Idiopathic Pleuroparenchymal Fibroelastosis as a Rare Entity within Idiopathic Interstitial Pneumonias: A Case Report
Received: 02-Jun-2016 / Accepted Date: 08-Jun-2016 / Published Date: 13-Jun-2016 DOI: 10.4172/2476-2024.1000115
Keywords: Fibroelastosis; Hypercapnia; Platythorax
6722Introduction
Pleuroparenchymal fibroelastosis (PPFE) is an upper lobe predominant progressive fibrosis with specific pathologic findings including visceral pleural thickening, subpleural elastosis, and intraalveolar collagenous fibrosis. This uncommon condition firstly described in the Japanese literature in 1992 [1] and subsequently reported in English literature in 2004 [2]. PPFE is now recognized by the American Thoracic Society (ATS)/European Respiratory Society (ERS) in their most recent statement on idiopathic interstitial pneumonia in the category of rare idiopathic interstitial pneumonias [3]. Many of the PPFE cases are associated with underlying conditions but the majority is idiopathic. In the English literature, about forty cases of idiopathic PPFE (IPPFE) have been described. We aimed to present the clinical, radiological and histopathological features of PPFE in a case diagnosed as IPPFE.
Case Presentation
A 37-year-old, non-smoker, woman had progressive dyspnea and nonproductive cough for six years and she had cachexia and lost 15 kg weight within last one year. On her medical history, she experienced recurrent respiratory tract infections starting from the childhood. There was no any other medical problem or drug use. Family history was unremarkable. She was a housewife without known environmental exposure including asbestos. She had been investigated in another tertiary care center for tuberculosis and interstitial lung disease. Videoassisted thoracoscopic surgery (VATS) was made with right upper and middle lobe wedge biopsies. Tuberculosis cultures of biopsy samples were negative and pathological investigations revealed nonspecific pulmonary fibrosis. Then patient was referred to our clinic for clarifying the interstitial lung disease etiology.
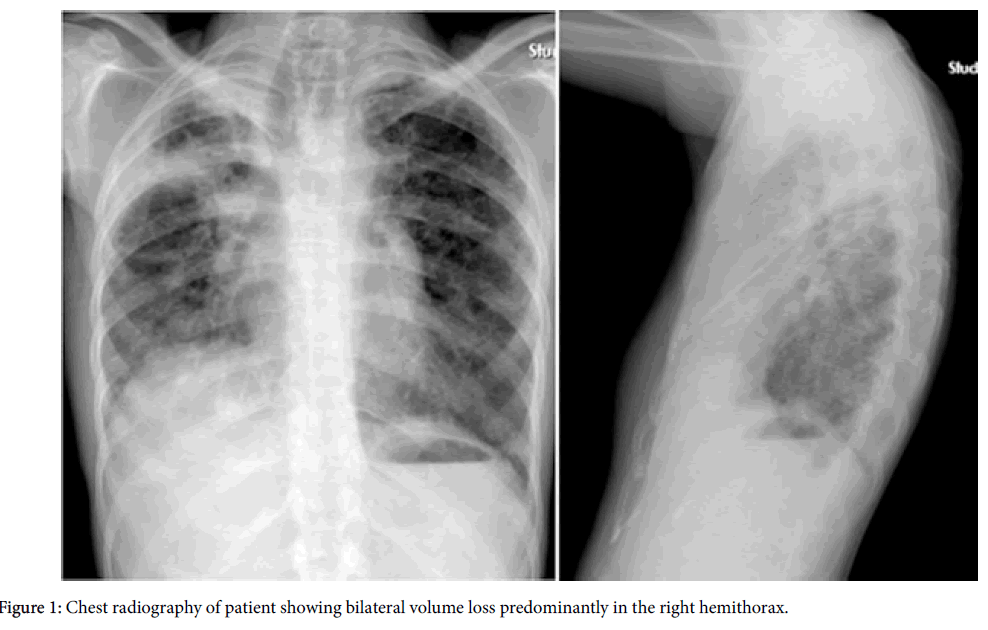
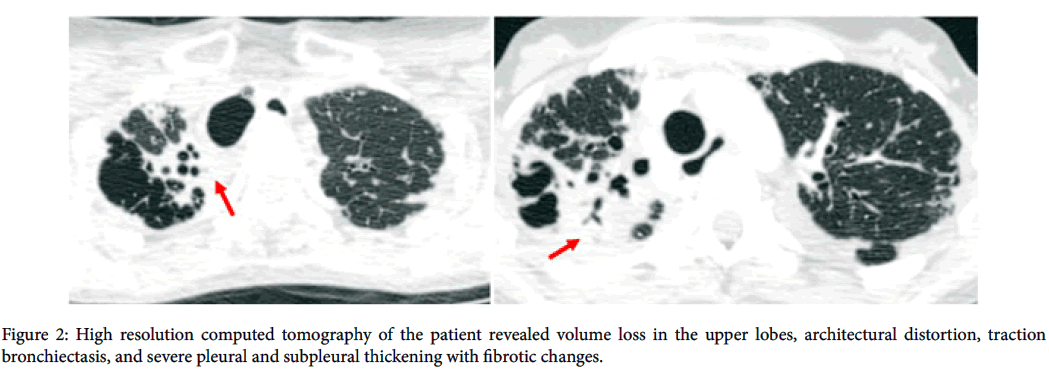
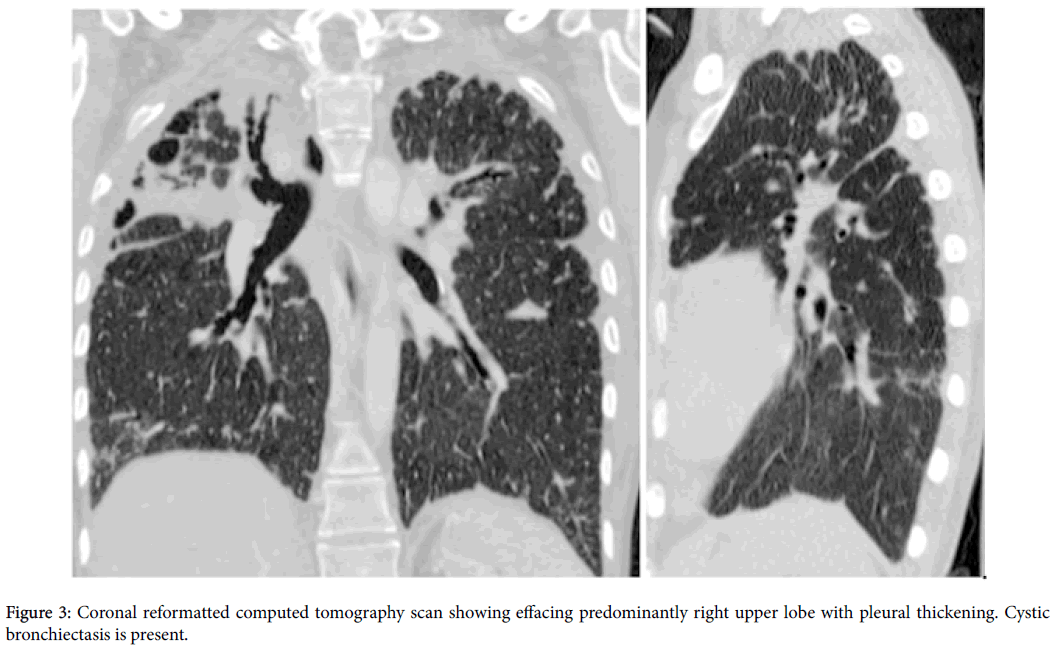
On physical examination, she was thin, cachectic with a body mass index of 14.0 kg/m2 and she was tachypneic and cyanotic. Chest auscultation revealed diminished breath sounds and bronchial sound was noted on the right lower lung zone. Cardiovascular, abdominal and rheumatologic examinations were normal. Arterial blood gas analysis demonstrated hypoxemia and hypercapnia; pH: 7.45, PaO 2:44 mmHg, PaCO 2:58 mmHg, HCO 3:39 mmol/L, SpO 2:84% at room air. Pulmonary function test results revealed restrictive pattern as: forced vital capacity (FVC) 14% of predicted, forced expiratory volume in one second (FEV1) 17% of predicted, FEV1/FVC:100% predicted, total lung capacity (TLC): 15% of predicted, residual volume (RV):36% of predicted. Also diffusion capacity of the lung for carbon monoxide was reduced. Chest X-ray showed significant reduction of the ratio of anteroposterior diameter of the thorax to the transverse diameter consistent with platythorax; besides this bilaterally but most significantly right apical pleural thickening, decreased right lung volume with elevated diaphragm and right upper lobe destruction and fibrosis was observed (Figure 1). High resolution computed tomography (HRCT) scan showed bilateral upper lobe predominant irregular pleuroparenchymal thickening, architectural distortion, and traction bronchiectasis without involvement of the lower lobes (Figures 2 and 3).
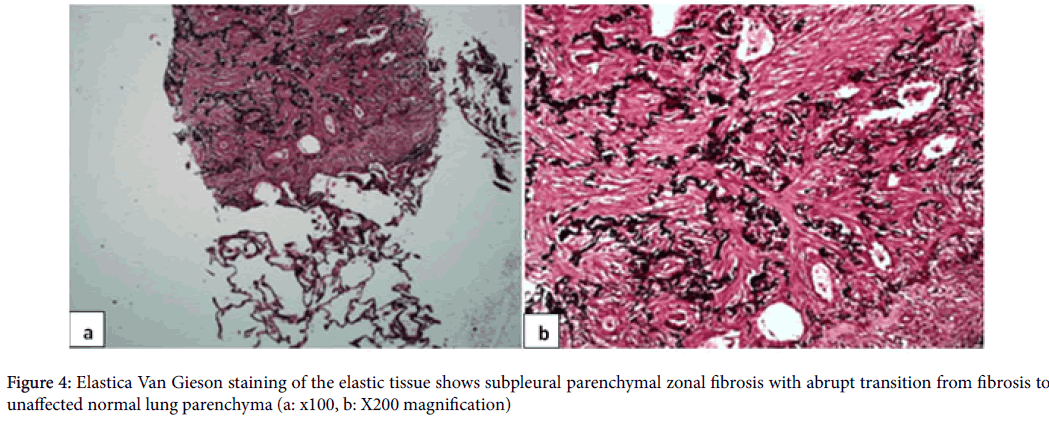
These HRCT characteristics posed a diagnostic dilemma for the radiologist, who made the differential diagnosis among PPFE, sarcoidosis, and chronic hypersensitivity pneumonia. Anti-nuclear antibody (ANA) and anti-neutrophil cytoplasmic antibodies (ANCA) were negative. Alpha-1 antitrypsin level was normal. Bronchoscopy was performed and bronchoalveolar lavage (BAL) differential cell count was 98% macrophages. Microbiologic analysis of BAL was negative for bacteria, fungi and mycobacteria. Histological specimens of surgical lung biopsy were obtained from other center and investigated once again in our pathology department. Histopathological findings from elastic fiber staining revealed distortion and marked pleural thickening, with the evidence of dense fibroelastic tissue in the pleura and alveolar walls, together with sparse fibroblastic foci near the transition from the lung to the pleura (Figure 4). On the basis of the clinical and radiological data, a diagnosis of IPPFE was made pathologically.
Discussion
There is currently no consensus regarding the diagnostic criteria used in order to classify IPPFE, as well as regarding whether or not it represents a real, new, specific entity.
The etiology of PPFE is unknown, with most cases considered idiopathic. Familial cases of PPFE and other interstitial lung disease have been reported, and the condition may be more common in the Japanese, suggesting genetic predisposition plays a role in pathogenesis [2,4]. PPFE has been associated with bone marrow transplantation and lung transplantation, and recurrent chest infections have been reported in 14% to 50% of all cases [5,6]. Various organisms have been isolated including Aspergillus species and Mycobacterium avium-intracellulare complex leading authors to postulate that repeated inflammatory damage leads to intraalveolar fibroelastosis and eventually to PPFE [7,8].
Our patient also had a history of recurrent respiratory tract infections but bronchoscopic samples didn't reveal any specific organism to suggest infectious etiology.
Clinically, patients with IPPFE usually present with chronic respiratory symptoms such as dyspnea and dry cough as in our case. Most patients are between 49 and 57 years old at the time of diagnosis. No active smokers have been reported, and the majority of cases occur in never smokers (62%-75%) [8,9]. Our case was younger than other cases in the literature and she was also never smoker.
The diagnosis of IPPFE is established on the basis of clinical, radiological, and histopathological findings. Anteroposterior flattening of the chest wall may occur. Pulmonary function test abnormalities are most commonly consistent with restrictive lung disease with reduced lung volumes and diffusion capacity of the lung for carbon monoxide [10]. Chest HRCT scans show marked subpleural consolidations with irregular thickening; distortion, usually located in upper lung lobes (“pleural cap”); subpleural bronchiolectasis; reticular opacities; and, in some cases, mild honeycombing. These features are similar to those of other forms of idiopathic pulmonary fibrosis. However, in IPPFE, they are seen mainly in upper lobes, being rare or absent in the lower lobes [7,11]. Chest X-ray, pulmonary function tests and HRCT findings of our case were consistent with above criteria.
Characteristic histopathological findings in PPFE include distortion and marked pleural thickening, with evidence of dense fibroelastic tissue in the pleura and alveolar walls, together with sparse fibroblastic foci near the transition from the lung to the pleura in elastic fiber staining [12]. These lesions are expected to cause mechanical restriction resulting both inspiratory and expiratory weakness.
Differential diagnosis of the imaging features includes late stage hypersensitivity pneumonitis, apical cap, connective tissue disease, advanced fibrosing sarcoidosis, drug induced lung disease, radiation, and prior tuberculosis [13,14]. In countries like Turkey where tuberculosis is common, sometimes tuberculosis may be first clinic condition for differential diagnosis of PPFE like our case. VATS pathological sample and BAL specimens were negative for tuberculosis culture and PCR in our case.
The natural history and intrinsic behavior of patients with PPFE is unpredictable as it varies from rapid progression to relatively long periods of stability. Survival of patients mostly depends on the stage of the disease at presentation and lung transplantation seems to be only effective treatment. Unfortunately we did not have enough time to send our patient to lung transplantation, since she presented and diagnosed at the later stages of disease. She passed away in intensive care unit due to severe hypoxic and hypercapnic respiratory failure and septic shock.
In conclusion, although there are specific HRCT features that are suggestive of IPPFE, the disease continues to be underdiagnosed, probably because there is still a significant lack of knowledge and awareness of this entity. A diagnosis of IPPFE should be considered in cases of pulmonary fibrosis that is located predominantly in the upper lobes. Further studies that include larger and more homogenous patient cohorts are needed in order to increase the awareness of this disease.
References
- Amitani R, Miimi A, Kuse F (1992) Idiopathic pulmonary upper lobe fibrosis (IPUF). Kokyu 11: 693-699.
- Frankel SK, Cool CD, Lynch DA, Brown KK (2004) Idiopathic pleuroparenchymal fibroelastosis: description of a novel clinicopathologic entity. Chest 126: 2007-2013.
- Travis WD, Costabel U, Hansell DM, King TE Jr, Lynch DA, et al. (2013) An official American Thoracic Society/European Respiratory Society statement: update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J RespirCrit Care Med 188: 733-748.
- Kusagaya H, Nakamura Y, Kono M, Kaida Y, Kuroishi S, et al. (2012) Idiopathic pleuroparenchymalfibroelastosis: consideration of a clinicopathological entity in a series of Japanese patients. BMC Pulm Med 12: 72.
- Hirota T, Fujita M, Matsumoto T, Higuchi T, Shiraishi T, et al. (2013)Pleuroparenchymalfibroelastosis as a manifestation of chronic lung rejection? Eur Respir J 41: 243-245.
- Ofek E, Sato M, Saito T, Wagnetz U, Roberts HC, et al. (2013) Restrictive allograft syndrome post lung transplantation is characterized by pleuroparenchymalfibroelastosis. Mod Pathol 26: 350-356.
- Piciucchi S, Tomassetti S, Casoni G,Sverzellati N, Carloni A, et al. (2011) High resolution CT and histological findings in idiopathic pleuroparenchymalfibroelastosis: features and differential diagnosis. Respir Res 12: 111.
- Watanabe K, Nagata N, Kitasato Y, Wakamatsu K, Nabeshima K, et al. (2012) Rapid decrease in forced vital capacity in patients with idiopathic pulmonary upper lobe fibrosis. Respir Investig 50: 88-97.
- Watanabe K (2013)Pleuroparenchymal fibroelastosis: its clinical characteristics. Curr Respir Med Re v9: 299-237.
- Harada T, Yoshida Y, Kitasato Y, Tsuruta N, Wakamatsu K, et al. (2014) The thoracic cage becomes flattened in the progression of pleuroparenchymalfibroelastosis. Eur Respir Rev 23: 263-266.
- Camus P, von der Thüsen J, Hansell DM, Colby TV (2014) Pleuroparenchymal fibroelastosis: one more walk on the wild side of drugs? Eur Respir J 44: 289-296.
- von der Thüsen JH (2013)Pleuroparenchymal fibroelastosis: its pathological characteristics. Curr Respir Med Rev 9: 238-247.
- Becker CD, Gil J, Padilla ML (2008) Idiopathic pleuroparenchymal fibroelastosis: an unrecognized or misdiagnosed entity? Mod Pathol 21: 784-787.
- Beynat-Mouterde C, Beltramo G, Lezmi G, Pernet D, Camus C, et al. (2014)Pleuroparenchymal fibroelastosis as a late complication of chemotherapy agents. Eur Respir J 44: 523-527.
Citation: Yildirim F (2016) Idiopathic Pleuroparenchymal Fibroelastosis as a Rare Entity within Idiopathic Interstitial Pneumonias: A Case Report. Diagn Pathol Open 1:115. DOI: 10.4172/2476-2024.1000115
Copyright: ©2016 Yildirim F. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Tools
Article Usage
- Total views: 14879
- [From(publication date): 6-2016 - Aug 19, 2025]
- Breakdown by view type
- HTML page views: 13865
- PDF downloads: 1014