Research Article Open Access
Mercury in Different Tissues of Grey Herons (Ardea cinerea) from the Volta Lake, Ghana
Kwaansa-Ansah EE*, Agyemang D and Opoku F
Department of Chemistry, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
- *Corresponding Author:
- Kwaansa-Ansah EE
Department of Chemistry
Kwame Nkrumah University of Science and Technology
Kumasi, Ghana
Tel: +233244298182
Fax: +233322060305
E-mail: eekwaansaansah@yahoo.com
Received date: October 29, 2015; Accepted date: December 02, 2015; Published date: December 07, 2015
Citation: Kwaansa-Ansah EE, Agyemang D, Opoku F (2015) Mercury in Different Tissues of Grey Herons (Ardea cinerea) from the Volta Lake, Ghana. J Marine Sci Res Dev 6:175. doi:10.4172/2155-9910.1000175
Copyright: © 2015 Kwaansa-Ansah EE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Marine Science: Research & Development
Abstract
Birds as high consumers and account for accumulative processes in marine pollution accumulate contaminants such as mercury (Hg). Therefore, in this study, the concentration of total mercury (THg) in ten (10) different tissues of Ardea cinerea from the Volta Lake, Ghana was measured. The THg concentrations in Ardea cinerea tissues follow the order: lungs
Keywords
Total mercury; Ardea cinerea; Tissues; Feather; Volta lake
Introduction
Mercury as a highly toxic non-essential metal strongly influences humans and wildlife with direct effects on behaviour, neurology, endocrinology and development [1,2]. The source of Hg is from both natural and anthropogenic sources as well as human activities [3]. Hg contaminates oceanic islands and polar and sub-polar regions due to its high volatility and long atmospheric residence time [4,5]. Exposure to mercury results in impaired vision and hearing, dizziness, vomiting, headache, muscular weakness, allergies, depressed immune system and brain damage [6].
The Volta Lake is a unique and an extraordinary natural resource that provide drinking water, food, recreation, employment and transportation to millions of Ghanaians as well as tourism. Volta Lake is the World’s second largest and Africa’s largest artificial inland water body which occupy 3.6% of the total land area of Ghana. The Volta Lake supply about 90% of the nation’s inland fish supply.
It is known that certain forms of mercury can readily accumulate within organisms tissues at much higher levels than those in the water column and in sediment [7]. Mercury as a non-biodegradable contaminant can enter aquatic food chains and consequently accumulates organisms at the various trophic levels [8]. Biological and ecological factors have significant influences on metals bioaccumulation and bioavailability [9].
Birds are valuable tool for environmental monitoring because they are always at the top of the food chain and often have high levels of Hg contamination [10-14]. Feathers are among the most attractive tissue to monitor, because they are considered as the main route for Hg elimination [14]. Mercury accumulation in piscivorous birds has extensively being studied in the past [15-18], but to the best of our knowledge data on mercury contamination in different tissues of grey herons from the Volta Lake, Ghana is lacking. This work is aiming to evaluate the residual accumulation of mercury in different tissues of grey heron’s in Ghana.
Materials and Methods
Study area
The Volta Lake was formed in 1965 as a result of the construction of the Akosombo dam which generates hydroelectric power for Ghana. It has a surface area of 8500 km2 and covers about 4% of Ghana’s total land area. It covers 1,232 communities with a population of over 300,000 people living along the shoreline whose livelihood depend on the Volta Lake [19].
Sampling
Our overall protocol, under appropriate state and federal permits was to collect grey herons from shorebirds netted during their migratory stopover on Volta Lake in Ghana. 10 grey herons were collected as by catch of long line fishing vessels or found recently dead on land killed following collision with the Scientific considered for this study. The grey herons were weighed in the field, immediately frozen and kept at -20°C until analyses. Birds were thawed overnight and dissected for kidney, intestines, lungs, heart, gizzard, blood, brain, liver, flesh and feathers that give the best measure of the Hg contamination of a bird. The birds were transported to the Department Of Wildlife, Faculty of Renewable Natural Resources, Kwame Nkrumah University of Science and Technology, Kumasi for measurement of their biometrical features. After measurement of biometrical features, the birds were kept frozen at -4°C until analysis.
Analysis
During the analysis, the birds were dissected and the various tissues separated into kidney, intestines, lungs, heart, gizzard, blood, brain, liver, flesh and feathers. Each tissue was washed with deionized water minced into smaller pieces and homogenized. The samples were digested for total mercury determination by an open flask procedure developed by Akagi and Nishimura [20]. In the procedure, approximately 0.5 g of each tissue was weighed into a 50 mL digestion tube and 1mL deionized water, 2 mL of a mixture of HNO3 and HClO4 (1:1) and 5 mL H2SO4 was added. The samples were digested at 200°C ± 5 for 30 minutes, cooled to room temperature and topped to the 50mL mark with deionized water. A blank and standard solution was digested using 25, 50 and 100 μL of l μg/mL standard Hg solution. Total mercury concentrations were determined using an automatic Mercury Analyser (Model HG-5000). The reducing reagent used for the analysis was 0.5 mL of 10% (w/v) SnCl2.2H2O in 1 MHCl. Quality assurance samples analysed included procedural blanks, replicate samples and post-digestion spikes. The accuracy of the procedure was determined by analysis of standard reference materials (SRMs). The SRMs included National Research Council of Canada (NRCC) DOLT-4 (dogfish liver) and TORT-2 (lobster hepatopancreas). All CRMs were recovered at ± 10% of the certified value; 0.27 ± 0.06 for Tort-2 and 2.58 ± 0.22 for Dolt-4. Statistical analysis was conducted using IBM Statistical Package for the Social Sciences ‘20. A significance level of α<0.05 was used for all tests.
Results and Discussion
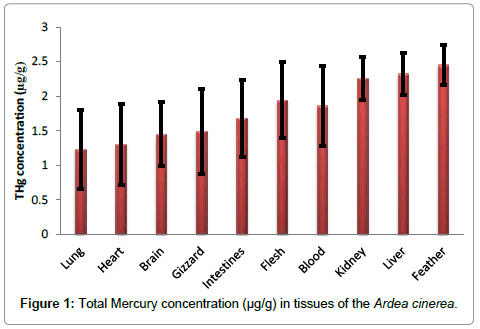
The mean concentration of mercury in the lungs, heart, brain, gizzard, intestines, fresh, blood, kidney, liver and feathers tissues were 1.23 ± 0.57, 1.30 ± 0.58, 1.45 ± 0.46, 1.49 ± 0.62, 1.68 ± 0.56, 1.94 ± 0.55, 1.86 ± 0.58, 2.26 ± 0.31, 2.32 ± 0.30 and 2.45 ± 0.29, respectively (Table 1).The lowest Hg concentration occurred in lung tissue and the highest in feathers tissues (1.23 ± 0.57 and 2.45 ± 0.29 μgg-1, respectively).The concentration of THg in the tissues studied was found to increase in the following order: lungs<heart<brain<gizzard<intestines<flesh<blood<kidney<liver<feathers.
| Tissue | Mean THg concentration | Range of THg Concentration |
|---|---|---|
| Lung | 1.23 ± 0.57 | 0.77 - 2.70 |
| Heart | 1.30 ± 0.58 | 0.79 - 2.76 |
| Brain | 1.45 ± 0.46 | 1.18 - 2.57 |
| Gizzard | 1.49 ± 0.62 | 0.96 - 2.79 |
| Intestines | 1.68 ± 0.56 | 1.15 - 2.79 |
| Flesh | 1.94 ± 0.55 | 1.91 - 2.80 |
| Blood | 1.86 ± 0.58 | 1.20 - 2.85 |
| Kidney | 2.26 ± 0.31 | 1.97 - 2.85 |
| Liver | 2.32 ± 0.30 | 2.02 - 2.86 |
| Feather | 2.45 ± 0.29 | 2.05 - 2.84 |
Table 1: Mean THg concentration (μgg-1) in tissues of Ardea cinerea.
During current study, mercury concentrations in tested tissues of all the fishes increased in order lungs < heart <brain<gizzard<i ntestines<flesh<blood<kidney<liver<feathers (Figure 1). Mercury concentrations are usually found at the highest levels in feathers followed by liver, kidney, and muscle tissues [17,21]. In birds, feathers play a major role in mercury excretion [22]. As a result of high affinity for the sulfhydryl groups in keratin, mercury can accumulates in bird feather [21,23]. Previous reports indicate that Hg accumulates more in liver than kidney follow by muscle [11,17,18]. Lewis and Furness [24] also reported lower mercury levels in kidney than liver in black-headed gull (Larus ridibundus).
Frodello et al. [25] and Storelliet al. [26] reported that the liver appeared to be the preferential organ for mercury accumulation, followed by the kidney. The THg concentration in the liver is accounted for the biotransformation of demethylates organic mercury into less toxic [27]. The levels of mercury found in the kidneys are explained on the basis that the kidney is the major reservoir of inorganic mercury in birds as well as mammals [28]. The kidney contains a thiol-rich protein called metallothionein and exposure to toxic metals triggers the production of this protein and binds tightly to the metal [29]. Spalding et al. [30], found that kidney disease and gout were present in great white herons are greater than 25 μg/g. Though elevated mercury levels were found in the kidney tissues of the birds in this study, the highest concentration of THg (2.85 μg/g) found in the kidney tissues of Ardea cinerea was about nine times less than the concentration reported to cause diseases and adverse effects in birds.
Higher concentration of mercury found in the feathers may be due to the fact that mercury is excreted mainly via the feathers [31]. Feathers which represent the major pathway for elimination of mercury in birds are useful indicators for assessment of whole bird mercury burdens [15,32,33]. Studies on the Great skua (Catharcta skua) shows that mercury was more contaminated in the feathers [34]. Mercury in the food eaten during feather growth considerably affects the mercury levels of the feathers [35].
Boening [36] and Ochoa-Acuna et al. [37] found some differences in Hg concentrations in feathers across taxonomic bird groups. Mercury concentrations in birds also depend on body size, moult strategy, migration patterns and physiology [38]. Fish-eating birds are at risk of higher contents of Hg because its circulation is associated mainly with water basins [37,39].
Pearson correlation coefficient between the age and THg concentration in all the tissues studied showed a very strong linear relationship. Total mercury concentrations in the tissues of Ardea cinerea were positively correlated with age (p<0.05). This relationship suggests that as grey herons mature, mercury is accumulated in all the tissues studied as such they can be useful bio-indicators for monitoring Hg pollution in the Volta Lake and other aquatic ecosystems.
Conclusion
The results showed that the accumulation of total mercury varied among the various tissues. However, Ardea cinerea species showed relatively high THg concentration in feather tissues. From a public health perspective, levels of mercury in the tissues of birds are very alarming. Therefore, studies on mercury contamination in birds especially around the coastal areas are relevant and useful for environmental monitoring.
Competing Interests
The authors declare that they have no competing interests.
Acknowledgement
The authors are very grateful to the National Council for tertiary Education (NTCE), Ghana for a research grant under the Teaching and Learning Innovation Fund (TALIF-KNUSTR/3/005/2005). We are also grateful to the Department Of Wildlife, Faculty of Renewable Natural Resources, Kwame Nkrumah University of Science and Technology (KNUST), Kumasi, Ghana for the use of their facilities for this work.
References
- ScheuhammerAM, Meyer MW, Sandheinrich MB, Murray MW (2007) Effects of environmental methylmercury on the health of wild birds, mammals, and fish. Ambio 36: 12-18.
- Grandjean P, Satoh H, Murata K, Eto K (2010) Adverse effects of methylmercury: environmental health research implications. Environ Health Persp 118: 1137-1145.
- Selin NE (2009) Global biogeochemical cycling of mercury: a review. Annu Rev Environ Resourc 34: 43-63.
- Fitzgerald WF, Engstrom DR, Mason RP, Nater EA (1998) The case for atmospheric mercury contamination in remote areas. Environ SciTechnol 32: 1-7.
- Ebinghaus R, Kock HH, Temme C, Einax JW, Löwe AG, et al. (2002) Antarctic springtime depletion of atmospheric mercury. Environ SciTechnol 36: 1238-1244.
- Risher JF (2003) Elemental mercury and inorganic mercury compounds: Human health aspects. World Health Organization, Geneva.
- Beltrame MO, Marco SGD (2010) Influences of sex, habitat, and seasonality on heavy-metal concentrations in the burrowing crab (Neohelice granulate) from a coastal lagoon in Argentina. Arch Environ ContamToxicol 58: 746-756.
- Hosseini M, Nabavi SMB, Parsa Y (2013) Bioaccumulation of Trace Mercury in Trophic Levels of Benthic, Benthopelagic, Pelagic Fish Species, and Sea Birds from Arvand River, Iran. Biological Trace Element Research 156: 175-180.
- Bustamante P, Bocher P, Cherel Y, Miramand P, Caurant F (2003) Distribution of trace elements in the tissues of benthic and pelagic fish from the Kerguelen Islands. Sci Total Environ 313: 25-39.
- Furness RW, Greenwood JD (1993) Birds as Monitors of Environmental Changes British Trust for Ornithology. 1stEdn UK: Thetford Norfolk 342.
- Horai S, Watanabe I, Takada H, Iwamizu Y, Hayashi T, et al. (2007) Trace element accumulations in 13 avian species collected from the Kanto area, Japan. Sci Total Environ 373: 512-525.
- Burger J, Gochfeld M (2004) Marine birds as sentinels of environmental pollution. Eco Health 1: 263-274.
- Burger J, Gochfeld M (1991) Cadmium and lead in common terns (Aves, Sterna hirundo): Relationship between levels in parents and eggs. Environ Monit Assess16:253-258.
- Monteiro LR, Furness RW (1995) Seabirds as monitors of mercury in the marine environment. Water Air Soil Pollut 80: 851-870.
- Thompson DR, Furness RW (1989) The chemical form of mercury stored in South Atlantic seabirds. Environ Pollut 60: 305-317.
- Monteiro LR, Granadeiro JP, Furness RW, Oliveira P (1999) Contemporary patterns of mercury contamination in the Portuguese Atlantic inferred from mercury concentrations in seabird tissues. Mar Environ Res 47: 137-156.
- Kim EY, Murakami T, Saeki K, Tatsukawa R (1996) Mercury levels and its chemical form in tissues and organs of seabirds. Arch Environ ContamToxicol 30: 259-266.
- Houserova P, Kuban V, Kracmar S, Sitko J (2007) Total mercury and mercury species in birds and fish in an aquatic ecosystem in the Czech Republic. Environ Pollut 145: 185-194.
- Braimah LI (2001) Volta lake fisheries management plan: Fisheries sub-sector capacity building project. 18-32.
- Akagi H, Nishimura H (1991) In: Suzuki T, Imura N, Clarkson TW (eds) Speciation of mercury in the environment. Advances in mercury toxicology New York: Plenum Press 53-76.
- Zamani-Ahmadmahmoodi R, Esmaili-Sari A, Ghasempoury SM, Savabieasfahani M (2008) Mercury levels in selected tissues of three kingfisher species; Cerylerudis, Alcedoatthis, and Halcyon smyrnensi, from Shadegan Marshes of Iran. Ecotoxicology 18: 319-324.
- Al-Hello AA, Al-Obaidy AM (1997) The chemistry of Shatt Al-Arab water from Qurna to Al-Fao. Mar Mesopotamica 12: 190-201.
- Saeki K, Okabe Y, Kim EY, Tanabe S, Fukuda M, et al. (2000) Mercury and cadmium in common cormorants (Phalacrocoraxcarbo). Environ Pollut 108: 249-255.
- Lewis SA, Furness RW (1991) Mercury accumulation and excretion in laboratory reared black-headed gull Larusridibundus chicks. Arch Environ ContamToxicol 21: 316-320.
- Frodello JP, Romeo M,Viale D (2000) Distribution of mercury in the organs and tissues of five toothed-whale species of the Mediterranean. Environ Pollut 108: 447-452.
- StorelliA, Marcotrigiano GO (2005) Accumulation of mercury, cadmium, lead and arsenic in Swordfish and Bluefin tuna from the Mediterranean Sea: A comparative study. Mar Pollut Bull 50: 1004-1007.
- Kinabo C, Lyatuu H (2009) Mercury contamination in domestic ducks in Geita Northwest Tanzania. Tanz J Sci 35: 38-45.
- United States Department of the Interior (1998) Guidelines for Interpretation of the Biological Effects of Selected Constituents in Biota, Water, and Sediment: Mercury. Report No. 3. National Irrigation Water Quality Program Information, United States Department of the Interior.
- Ware RA, Burkholder PM, Chang LW (1975) Ultra structural changes in renal proximal tubule after chronic organic and inorganic mercury intoxication. Environ Res 121-140.
- Spalding MG, Bjork RDG, Powell VN, Sundlof SF (1994) Mercury and cause of death in great white herons (Ardeaherodiasoccidentalis). J Wildlife Manage 58: 735-739.
- Parslow JLF, Jeffries DJ, Hason HM (1973) Gannet mortality incidents in 1972. Marine Poll Bull 4: 41-43.
- Liu QG, Chen Y, Li JL, Chen, LQ (2007) The food web structure and ecosystem properties of a filter-feeding carps dominated deep reservoir ecosystem. Ecol Model203: 279-289.
- Furness RW, Muirhead SJ, Woodbum M (1986) Using bird feathers to measure mercury in the environment: relationships between mercury content and moult. Marine Pollution Bulletin 17: 27-30.
- BearhopS, Waldron S, Thompson D, Furness R (2000) Bio-amplification of mercury in great skuaCatharactaskuachicks: The influence of trophic status as determined by stable isotope signatures of blood and feathers. Mar Pollut Bull40:181-185.
- Mautinom M, Bell JU (1986) Experimental Lead Toxicity in the Ring-necked Duck. Environmental Research 41: 538-545.
- Boening DW (2000)Ecological effects, transport, and fate of mercury: A general review. Chemosphere40:1335-1351.
- Ochoa-Acuna H, Sepulveda MS, Gross TS (2002) Mercury in feathers from Chilean birds: influence of location, feeding strategy, and taxonomic affiliation. Mar Pollut Bull 44: 340-349.
- Dauwe T, Bervoets L, Pinxten R, Blust R, Eens M (2003) Variation of heavy metals within and among feathers of birds of prey: effects of molt and external contamination. Environ Pollut 124: 429-436.
- Stewart FM, Phillips RA, Catry P, Fumess RW (1997) Influence of species, age and diet on mercury concentrations in Shetland seabirds. Marine Ecology Progress Series 151: 237-244.
Relevant Topics
- Algal Blooms
- Blue Carbon Sequestration
- Brackish Water
- Catfish
- Coral Bleaching
- Coral Reefs
- Deep Sea Fish
- Deep Sea Mining
- Ichthyoplankton
- Mangrove Ecosystem
- Marine Engineering
- Marine Fisheries
- Marine Mammal Research
- Marine Microbiome Analysis
- Marine Pollution
- Marine Reptiles
- Marine Science
- Ocean Currents
- Photoendosymbiosis
- Reef Biology
- Sea Food
- Sea Grass
- Sea Transportation
- Seaweed
Recommended Journals
Article Tools
Article Usage
- Total views: 10837
- [From(publication date):
February-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 9928
- PDF downloads : 909