Research Article Open Access
Patient Radiation Risk in Interventional Cardiology
| Eleni-Theano Samara1*, Abbas Aroua1, François Oswald Bochud1, Alain Delabays2, Jean-Pascal Laedermann1 and Francis Robert Verdun1 | |
| 1Institute of Radiation Physics, Lausanne University Hospital, Rue du Grand-Pre 1, CH-1007, Lausanne, Switzerland | |
| 2Department of Cardiology, Lausanne University Hospital, Rue du Bugnon 46, CH-1011 Lausanne, Switzerland | |
| Corresponding Author : | Eleni-Theano Samara Institute of Radiation Physics Lausanne University Hospital Rue du Grand-Pre 1, CH-1007 Lausanne, Swit- zerland E-mail: eleni-theano.samara@chuv.ch |
| Received April 12, 2012; Accepted April 23, 2012; Published April 30, 2012 | |
| Citation:Samara ET, Aroua A, Bochud FO, Delabays A, Laedermann JP, et al. (2012) Patient Radiation Risk in Interventional Cardiology. OMICS J Radiology. 1:103. doi: 10.4172/2167-7964.1000103 | |
| Copyright: © 2012 Samara ET, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Radiology
Abstract
Radiological imaging used in cardiac procedures may result in significant radiation doses to the patient. The aim of this study was to estimate radiation risks from typical cardiac procedures. Monte Carlo simulations were performed to assess absorbed organ doses. Equivalent organ doses and effective doses were calculated using the recommendations in ICRP Publications 60 and 103 [1,2]. The differences in effective dose applying the two recommendations were found to be small. Cancer incidence risks for different organs were calculated for different sex and age at exposure using the lifetime attributable risks provided by the Biological Effects of Ionizing Radiations report VII. These results are a straightforward method to comprehend radiation risks.
| Introduction |
| A large increase in both diagnostic and therapeutic interventional procedures has been observed in recent years [3,4]; the most common of them being coronary angiography (CA) and percutaneous coronary intervention (PCI) [4,5]. Cardiac conditions previously treated by open heart surgery may now be safely and effectively performed by x-ray-guided interventions [6]. However, advances in technology have allowed complex cases to be treated leading to longer fluoroscopy times and thus to higher levels of radiation exposure [6,7]. Exposure to x-ray radiation has been demonstrated to increase the risk of cancer [2,8]. Thus, the use of radiation in medicine is only accepted when the benefits are greater than the associated risks [2]. Therefore, every procedure that involves exposure to ionizing radiation has to be justified. Before prescribing or performing a radiological procedure for any individual patient, the physician has to weigh the benefits with the risks taking into account the suspected pathology, the characteristics of the patient, alternative medical procedures, etc. The risk-benefit analysis is particularly important for high-dose procedures, such as interventional radiology. Risk assessment is a critical part of the justification process and pertinent quantities have to be used to perform such an evaluation. In addition, radiological procedures need to be optimized on the basis of reliable indicators. |
| The effective dose (E) as defined by International Commission on Radiological Protection (ICRP) is based on the use of equivalent doses, i.e. mean tissue and organ absorbed doses each weighted by radiation weighting factors which take account of the differences in radio-biological effectiveness of various radiations. Effective dose is the sum of all organ and tissue equivalent doses each weighted by specific tissue weighting factors which are intended to take account of the differences in sensitivity of tissue and organs with respect to stochastic effects. The aim of the E was to define a radiation protection quantity is which is related to the probability of a detriment from low- dose exposure to ionizing radiation where only stochastic effects occur and which is suited for setting exposure limits and for implementing the optimization principle in the context of occupational and public exposure. The concept of E was adopted by the ICRP 26 (1977) [9] and further developed in its recommendations ICRP 60 (1991) and 103 (2007) [1,2]. E was initially proposed for the radiation protection of the occupational workers and the general public. However, the convenience and ease of evaluating E in given exposure situations as well as the lack of established methods to estimate patient risks has led to its widespread use in the estimation of patient risks [10]. Yet, publications on awareness of the physicians on radiation protection show that radiation risks are not well understood and assessed [11-15]. Moreover, it seems that the language of radiation protection is confusing for non-specialists, for instance the use of Sv as unit of both the equivalent organ dose and the effective dose [16,17]. Therefore, there is a need for a quantity that is more dedicated to the assessment of radiation risks, and comprehensive by non-specialists. Equivalent organ dose was intended to quantify the organ risk to assess a certain detriment by multiplying the dose to the organ by the respective risk coefficient [18]. Brenner [16] proposed the use of cancer incidence risks for different organs and tissues provided in the Biological Effects of Ionizing Radiations (BEIR) report VII (NRC 2006) [8] and the estimation of the effective risk (R) [9]. |
| The objective of our study was to examine the possible use of R as an estimate of radiation risk during common cardiac procedures. To this purpose, two typical cardiac interventional procedures were simulated and the corresponding organ doses and potential risks for cancer induction were estimated. |
| Materials and Methods |
| Patient study |
| Patient data were derived from the database of a cardiology department. For our study, data for all the patients referred to CA and PCI procedures during one year were exported. Patient data included patient demographics (sex, weight, height, body mass index (BMI) and age), volume of the contrast agent as well as dose parameters, such as kerma-area product (PKA), fluoroscopy time (T) and cumulative dose (CD) measured at the interventional reference point. The number of images obtained during a procedure was not automatically registered and thus, not included in the database. Patients were divided into different categories according to their BMI (the weight (kg) of the patient divided by the square of the height (m2)). No patients with a BMI less than 18.5 undergoing a CA or PCI procedure were found. In this study, only data of normal patients were taken into account in order to describe a typical procedure for an average patient. Independent samples Student t-tests were performed to test any differences between a) male and female patients and b) CA and PCI procedures. The statistical analysis was performed with the SPSS software (version 18, SPSS Inc. Chicago, IL). |
| During a four-month period, a detailed study was performed for the patients referred to CA and PCI procedures. The sample included 32 male and 15 female patients. For these patients technical and physical parameters were recorded, such as source-to-detector distance, number of images acquired during the procedure, tube angulation, and tube voltage (kV) for each series of images. All procedures were performed with a monoplane X-ray unit (Allura Xper FD10, Philips Healthcare, Philips, Eindhoven, Netherlands) equipped with a dynamic flat-panel detector. Pulsed fluoroscopy and cineangiography were used during the procedures. |
| Monte Carlo simulations |
| To estimate organ doses during coronary procedures, Monte Carlo simulations were performed. Monte Carlo simulations are widely used to estimate the radiation dose distribution in the body [19-22]. The general-purpose radiation transport code MCNPX was employed (version 2.5.0, Los Alamos National Laboratory, Los Alamos, NM). This code can handle the transport and interactions of photons, electrons, neutrons and other particles for a wide range of energies and is three-dimensional and time-dependent. |
| The simulation of the human body was performed using the ICRP 110 voxelized adult reference phantoms [23]. The two phantoms are the official computational models representing the Reference Male and Female. The models are based on computed tomography images of real individuals and represent the human anatomy in three dimensions. The voxel volume for the Reference Male was 36.54 mm3 and that for the Reference Female 15.25 mm3. The phantoms represent an adult male of 73 kg weight and 1.76 m height and an adult female of 60 kg and 1.63 m. All the organs and tissues required for the calculation of the effective dose according to ICRP 103 [2] are included in the phantoms. |
| Three x-ray spectra (70,80 and 90 kVp) and 3 mm Al total filtration were produced according to the method developed by Boone and Seibert [24]. The distance of the x-ray tube to the detector was assumed to be constant at 100 cm. Five main projections (posterior anterior (PA), right anterior oblique 30° (RAO30°), left lateral (LLAT), left anterior oblique 10° (LAO10°), and left anterior oblique 60o (LAO60°) and three additional ones (right anterior oblique 30° and caudal 20° (RAO30°CAD20°), left anterior oblique 10° and cranial 10° (LAO10°/CRA10°), and left anterior oblique 60° and cranial 10° (LAO60°/CRA10°) were studied. For all tube angulations, the field of view was chosen to be 10x10 cm2 centred at the heart of the patient. In all simulations, 200 million histories were performed to ensure that the statistical error was acceptable. Typical CPU time for the simulations varied between 11 and 20 hours on a nine-PC cluster (HP 3646/3GHz/dual core/4Gb RAM, Linux 2.6.32-35-generic, Ubuntu 10.04), depending on the source geometry and energy spectrum. |
| Verification measurements |
| To verify the Monte Carlo results, a physical anthropomorphic phantom (Model 702-D, ATOM, CIRS, Norfolk, VA) that simulates the body trunk of an adult female 1.60 m in height and 55 kg in weight was used. The phantom is sectional in design with thirty-eight 25-mmthick slabs of tissue equivalent material. The tissue-equivalent epoxy resins simulate soft tissue, skeleton and lungs and allow the phantom’s use for radiation energies in the field of diagnostic radiology. The phantom provides thermo-luminescent dosimeters (TLDs) locations specific to 21 inner organs. Eighty TLDs were loaded in the phantom in different organs to measure the absorbed dose. The same geometrical setup with that of Monte Carlo simulations was followed to verify the Monte Carlo results. The phantom was exposed to 50 Gyâ�?�?cm2 to reduce the statistical errors of the TLD measurements. Normalized organ dose values were calculated as the mean dose gauged by TLDs divided by the total KAP for each exposure. Data derived by using TLD dosimetry were compared with corresponding data derived from Monte Carlo simulations. |
| The TLD crystals (LiF:Mg,Tl chips, TLD-100; Harshaw, Solon, Ohio) were calibrated towards an ionization chamber NE 2575 600 cm3 (Nuclear Enterprise, U.K.) connected to an electrometer Keithley 6517A (Keithley Instruments Inc, Cleveland, Ohio) and a Co-60 source (mean energy 1.25 MeV). The individual sensitivity of each TLD was determined taking into account the energy correction factor calculated by mass energy absorption coefficients. Thus, the overall uncertainty of TLD measurements was lower than 5%. The TLD reading was performed by a Harshaw 5500 reader system (Harshaw, Solon, Ohio). The annealing process for the TLDs was carried out at a determined oven Hengstler grado 921 (Hengstler GmbH, Aldingen, Germany) for an annealing cycle at 385°C for 75 minutes followed by another annealing cycle at 100°C for 135 min in the oven Venticell 222 (MMM Medcenter Einrichtungen GmbH, Munich, Germany). The first annealing cycle is performed for the re-crystallization of the TLDs while the second one is performed to erase the first two peaks of the thermo-luminescent signal, which reduce the dosimeter “fading”. |
| Estimation of effective dose |
| The energy deposition to the organs and tissues was estimated using the *f8-type tally (in MeV). This is a pulse height tally. It is defined by the energy entering a cell (in our case each cell was an organ or tissue) minus the energy leaving this cell for the entire history for each particle emitted by the source. The amount of energy deposited in a cell is the pulse height for that history. A full pulse height spectrum is created when the distribution of the energy deposited by all incident source particles is recorded. The pulse height tally requires the entire set of tracks for a history to be completed. The absorbed dose in the organ or tissue (DT) was calculated by the energy deposited in the organ or tissue over the corresponding mass (MeV/kg). KAP was also calculated by multiplying the free-in-air-kerma value with the area of the x-ray field. The KAP-normalized organ dose (DTKAP) was calculated by the value of the organ dose divided by the KAP, as summarized in the equation: |
| |
| The KAP-normalized equivalent dose (HTKAP) in an organ or tissue was calculated by: |
| |
| Where the wR is the radiation weighting factor for radiation R, in our case, the radiation weighting factor for the photons was 1. |
| E is based on the mean equivalent doses in organs or tissues of the human body. The tissue weighting factors represent average values for individuals of both sexes. Thus, the equivalent doses in the organs and tissues of the Reference Male and the Reference Female were averaged. The averaged equivalent doses were then multiplied with the corresponding tissue weighting factor. The KAP-normalized effective dose (EKAP) was calculated as following: |
| |
| Where the summation includes the gonads dose (testes in the male and ovaries in the female) |
| The tissue weighting factors wT are given in table 1. E was calculated using both ICRP 60 and 103 [1,2]. The new recommendations define the colon and the remainder tissues differently from the old ones and this was also taken into account in the E calculations. |
| Estimation of effective risk |
| The term “effective risk” R was proposed by [16] in order to estimate the cancer risk from a radiological procedure. R may be calculated as following: |
| |
| where rT is the lifetime radiation-attributable tissue- specific cancer risks, as defined by BEIR VII (NRC 2006) [8] and DT is the equivalent dose in tissue T. The unit of R is the cancer incidence. Table 2 shows the risk for cancer incidence according to BEIR VII. The risks are age- and sex-dependent. The dose to “other” (Dother) was calculated as following: |
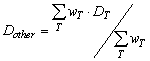 |
| Where DT is the mean organ dose for a typical cardiac procedure, wT is the tissue weighting factors for the corresponding organs according to ICRP 103 [2]. T concerns the oesophagus, brain, bone surface, skin, salivary glands, the gonads (for male calculations), and the remainder tissue mentioned in ICRP 103 [2] except for the prostate and the uterus. |
| Results and Discussion |
| Patient characteristics |
| In total, 269 patients were included in the study. In table 3, patient demographics, i.e. age, weight, height and BMI, are presented for CA and PCI procedures. Of the 137 patients who underwent a CA procedure, 76 were males and 61 were females with a mean age of 67 years and mean BMI of 22.7 kg/m2. The 136 patients that underwent PCI procedure were 91 males and 41 females with mean age of 67 years and mean BMI 23.1 kg/m2. |
| The parameters related to the patient dose, i.e. KAP, fluoroscopy time, etc are presented in Table 4 and 5 for the CA and PCI procedures, respectively. The mean values for exposure (KAP, T, number of images, CD) during CA procedures were found significantly lower than those for PCI procedures (p-values < 0.001). The number of images acquired during the procedures was determined by the sample patient study. This study showed also that the mean tube voltage applied was 77 and 83 kV for CA and PCI procedures, respectively. |
| Equivalent organ dose |
| KAP-normalized doses for the all organs and tissues recommended by the ICRP were estimated. Tables 6 and 7 present the KAP-normalized doses for the main organs and the remainder organs and tissues as defined by both ICRP 60 and 103 [1,2]. The heart is included in the remainder organs in ICRP 103; however, we decided to also provide separately the dose to the heart as it is the target organ during the procedures studied. |
| The KAP-normalized doses for the colon were calculated with both ICRP 60 and 103 [1,2] and were found to differ in average by 36 times. |
| This was expected as the colon according to ICRP 60 was defined as the lower large intestine, but according to ICRP 103 the colon includes also the upper large intestine. The difference in the doses of the remainder tissue was less noticeable. In ICRP 60, the remainder tissue included 10 organs and tissues, while in ICRP 103 by 14 organs (13 for each sex). |
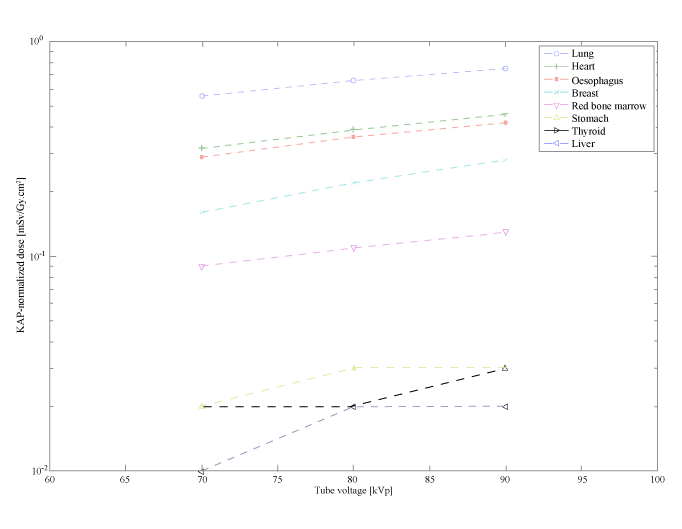
| The dependence of the organ dose on the tube voltage is presented in Figure 1 for the LAO10° projection. As expected, the higher the tube voltage, the higher the dose. Organs that are inside the x-ray field have higher KAP-normalized doses, as for example the lungs, the oesophagus and the heart in Figure 1. In contrast, organs that lie outside the x-ray field, such as the thyroid, receive lower doses. In average the KAP-normalized doses differ by 1.8 times for tube voltages between 70 and 90 kVp. The higher differences (up to 6 times) were observed for low KAP-normalized doses, such as the case of gonads for RAO projection. Organs with low KAP-normalized doses are exposed to scatter radiation; thus, their exposure highly depends on the energy of the scatter radiation. |
| The KAP-normalized doses for three tube angulations RAO30°, LAO10° and LAO60° were compared to the RAO30°CAD20°, LAO10°/CRA10°, and LAO60°/CRA10°. A tilt of 10 to 20 degrees in the sagittal plane led to a mean difference of 15% in organ doses. As expected, the difference in organ doses increases with the angle increase, for instance 30% difference in the heart dose for RAO30° and RAO30°CAD20°. |
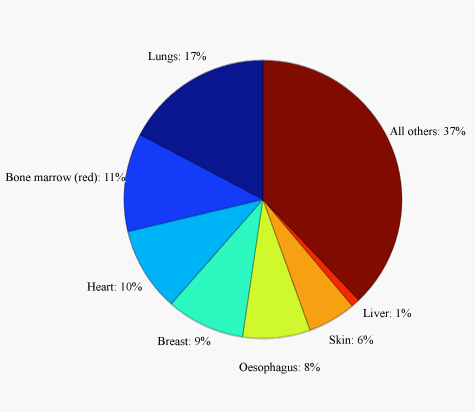
| A typical cardiac procedure may be described by the five main projections (PA, RAO30°, LAO10°, LAO60° and LLAT) for 80 kVp. The KAP-normalized doses for the organs and tissues for these angulations were averaged and multiplied by the mean KAP values for the CA and PCI procedures. The mean organ doses are presented in Table 6. During a typical cardiac intervention, the most irradiated organs were found to be the lungs, heart, red bone marrow, oesophagus and breast, as illustrated in Figure 2. Stomach and liver may also receive high doses. A special remark has to be made for the skin dose. The skin dose provided in tables 6-8 concerns the total skin and may be used only for the estimation of the effective dose. In real practice, only a small portion of the skin is directly irradiated at the body entrance. Moreover, skin areas may be irradiated by overlapping x-ray fields and may lead even to skin burns [25]. A quite good indicator for the skin dose is the CD at the interventional reference point, which is provided in modern x-ray units [26]. |
| KAP-normalized organ doses were also provided in previous [27- 29]. An extensive study in cardiac interventional procedures was performed in 1995 by Stern et al. [29]. Schlattl et al. [28] simulated the voxel phantoms Regina and Rex which were then adopted by the ICRP as the Reference Male and Female phantoms. However, differences in the phantom used by Stern et al [29] as well as the tube filtration and voltage, used by [28], do not allow a meaningful comparison between the results. A comparison was possible with the results of Bozkurt and Bor [27] for the male phantom KAP-normalized organ dose. In this publication, the voxel VIP-Man phantom was used and simulations were performed for tube voltage from 60 to 120 kVp and a 3.5 mm Al filtration. Our results are in a quite good agreement with the results of Bozkurt and Bor [27]. For example, the lung KAP-normalized dose for RAO30° projection at 70 kVp in this study was equal to 0.561 mSv/Gyâ�?�?cm2 and the corresponding value in the Bozkurt and Bor’s [27] work was 0.458 mSv/Gyâ�?�?cm2. Differences may be attributed to different phantoms and spectrum quality. |
| Coefficient of variance for large volume organs and organs within the x-ray field, such as liver, lung, skin, heart, etc., was below 0.01%. Although 200 million histories were carried out for each simulation to reduce the statistical errors, coefficients of variance of certain organs remained above 50%, for example left ovary 53% for 80 kVp. |
| Effective dose |
| The KAP-normalized effective doses according to ICRP 60 (1991) and ICRP 103 (2007) [1,2] for cardiac interventional procedures were calculated and are presented in Table 9. The effective doses were found to be higher when using the ICRP 103 than when using ICRP 60. The last column in Table 9 shows the ratio of the effective doses between ICRP 103 and ICRP 60. The differences were up to 20% and were most significant for the tube angulation LAO60°. In the case of cardiac procedures, this is attributed to the breast weighting factor, which was 0.05 in the ICRP 60 and increased to 0.12 in the new recommendations. |
| Our KAP-normalized effective doses according to ICRP 60 were found quite lower than published ones (range from 0.09 to 0.24 mSv/ Gy��?cm2), but are in general in good agreement [27]. Any differences may be attributed to the different phantoms and energy spectra used for the simulations. The KAP-normalized effective doses estimated in this work according to ICRP 103 could not be compared, as to the best of our knowledge, no previous publication estimate effective dose for cardiac procedures according to ICRP 103. |
| Effective doses for a typical (as previously described) CA and PCI procedure are presented in Table 10. Dose calculations were performed for both ICRP 60 and 103. The KAP-normalized effective doses for 80 kV were averaged to respect the change in the tube angulation during an actual procedure. The mean KAP-normalized E values for ICRP 60 and 103 were found 0.12 and 0.14 mSv/Gy��?cm2, respectively. This difference is quite small and may be attributed mainly to the increase of the breast weighting factor. The KAP values from tables 4 and 5 were used to calculate E, resulting to a range from 0.2 to 48.3 mSv for CA procedures and 1.2 to 33.1 mSv for PCI procedures. Effective doses in bibliography ranged between 0.3 to 15.8 mSv for a CA procedure and 0.9 to 44.7 mSv for a PCI procedure [30]. These wide ranges in patient doses may be attributed to the procedure complexity (due to patient anatomy, physician experience, etc.) as well as the different conversion coefficients used for the estimation of the effective dose. |
| Effective risk |
| Organ doses for typical CA and PCI procedures (Table 8) were used to estimate the corresponding risks, according to BEIR VII. Risks for cancer incidence per 100,000 persons are presented in Table 11 as a function of age and sex for both procedures. In this table11, the risks for lungs, breast, colon and leukaemia are provided. Risks for organs, such as stomach, liver, urinary bladder, and prostate were found to be very low; thus, not provided in the Table. The estimated risks for “other” were dominated by oesophagus cancer. R was estimated for different age and sex and is provided in the Table. R simply represents the total risk of cancer incidence or mortality. Cancer risks during cardiac procedures are dominated by lung and breast cancer. |
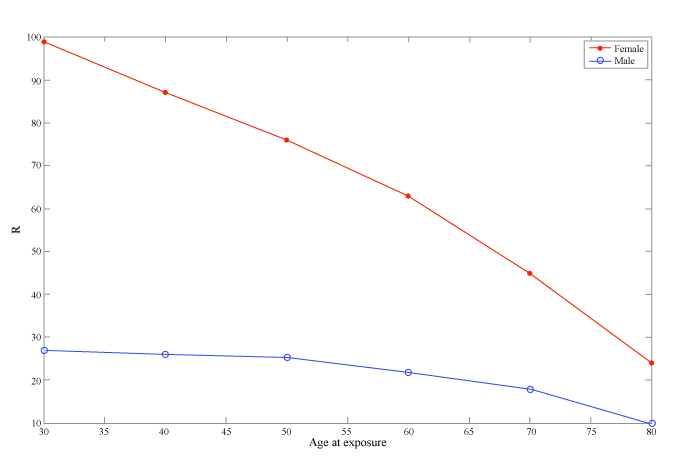
| Figure 3 illustrates the effective risk for incidence as a function of age and sex for a typical CA procedure. R is in general greater at younger ages. This may be seen in the figure, where the risk for cancer incidence is five times greater at the age of 30 than that at the age of 70. Women are more radiation-sensitive than men. Figure 3 shows that R for a woman is two to five times greater than that of a man who undergoes the same cardiac procedure at the same age, with the difference being more intense at younger ages. This difference is mainly due to the breast and lung cancer risks. |
| Comparison of effective dose with effective risk |
| According to the ICRP [2,31], the purposes of E in radiation protection for occupational workers and general public are the exposure limitation and prospective dose assessment for optimization and the retrospective dose assessment for demonstrating compliance with dose limits, or for comparing with dose constraints or reference levels. ICRP accepts the use of E for medical procedures in order to compare the relative risk i) among different procedures, ii) of similar procedures in different hospitals or iii) when using different technologies for the same procedure [31]. However, its use involves a number of assumptions and limitations. Firstly, the tissue weighting factors for the calculation of E are derived by the risks of cancer incidence, cancer mortality, life shortening and hereditary risks. These risks cannot be combined in one number, as they are rather irrelevant to each other [16]. Moreover, E has been widely used to estimate patient risks Borras et al. [10], as it can be easily converted into an approximate radiation risk, using a risk of fatal cancer of 5% per Sv averaged over the whole population [1,2]. The estimation of excess lifetime risk of developing cancer due to a radiation exposure is more reasonable to be based only on cancer risks. Therefore, the use of R seems more appropriate for such calculations. Some attention should be paid at this point. The obtained results should only be used to compare the relative risk for examinations involving radiation exposure and not to predict number of cancers caused by a medical procedure. Cancer risk estimations for the individual patient is currently impossible due to different patient anatomy which cannot be simulated by the average phantoms, genetic susceptibility to cancer, uncertainties in cancer risk factors, etc. |
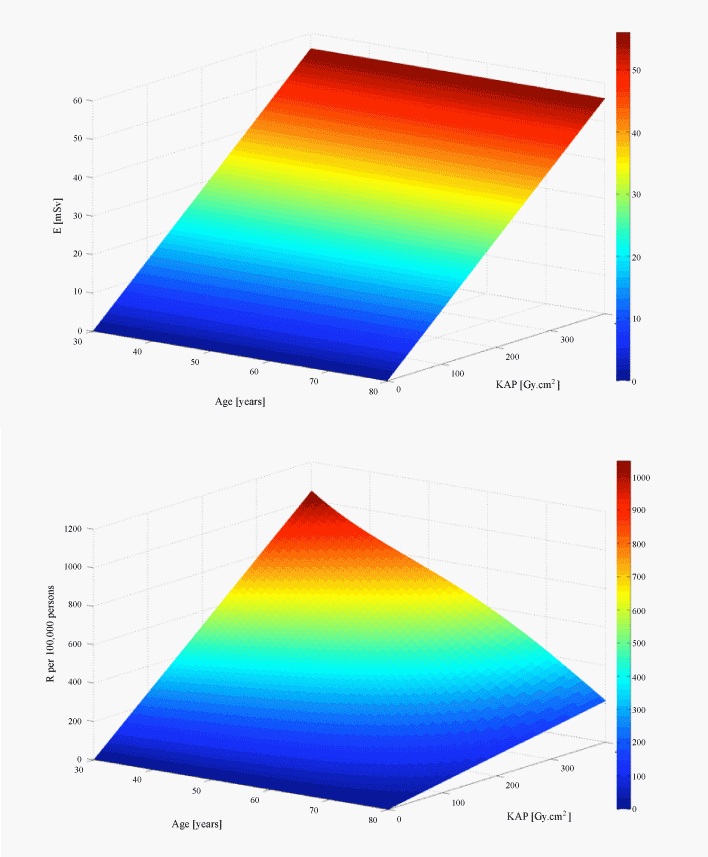
| Secondly, E is defined to be age- and sex- independent. This means that E does not take into account the age at exposure, although radiation risks are higher for paediatric and young patients than for adults and the fact that women are more radiation-sensitive than men. The most important advantage of R is that it takes into account different radiation sensitivities among ages and between male and female patients. Our calculations showed that for a typical cardiac procedure the risk of cancer incidence for a 30-year-old male is five times higher than a seventy-year-old and that the cancer incidence risk for a female patient is two times higher than the risk of a male patient (Figure 3). Figure 4 shows the dependence of E and R on radiation exposure. The KAP goes up to 400, which is the maximum KAP value found in our study. Both E and R increase with the increase of the KAP. While E remains stable for all ages, R decreases with patient age. The risk is higher at younger ages (shown in red in Figure 4b). |
| Thirdly, the use of E may lead to confusion, as both equivalent organ dose and effective dose, which is the weighted sum of equivalent doses, are measured in Sv. The unit of R is a number of induced cancers per 100,000 procedures. This may eliminate the confusion between organ dose and risk. Comprehension and awareness of radiation risks is particularly important as it allows physicians to balance risks with benefits for each patient. As already discussed, R may be used to estimate the number of induced cancers by medical exposure, while the purpose of the radiation protection is to provide physicians with a way to estimate radiation exposure in order to protect the patient. A wrong interpretation of the results that lead to fear and avoidance of medical exposure is as wrong as the ignorance of potential risks. Referring physicians should understand radiation risks, and be able to explain to patients the potential risks and help them take the right decision. The correct use of radiation protection quantities is useful for the radiation protection of the patient and the good communication between medical physicists, physicians and patients. This becomes more urgent nowadays because the use of radiation in medicine is expanding [4] and modern medicine demands shared decision making between physicians and patients [17]. Therefore, risks due to ionizing radiation have to be put into perspective with other risks (in-hospital complications, false positive or negative results, etc.) and communicated to the patient to get his/her consent. |
| Examining the practical aspects of R, we found that calculating R was as complicated as calculating E. The results for R, depending on patient sex and age, may be considered as much more complex and disturbing than the effective dose where only one number is acquired. Although this may be considered as a limitation of R, it provides important information about patient risk and may help for deep understanding and active reduction in patient exposure. Changing from E to R is difficult, as E has been extensively used for the estimation of medical exposures. However, we believe that R is currently the most accurate method of estimating radiation risks. More studies should be performed on how physicians and radiation protection experts perceive the use of R. |
| Limitations of the study |
| The first limitation of this study is that the patient study did not allow us to estimate the KAP for different angulations of the tube. However, our approximation to consider the contribution of the different angulations as equal may be considered satisfactory in order to simulate a typical cardiac procedure. Another source of error in the estimation of the organ dose is related to variations of organ location and size from the average. Nevertheless, the use of voxelized anthropomorphic phantoms that represent the average patient is the most realistic approach to the issue. Finally, the linear non-threshold model was used to derive risks for low doses from epidemiologic studies on effects of individuals exposed to high levels of ionizing radiation, such as atomic bomb survivors, patients exposed to radiation for medical treatments and animal experiments. This model has been thoroughly argued. However, the main radiation protection organizations and advisory authorities assume that low-radiation doses are associated with hazard risks [1,4,8] and this assumption was also adopted in this study. |
| Conclusion |
| Cardiac procedures can result in considerable radiation doses to the patient, even when performed with modern fluoroscopic equipment. The patient may be exposed to even higher radiation doses due to extended exposure times affected by his clinical condition. Cancer risks cannot be neglected, especially for relatively young patients. Radiation protection needs to be better communicated in order to be well accomplished. Monte Carlo simulations were performed to simulate two typical cardiac procedures. Normalized dose data were derived for the estimation of organ doses. Data provided in this study allow organ dose estimation, regardless of the operating parameters and equipment used. Equivalent organ doses were estimated and the effective dose was calculated using both ICRP recommendations 60 and 103. The differences in effective dose according the two recommendations were found small. Effective dose allows comparisons of the radiation doses among different medical procedures; however it appears to be rather inappropriate for risk communication among specialists and non-specialists. Moreover, effective dose suppresses the effect of age and sex on radiation risk. On the other hand, the effective risk is a straightforward method to communicate and understand radiation risks. We believe that the effective risk is the most accurate method, currently available, to estimate radiation risks. Work still needs to be done, however, to examine how physicians perceive the use of effective risk and the impact of its use in the day-to-day practice. |
| Acknowledgements |
| The authors would like to thank Dr. Hans Menzel for his helpful advice. |
References |
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 | Table 4 |
| Table 5 | Table 6 | Table 7 | Table 8 |
| Table 9 | Table 10 | Table 11 |
Figures at a glance
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 14513
- [From(publication date):
April-2012 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 9930
- PDF downloads : 4583
