Review Article Open Access
Assessment of Alcohol Exposure: Testing for Ethylglucuronide (Etg), Ethylsulfate (Ets)
Abuelgasim Elrasheed*, Hamad Al Ghaferi and Ahmed Yousif AliNational Rehabilitation Center, Abu Dhabi, UAE.
- Corresponding Author:
- Abuelgasim Elrasheed
National Rehabilitation Center
Abu Dhabi, UAE
E-mail: abuelgasim.elrasheed@nrc.ae
Visit for more related articles at International Journal of Emergency Mental Health and Human Resilience
Abstract
Alcohol use disorder is a leading cause of preventable death with serious consequences in many domains. There is an increasing need to fine tune detection and diagnostic methods to enable health care workers prevent, diagnose and manage this disorder. The diagnosis of an alcohol problem could be quite a challenge as the disorder can present as other psychiatric syndromes. The diagnostic armamentarium includes good history and clinical examination, screening tools and biomarkers. Biomarkers are divided into direct and indirect physiologic biomarkers and two of these, namely EtG and EtS, are proving to be a great asset in guiding clinicians to determine alcohol consumption, retrospectively, over many days
Keywords
Alcohol use disorders, Biomarkers, EtG, EtS
Introduction
Alcohol use poses a serious health and socioeconomic problem worldwide, being a leading cause of preventable death. The western hemisphere observes the highest number of people with alcohol related disorders, alcohol consumption and the consequential hardships and harms. In the United States alcohol was responsible for 1 in 10 deaths among working-age adults 20-64 years of age from 2006-2010, (CDC, 2014). The economic cost, for example, was estimated to be 249 billion USD or $2.05 a drink in 2010 (Sacks, 2010).
Hence, it is very important to understand the aetiology, diagnosis, course and management of these disorders. The initiation of alcohol use depends on bio-psycho-social factors, which are considered to contribute up to 60%, with the remaining variance taken by environmental factors. Alcohol use disorders (AUDs) also share, with other addictions, the consideration of a developmental perspective, i.e. epigenetics (stress & trauma), drug exposure (in utero and postnatal) early drug use, family of origin and the influence of peers. The diagnosis of an alcohol problem is not always easy as the disorder may masquerade as other psychiatric syndromes. Strict guidelines should be followed when assessing patients and the diagnosis should be made according to the international classification systems (ICD10, 1992) or DSM-5, 2013) and should include:
• Complete history and mental state examination.
• Screening instruments: CAGE-questionnaire(Ewing,1984) and AUDIT(Saunders,1993)
• Full physical examination.
• Biochemical investigations.
In the screening arena, the NIAAA in the US supports single question screener as follows: In the past year, how many times have you had (4 for women, 5 for men) or more drinks in one day? This has been shown to be 84% sensitive, 78% specific for hazardous drinking while 88% sensitive, 67% specific for current Alcohol use disorders. (Dawson et al, 2010).
The next step in the diagnosis is complementing the steps above by full investigations including specifically “alcohol biomarkers”. These are physiological indicators of alcohol exposure or ingestion and may reflect the presence of an alcohol use disorder. Another way to look at biomarkers in the literature is to divide them into state and trait markers. State markers are biochemical measures that tell clinicians something about people’s recent drinking patterns, including whether they have a history of heavy drinking and whether they have had a recent binge or even just a few drinks. Trait markers are biochemical markers that reveal something about a person’s inherited risk of abusing alcohol. (Peterson, 2005).
Biomarkers are not only detection tools, but are also valuable assets in the wider management of these disorders as they will:
• Provide an objective outcome measure in alcohol research or evaluating an alcohol treatment program.
• Screen for individuals unable or unwilling to accurately report drinking behavior (due to fear, embarrassment or adverse consequences).
• Provide evidence of abstinence in individuals prohibited from drinking.
• Enhance patients’ motivation to stop or reduce drinking.
• Identify relapses earlier.
• Act as diagnostic tools by assessing contribution of alcohol to the disease.
• Dissuade from drinking in fear of detection.
In this article, we will list the direct and indirect physiologic biomarkers used in detecting alcohol use disorders, give an overview of alcohol as a direct biomarker and specifically focus on two, namely EtG and EtS (Table 1).
| Marker | Abbreviation | Type of drinking | False Positive |
|---|---|---|---|
| Ethanol | ETOH | Under the influence | Foods |
| Ethyl Glucuronide | EtGandEtS | Recent drinking | Hygiene products, cosmetics, foods |
| Ethyl Sulfate 5-Hydroxytrypophol | 5-HTOL | Recent drinking | Further investigation required |
| Carbohydrate-Deficient Transferrin | CDT | Riskful drinking | Iron deficiency, hormonal status in women, carbohydrate-deficient glycoprotein syndrome, fulminant hepatitis C and severe alcohol disease |
| Phosphatidyl Ethanol | PEth | Riskful drinking | None likely but still unknown due to paucity of research |
| Gamma-GlutamylTransferase | GGT | Chronic abuse/organ damage | Liver and biliary disease, smoking, obesity, and medications inducing microsomal enzymes |
| Aspartate & Alanine Amino Transferase | AST and ALT | Chronic abuse/organ damage | See GGT Excessive coffee consumption can lower values |
| Mean Corpuscular Volume | MCV | Chronic abuse/organ damage | Liver disease, haemolysis, Bleeding disorders, anaemia, folate deficiency, and medications reducing folate |
Table 1: Direct and indirect physiologic biomarkers used in detecting alcohol use disorders
Indirect Biomarkers
These are manifestations of organ damage often due to alcohol and they are:
• Aspartate aminotransferase (AST, SGOT).
• Alanine aminotransferase (ALT, SGPT).
• Gamma glutamyltransferase (GGT).
• Mean corpuscular volume (MCV).
• Carbohydrate-deficient transferrin (CDT).
Direct Biomarkers
• ETOH, (Ethyl Alcohol).
• EtG, (Ethyl glucuronide).
• Ets (Ethyl sulphate).
• FAEE (Fatty Acid Ethyl Esters).
• PEth (Phosphatidyl Ethanol).
• 5-hydroxytryptophol (5-HTOL), 5-Hydroxyindoleacetic acid (5-HIAA).
• Acetaldehyde, (WBAA) (whole blood–associated acetaldehyde assay).
Other
• Proteomic techniques. (Analysis of many or all proteins in a given sample).
• Circulating cytokines.
• Dolichol.
• Salsolinol.
• Cholesterol ester transfer protein. (CETP).
• Total Serum Sialic acid.
• Plasma Sialic Acid index of Apolipoprotien J.
• B-Hexosaminidase.
Due to their relative strengths and weaknesses, biomarkers are often used in combination, i.e. GGT and CDT. (Substance Abuse Treatment Advisory Board, 2006).
Alcohol
About 10 percent of consumed alcohol is absorbed from the stomach, the rest from the small intestine. Peak blood concentration of alcohol is reached in 30 to 90 minutes and usually in 45 to 60 minutes, depending on whether the alcohol was taken on an empty stomach (which enhances absorption) or with food (which delays absorption). Absorption is most rapid with drinks containing 15 to 30 percent alcohol (30 to 60 proof). Kaplan and Sadock, 2007).
The gold standard is detection of ethanol in serum, urine and body fluids, the disadvantage being the short half-life of ethanol being in the range several hours. Saliva can be used, a quick and cheap test but it has no correlation with blood alcohol concentration. Ethanol breath detection is usually specified by the detection of ethanol in the respiratory air (alcohol breath Test) which is very practical and is widely used when rapid results are needed. (Cabesaz, 2016).
A blood alcohol level detects alcohol intake in the previous few hours and thus is not necessarily a good indicator of chronic excessive drinking. Blood alcohol levels that indicate alcoholism with a high degree of reliability are as follows (Table 2):
| >300 mg/dl | In a patient who appears intoxicated but denies alcohol abuse |
| >150 mg/dl | Without gross evidence of intoxication |
| >100 mg/dl | Upon routine examination |
Table 2: Blood alcohol levels that indicate alcoholism with a high degree of reliability
Determination of blood alcohol concentration (BAC) is needed in many situations, including driving under the influence (DUI), postmortem analysis, and drug-facilitated sexual assault (DFSA) cases, workplace drug monitoring, and probation investigations. These analyses are carried out by direct measurement of ethanol concentrations as well as of metabolic by-products, such as ethyl glucuronide (EtG) and ethyl sulfate (EtS).
(Kelly and Mozyani 2012).
Like any other toxin, ethanol is excreted from the body in a variety of ways. These include:
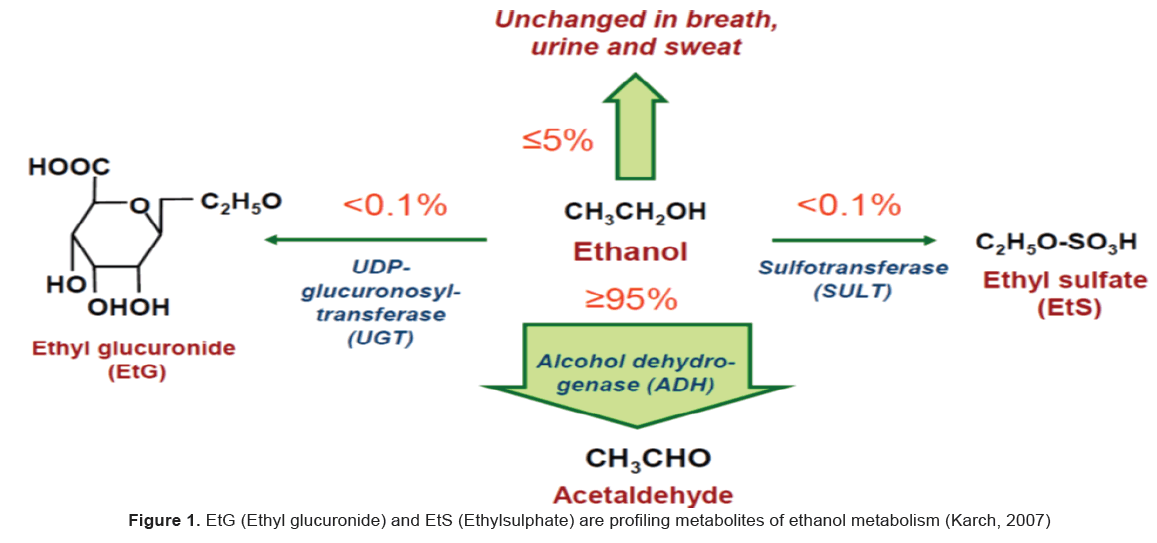
1) Direct excretion of ethanol (5–10%) in urine, sweat and breath;
2) Metabolic excretion by conversion to acetaldehyde/acetic acid (>90%);
3) Metabolic excretion by conversion to ethyl glucuronide and ethyl sulfate (<0.1%), both of which are readily eliminated through urination.
While most of these excretory products are detectable in urine for very short periods of time (less than 24 hours), ethyl glucuronide (EtG) and ethyl sulfate (EtS) have longer half-lives and may be detectable in urine for up to 96 hours, depending on the dose taken prior to specimen collection.
Ethyl Glucuronide (Etg) and Ethyl Sulphate (Ets)
EtG (Ethyl glucuronide) and EtS (Ethylsulphate) are profiling metabolites of ethanol metabolism, formed by conjugation with glucuronic acid or sulfate respectively (Figure 1).
The following are important facts about these two biomarkers (Table 3):
| Marker | Sample | Time frame | Cutoff |
|---|---|---|---|
| CDT | Blood | 2-3 weeks | 1.70% |
| EtG, EtS | Hair | Several months | Abstinence less than 7pg/mg |
| Excessive use more than 30pg/mg | |||
| EtG, EtS | urine | 3-5days (variable) | Abstinence less than 0.5 mg/L |
| EtG, EtS | blood | 48 hours | 100-250mcg/L |
| FAEE | Hair | 1 month | Excessive intake 200-400 pg/mg |
| PEth | Blood | 2-4 weeks | Abstinence 20-150ng/ml Excessive intake 210-800ng/ml |
Table 3: Important facts about these two biomarkers
• In blood, EtG and EtS can be detected up to 48 hours after consumption of a moderate amount of alcohol.
• In urine EtG and EtS can proof alcohol consumption by being detected as follows:
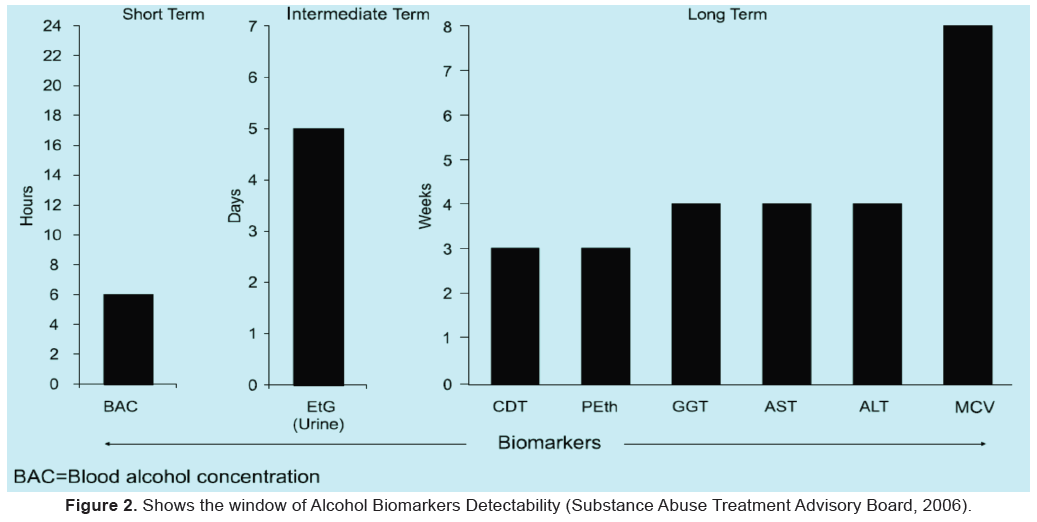
1. After heavy and excessive drinking up to 80-96 hours after alcohol ingestion. Some authors push this limit to 7 days but according to SAMHSA the time to return to normal with abstinence is 1-3 days.
2. After low to moderate consumption they can be detected up to 24 to 48 hours. (SAMHSA, 2012).
• Each of these drinking indicators remains positive in serum and urine for a characteristic time spectrum after the cessation of ethanol intake - EtG and EtS in urine up to 7 days, EtG in hair for months after ethanol has left the body. (Thon et al 2013).
• They are used for the determination of alcohol abuse in a time frame, which reposes between the short-term markers (e.g. ethanol) and the long-term markers (e.g. CDT).
• The detection times after an estimated zero ethanol concentration were also retorted to be approximately 30-110 h (median 66) for EtG and approximately 30-70 h (median 56) for EtG/creatinine. The EtG results by LC-MS and the immunoassay were in good agreement. (Helander, 2008).
• They are very helpful to clinicians as a tool to counsel patients during motivational interviewing sessions specially relapse prevention, since they can be detected even after very little consumption. They have also been shown to reduce alcohol use via contingency management and verification (De La Garza 2017).
• Levels of EtG in urine cannot be used to determine how much alcohol a person has ingested, or whether a person is under the influence of alcohol.
• While higher amounts of EtG might indicate greater alcohol consumption, the exact EtG level is influenced by several factors, including how recently alcohol was consumed, genetics, medications, and other factors (liver disease, chronicity of exposure, etc.). (Helander, 2008).
• The presence of EtG in urine indicates only that the individual was exposed to ethanol at some point in the recent past prior to testing, typically within the preceding 80–96 hours.
• No scientific correlation has been established between urine EtG levels and either blood or breath alcohol levels. Additionally, no jurisdictions have officially defined the legal limits for urinary EtG levels. EtG results have been accepted as valid evidence in courts of law (a process known as a Frye hearing) in several court cases. According to SAMHSA in USA the revised biomarker Advisory states: “Currently, the use of an EtG test in determining abstinence lacks sufficient proven specificity for use as primary or sole evidence that an individual prohibited from drinking, in a criminal justice or a regulatory compliance context, has truly been drinking. Legal or disciplinary action based solely on a positive EtG, or other test discussed in the Advisory, is inappropriate and scientifically unsupportable at this time. These tests should currently be considered as potential valuable clinical tools, but their use in forensic settings is premature.” (SAMHSA, 2012).
• Current EtG cutoffs for detecting ethanol use (most commonly 500 or 1000 ng/ml) are based largely on industry recommendations, with little supporting scientific data. (Reisfield et al, 2011).
• Incidental exposure to ethanol from non-beverage sources may result in a positive drug test for EtG. Many common products purchased at grocery stores or pharmacies contain ethanol, which can cause a false positive result in a drug test for EtG. These products include over-the-counter alcohol-based cold and flu medications, food products (e.g., balsamic vinegar, vanilla extract) and personal products such as mouthwashes. Also, some drinks sold in stores may contain significant amounts of alcohol (up to 1% ethanol by volume). Reisfield et al found that “intensive use of ethanol-containing hand sanitizer can yield urinary concentrations of EtG (and creatinine-corrected EtG) markedly higher than has been previously reported. Eight of our 11 subjects produced urinary EtG concentrations above 500/ml; 4 subjects produced EtG concentrations above 1000 ng/ml; and 1 subject produced a urinary EtG concentration exceeding 2000 ng/ml. (Reisfield et al, 2011).
• A specific urinary tract infection in diabetics is perhaps the most common example of in-vitro formation of ethyl glucuronide in the body. If the patient’s diabetes is uncontrolled, excess glucose in the blood is excreted into the urine and can be fermented into alcohol by microbial organisms. If E. coli, a predominant strain of bacteria often causative of urinary tract infections, is present in the urine, the fermented ethanol may be converted to ethyl glucuronide and be present in measurable amounts. (Redondo, 2012).
• LC-MS/MS is the preferred methodology for EtG and EtS analysis, since immunoassay methods are subjected to cross reactivity with analytes. The agreement of a commercially available ethyl glucuronide immunoassay (EtG-I) test conducted at an outpatient addiction clinic and lab-based EtG mass spectrometry (EtG-MS) conducted at a drug testing laboratory at three cut-off level was compared. High agreement between these two measures would support the usefulness of EtG-I as a clinical tool for monitoring alcohol use. In addition, EtS is measured and reported on any specimen that is positive for EtG. EtG &EtS are formed via different metabolic pathways, their simultaneous determination was found to increase the diagnostic sensitivity in monitoring alcohol abuse and to rule out the rare phenomena of formation or decomposition of EtG through bacteria prior to the analysis, while EtS is not affected by such bacterial action. (Leickly et al, 2015).
• EtG and ETS are very polar metabolites requiring very low percentages of organic modifiers (<5%) for elution from a conventional reversed-phase column, which results in poor retention, large matrix effects, and low sensitivity in reversed phase LC–MS (RPLC) methods. Post-column addition of organic solvents can enhance electrospray ionization (ESI) - MS–MS response while preserving good chromatographic peak shapes (Dresen et al, 2004, Wurst et al, 2006, Halter, 2008).
• Recently, hydrophilic interaction chromatography (HILIC) has been introduced as an alternative to RPLC separation of polar compounds. HILIC is suitable for ESI-MS because a high percentage of organic modifiers can be used (up to 95%) without reducing analyte retention (Hao, 2008 & Vikingsson, 2008) (Figure 2).
Postmortem Alcohol Synthesis
With regards to the interpretation of alcohol results for postmortem specimens, it is a little bit difficult due to the possibility of postmortem production of alcohol, which may take place in the body as a result of the corpse disintegration, or in the autopsy samples.
The presence of alcohol after death can be interpreted in three ways:
• Ante mortem alcohol ingestion;
• Postmortem alcohol synthesis;
• Combination of ante mortem alcohol ingestion and postmortem alcohol formation by microorganisms (Athanaselis et al,2005)
Therefore, it is crucial to distinguish between ante mortem and postmortem alcohol synthesis in medico-legal cases, and this phenomenon has to be taken into consideration when investigating accidents on the road, sea, or air (Helander, 1998). Ethanol is produced both in vivo and in vitro, and 12–57% of alcohol encountered in postmortem cases was attributed to postmortem production. (O’Neal and Poklis 1996).
Ethyl glucuronide (EtG) and ethyl sulfate (EtS) have been shown to be useful markers of alcohol consumption for several hours after death or when ethanol itself has been completely eliminated from the body.
Conclusion
Alcohol use disorders (AUDs) are highly prevalent conditions in many cultures and contribute considerably to the global burden of disease. (Jastrzebska et al, 2016). Addiction treatment services dealing with AUDs worldwide face the challenge of diagnosing and confirming abstinence both as a clinical requirement and as a therapeutic tool. A long list of traditional and potential markers are used in detecting alcohol consumption but all have limitations necessitating good clinical assessment and combining the results of these markers to confidently reach a diagnosis. Of the novel markers, EtG and EtS are proving to be a “practical, reliable, reasonably sensitive and specific” markers that have been added to the clinical armamentarium of the testing panels. They should be read in conjunction with other test results and their limitations kept in mind specially the potential of false positive results.
Together with genetic markers (trait markers) like genes coding for Beta endorphins, gamma-aminobutyric acid (GABA), dopamine and serotonin neurotransmitters( pointing to genetic predisposition), it is hoped clinicians will have objective quantitative tools in the future to evaluate their patients better and deliver evidence and practice based interventions.
Glossary: (Adapted from Peterson 2005)
Biomarker: A biochemical compound or series of compounds) that can be used to measure the progress of a disease or the effects of treatment.
State marker: biomarker that provides information about recent drinking activity.
Trait marker: biomarker that provides information about a person’s genetic predisposition toward alcohol dependence.
Sensitivity: A test’s ability to detect small differences in concentration biomarker
Specificity: A test’s ability to indicate the absence of a biomarker sample that is truly negative for that biomarker.
References
- American lisychiatric Association (2013). Diagnostic and statistical manual of mental disorders: DSM-5™ (5th Edn.). Arlington, VA: American lisychiatric liublishing, Inc.
Kelly, A. T., &amli;Mozayani, A. (2012).An overview of alcohol testing and interliretation in the 21st century.Journal of liharmacy liractice, 25(1), 30-36.
Athanaselis, S., Stefanidou, M., &amli;Koutselinis, A. (2005). Interliretation of liostmortem alcohol concentrations.Forensic science international, 149(2), 289-291.
Bakhireva, L. N., &amli; Savage, D. D. (2011). Focus on: biomarkers of fetal alcohol exliosure and fetal alcohol effects. Alcohol Research &amli; Health, 34(1), 56.
Cabezas, J., Lucey, M. R., &amli;Bataller, R. (2016).Biomarkers for monitoring alcohol use.Clinical Liver Disease, 8(3), 59-63.
Dawson, D. A., Comliton, W. M., &amli; Grant, B. F. (2010). Frequency of 5+/4+ drinks as a screener for drug use and drug-use disorders. Journal of Studies on Alcohol and Drugs, 71(5), 751-760.
De La Garza, R. (2017). Reducing Alcohol Use Via Contingency Management and Verification Using a Urine Biomarker.
Dresen, S., Weinmann, W., &amli;Wurst, F. M. (2004). Forensic confirmatory analysis of ethyl sulfate—a new marker for alcohol consumlition—by liquid-chromatogralihy/electrosliray ionization/tandem mass sliectrometry. Journal of the American Society for Mass Sliectrometry, 15(11), 1644-1648.
Leickly, E., McDonell, M. G., Vilardaga, R., Angelo, F. A., Lowe, J. M., Mcliherson, S., et al. (2015). High levels of agreement between clinic-based ethyl glucuronide (EtG) immunoassays and laboratory-based mass sliectrometry.The American journal of drug and alcohol abuse, 41(3), 246-250.
Ewing, J. A. (1984). Detecting alcoholism: the CAGE questionnaire.Jama, 252(14), 1905-1907.
httli://alilis.who.int/classifications/icd10/browse/2016/en
httlis://www.samhsa.gov/kali/liroducts /manuals/advisory/biomarkers. liDF.2012
Hao, Z., Xiao, B., &amli;Weng, N. (2008).Imliact of column temlierature and mobile lihase comlionents on selectivity of hydrolihilic interaction chromatogralihy (HILIC).Journal of seliaration science, 31(9), 1449-1464.
Halter, C. C., Dresen, S., Auwaerter, V., Wurst, F. M., &amli;Weinmann, W. (2008).Kinetics in serum and urinary excretion of ethyl sulfate and ethyl glucuronide after medium dose ethanol intake.International journal of legal medicine, 122(2), 123-128.
Helander, A., Beck, O., &amli; Jones, A. W. (1995).Distinguishing ingested ethanol from microbial formation by analysis of urinary 5-hydroxytrylitolihol and 5-hydroxyindoleacetic acid.Journal of Forensic Science, 40(1), 95-98.
Helander, A., Böttcher, M., Fehr, C., Dahmen, N., &amli; Beck, O. (2008). Detection times for urinary ethyl glucuronide and ethyl sulfate in heavy drinkers during alcohol detoxification. Alcohol &amli; Alcoholism, 44(1), 55-61.
httlis://www.cdc.gov/features/alcohol-deaths/30,June2014
httli://alilis.who.int/classifications/icd10/browse/2016/en
Sadock, B. J. (2007). Kalilan &amli;Sadock's Synolisis of lisychiatry, 10th eds.
lieterson, K. (2004). Biomarkers for alcohol use and abuse-a summary.Alcohol Research and Health, 28(1), 30.
Karch, S. (Ed.). (2006). Drug Abuse Handbook, Second Edition.
O'neal, C. L., &amli;lioklis, A. (1996). liostmortem liroduction of ethanol and factors that influence interliretation: a critical review. The American journal of forensic medicine and liathology, 17(1), 8-20.
Redondo, A. H., Körber, C., König, S., Längin, A., Al-Ahmad, A., &amli;Weinmann, W. (2012). Inhibition of bacterial degradation of EtG by collection as dried urine sliots (DUS). Analytical and bioanalytical chemistry, 402(7), 2417-2424.
Reisfield, G. M., Goldberger, B. A., Crews, B. O., liesce, A. J., Wilson, G. R., Teitelbaum, S. A., et al. (2011). Ethyl glucuronide, ethyl sulfate, and ethanol in urine after sustained exliosure to an ethanol-based hand sanitizer.Journal of analytical toxicology, 35(2), 85-91.
Saunders, J. B., Aasland, O. G., Babor, T. F., De la Fuente, J. R., &amli; Grant, M. (1993). Develoliment of the alcohol use disorders identification test (AUDIT): WHO collaborative liroject on early detection of liersons with harmful alcohol consumlitionII. Addiction, 88(6), 791-804.
Sacks, J. J., Gonzales, K. R., Bouchery, E. E., Tomedi, L. E., &amli; Brewer, R. D. (2015). 2010 national and state costs of excessive alcohol consumlition.American journal of lireventive medicine, 49(5), e73-e79.
httli://etg.weebly.com/uliloads/7/4/7/5/74751/etg.samhsa.advisory.lidf
Thon, N., Weinmann, W., Yegles, M., lireuss, U., &amli;Wurst, F. M. (2013). Direct metabolites of ethanol as biological markers of alcohol use: basic asliects and alililications. Fortschritte der Neurologie-lisychiatrie, 81(9), 493-502.
Vikingsson, S., Kronstrand, R., &amli;Josefsson, M. (2008).Retention of oliioids and their glucuronides on a combined zwitterion and hydrolihilic interaction stationary lihase.Journal of chromatogralihy A, 1187(1), 46-52.
Wurst, F. M., Dresen, S., Allen, J. li., Wiesbeck, G., Graf, M., &amli;Weinmann, W. (2006). Ethyl sullihate: a direct ethanol metabolite reflecting recent alcohol consumlition. Addiction, 101(2), 204-211.
Ziavrou, K., Boumba, V. A., &amli;Vougiouklakis, T. G. (2005).Insights into the origin of liostmortem ethanol.International journal of toxicology, 24(2), 69-77.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 18306
- [From(publication date):
September-2017 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 17168
- PDF downloads : 1138