects by interfering with their effects on redox parameters. Supplemental anti-oxidants may protect tumor cells against chemotherapy and radiotherapy induced cell death, thus reducing patient survival rates. One the other hand, there is a significant amount of data to support the anti-cancer preventive effects of diets rich in anti-oxidants.
These apparently paradoxical observations may be reconciled if one applies the non-linear deterministic model of oncogenesis and disease progression driven by redox parameters that directly impact cellular bioenergetic mechanisms. Just as oxidizing agents are used in chemotherapy to exacerbate oxidative stress, perhaps anti-oxidants exert anti-cancer effects in the same way-by exacerbating oxidative stress to induce cell death. This model would explain the documented anti-cancer effects of some anti-oxidants as well as their selective cytotoxicity in tumor cells versus normal cells.
Moreover, this model proposes that ROS play central role in tumor formation and progression, both of which require altered energy metabolism. ROS initiate glycolytic activation coincidental with mitochondrial aerobic energy reduction as the basis of the Warburg effect. Anti-oxidants block these bioenergetic changes responsible for tumor formation/progression.
Conclusion
The relationship between chaos and cancer suggests that therapeutic approaches that target chaotic tumor growth and/or specific gene dysfunction may be insufficient to induce the longterm remission of many cancers, particularly later stage cancers that involve multiple chromosome/gene mutations and highly anaplastic growth patterns. Perhaps greater focus should be placed on targeting the “deterministic box” that shapes the ultimate disease trajectory. Non-linear redox parameters may both initiate and set deterministic limits of cell survival/death phenomena. The preventive effects of antioxidants may, therefore, result from their stabilizing effects on ROS/ redox equilibrium. Moreover, exacerbation of ROS imbalances may be an important therapeutic mechanism in the treatment of advanced cancer. To paraphrase Paget, “the seed cannot exist without the soil”. Thus, on this biological stage, it is imperative to target the deterministic, systemic box that pulls its silent strings around the tumor cells to determine their path and the way the story ends.
References
- Dalgleish A (1999) The relevance of non-linear mathematics (chaos theory) to the treatment of cancer, the role of the immune response and the potential for vaccines. QJM 92: 347-59.
- Davoli T, Xu AW, Mengwasser KE, Sack LM, Yoon JC, et al. (2013) Cumulative Haploinsufficiency and Triplosensitivity Drive Aneuploidy Patterns and Shape the Cancer Genome. Cell 155: 948-962.
- Klein K, Maier T, Hirschfeld-Warneken VC, Spatz JP (2013) Marker-free phenotyping of tumor cells by fractal analysis of reflection interference contrast microscopy images. Nano Lett 13: 5474-5479.
Relevant Topics
- Acupuncture Therapy
- Advances in Naturopathic Treatment
- African Traditional Medicine
- Australian Traditional Medicine
- Chinese Acupuncture
- Chinese Medicine
- Clinical Naturopathic Medicine
- Clinical Naturopathy
- Herbal Medicines
- Holistic Cancer Treatment
- Holistic health
- Holistic Nutrition
- Homeopathic Medicine
- Homeopathic Remedies
- Japanese Traditional Medicine
- Korean Traditional Medicine
- Natural Remedies
- Naturopathic Medicine
- Naturopathic Practioner Communications
- Naturopathy
- Naturopathy Clinic Management
- Traditional Asian Medicine
- Traditional medicine
- Traditional Plant Medicine
- UK naturopathy
Recommended Journals
Article Tools
Article Usage
- Total views: 2584
- [From(publication date):
August-2017 - Aug 20, 2025] - Breakdown by view type
- HTML page views : 1707
- PDF downloads : 877