Research Article Open Access
Purification of Schizothorax Immunoglobulin Raised Against Metazoan Parasite: A Preliminary Study
Feroz A. Shah1*, J. Mohanty2, M.H. Balkhi1, F.A. Bhat1 and Inayat Chishti1
1Faculty of Fisheries, SKUAST-K, Shuhama, J&K, India
2Central Institute of Freshwater Aquaculture, Kausalyagung, Bhubaneshwar, India
- *Corresponding Author:
- Feroz A. Shah
Faculty of Fisheries
SKUAST-K
Shuhama, J&K, India
E-mail: drferozshah@yahoo.com
Received Date: April 27, 2013; Accepted Date: May 20, 2013; Published Date: May 26, 2013
Citation: Shah FA, Mohanty J, Balkhi MH, Bhat FA, Chishti I (2013) Purification of Schizothorax Immunoglobulin Raised Against Metazoan Parasite: A Preliminary Study. J Fisheries Livest Prod 1:105. doi:10.4172/2332-2608.1000105
Copyright: © 2013 Shah FA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Fisheries & Livestock Production
Abstract
Attempts were made to purify immunoglobulins from serum of Schizothorax, Schizothorax niger, which had been immunized with Pomphorhynchus (Acanthocephalan) antigen. Initially, the protein in the serum was salted out at 50% saturation with ammonium sulphate and was chromatographed successively by gel filtration and ion exchange columns. The Pomphorhynchus kashmirensis agglutination positive fractions from ion exchange column when concentrated and checked in SDS-PAGE, three bands where observed. Since teleosts immunoglobulins have been shown to belong to a single class, the two extra bands found in our study might be the degradation products of immunoglobulin or some unpurified contaminants.
Keywords
Schizothorax niger; Immunoglobulins purification; Pomphorhynchus
Introduction
The ability of fish to synthesize Immunoglobulins in response to antigenic stimulation is well-known Branstein et al. (1998). These Immunoglobulins in fish are predominantly present in the serum, but also in the mucus of skin and intestine, as well as bile. The Immunoglobulins have been purified investigated in a number of fish species Litman (1976), Pilstrom [1]. Purification of immunoglobulin (Ig) to homogeneity is a necessary prerequisite for its further biochemical characterization. Many procedures have been developed in the past using different physical and chemical tools. In general, a combination of several tools is essential to obtain purified Immunoglobulins and specific structural characteristics have been widely investigated in fresh water teleosts Marchalonis (1969) used starch gel electrophoresis followed by gel filtration to purify Immunoglobulins from various fresh water fish species. However, no data are available until date for the immunoglobulin of Schizothorax species; the study will facilitate further studies on structural analysis of these immunoglobulin proteins.
Materials and Methods
Animals
Ten numbers of Schizothorax niger (100-150 g) were maintained at SKUAST-K Shuhama fish farm in plastic pools with continuous aeration. These were fed with standard pelleted carp diet twice daily at a rate of 3% of the body weight.
Immunization
The fish were immunized intraperitoneally with WW antigen of Pomphorhynchus kashmirensis at a dose rate of 1 mg wet weight in 0.2 ml volume per fish along with FCA. Those were boostered after 7 days of injection with the same doses and were bled after 14 days of booster by cardiac puncture, serum were separated by centrifugation, pooled and stored at –20°C until use.
Protein determination
Total protein concentrations in serum or chromatographic fractions were quantified by Bradford [2] dye binding method. Bovine serum albumin was used as the standard.
Ammonium sulphate precipitation
Precipitation concentration was determined. After concentration these fractions, the protein concentration was determined to be 4.0 mg/ml. 4 ml of pooled sera was diluted with equal volume of 0.1 M phosphate buffer with 0.14 M NaCl, pH 7.2 (PBS) and 8 ml of saturated ammonium sulphate solution was then added drop wise with continuous stirring, the solution was stirred for 45 minutes further at room temperature. The precipitate was collected by centrifugation at 10,000 x g for 30 minutes and was dissolved in 2 ml PBS. The solution was then dialyzed extensively against frequent changes of PBS followed by clarification centrifugation at 10,000 g for 30 minutes at 4°C. The supernatant was loaded onto the gel filtration column after determining the protein concentration and the Ig specificity by agglutination.
Gel filtration chromatography
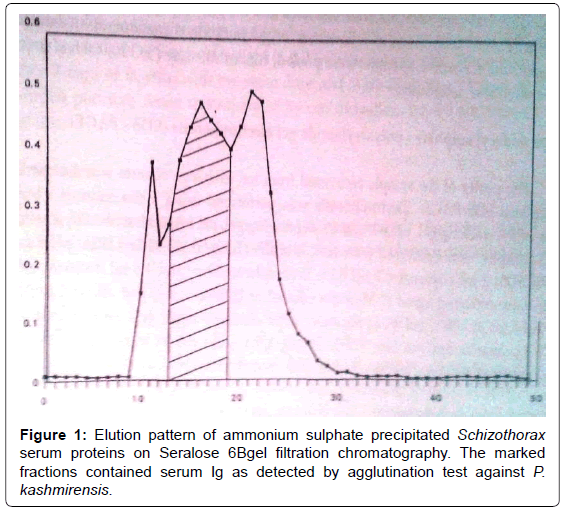
Gel filtration was carried out on Seralose 6B (SRL, India) column (2.2 x 25 cms) with PBS, at a flow rate of 6 ml/hr with automatic collection of 3 ml fraction by LKB 2112 redifrac fraction collector. The absorbance of the fraction was monitored at 280 nm and the immunoglobulin specificity was checked by agglutination test. The agglutination positive fractions were pooled, dialyzed against 10 mM Tris HCL, pH 7.2, concentrated to 2 ml by reverse dialysis against polyethylene glycol (PEG) 20,000 and applied on to ion exchange column after determining the protein concentration.
Ion exchange chromatography
Ion exchange chromatography was run on DEAE cellulose (DE- 32 Whatman) column (1.6 × 7) in 10 mM Tris HCL, pH 7.2. The concentrated fractions from gel filtration chromatography were loaded and run at 20 ml/h. after an initial elution period (about 20 ml); a linear sodium chloride gradient (0-0.3 M) in 10 mM in Tris HCL, pH 7.2 was applied. 3 ml fractions were collected and the protein absorbance at 280 nm and Ig specificity were determined. The agglutination fractions were pooled, concentrated by PEG 20,000.
SDS-PAGE
The purity of the sample recovered from ion exchange column was assessed by SDS-PAGE. Electrophoresis was performed using a discontinuous buffer system at 3% stacking gel, pH 6.8 and 4% separating gel, pH 8.8 as described by Laemmli [3]. Samples were processed with sample buffer (1.7 ml 0.625 M Tris HCL, pH 6.8; 4.5 ml 10% SDS; 2ml Glycerol; 0.2 ml 0.1% Bromophenol blue; and 1.6 ml distilled water) without any reducing agent and allowed to stand for 2 minutes at 100°C before application. After electrophoresis, gels were stained with 0.25% Coomassie brilliant blue R250. Photographs could not be taken due to the high fragile nature of the gel at this low concentration of acrylamide.
Results and Discussion
The present study was aimed at to purify the serum Immunoglobulins from Schizothorax spp. fish. Anti- Pomphorhynchus kashmirensis activity detected by agglutination served as serological marker for these Immunoglobulins. The initial pooled sera had a protein concentration of 40 mg/ml and a protein agglutination titre of 1:120 after ammonium sulphate precipitation, the solubilized pellet was found to be agglutination positive and the protein concentration was 8.5 mg/ml. A combination of Ammonium sulphate precipitation with gel filtration chromatography and ion exchange chromatography has been successfully used by several workers to purify the serum Immunoglobulins from fish species [4-7]. The solubilized pellet when fractionated on gel filtration chromatography, the elution profile revealed 3 peaks, However, agglutination was only associated with the Ig. Since the pooled agglutination-positive fractions from ion exchange column belonged to two separate peaks, the presence of contaminating proteins could be a certain possibility. Otherwise, there might be certain degree of degradation of Ig during our attempt to concentrate these proteins from the dilute column fraction. Lim [8] has also not been able to concentrate the eluted proteins from weak solutions (<1 μg/ml) without gross denaturation. Further studies are being continued in this aspect to eliminate the anomaly and to purify the Ig to homogeneity. On further purification of the concentrated Ig containing fractions from gel filtration on DEAE cellulose chromatography, an elution profile of four peaks was obtained (Figure 2) however, only the second and third peaks eluted at 0.16 M and 0.18 M NaCl concentrations, respectively were found to be agglutination positive after concentrating these pooled fractions, the protein level was found to be 0.95 mg/ml (Figure 1 and 2).
When 20 μg protein samples from ion exchange column were analyzed by SDS-PAGE under non reducing conditions, three distinct bands were observed. Since the fish species of order Teleost has been characterized as single tetrameric IgM like macroglobulin [1,9,10]. So the purified products in our experiment should have shown a single band in SDS-PAGE under non-reducing condition. The two extra bands present could be due to some contaminated proteins or due to some proteolytic degradative products of the Ig. Further studies are being continued in this aspect to eliminate the anomaly and to purify the Ig to homogeneity.
Acknowledgements
Authors are thankful to DST New Delhi for providing financial assistance to this work through a sponsored Fast Track Project.
References
- Pilstorm L, Bengton E (1996) Immunoglobulin in fish- genes, expression and structure. Fish Shellfish Immunol 6: 243-262.
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254.
- Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature 227: 680-685.
- Voss EW Jr, Groberj WJ Jr, Fryer JL (1978) Binding affinity of tetrameric coho salmon Ig anti-hapten antibodies. Immunochemistry 15: 459-464.
- Clerx JPM, Castle AD, Bol JF, Gerwig GJ (1980) Isolation and characterization of the immunoglobulin of Pike (Esox lucius L.). Vet. Immunopathol 1: 125-144.
- Itami T, Takahashi Y, Okamoto T, Kubono K (1988) Purification and characterization of immunoglobulins in skin mucus and serum of Ayu. Nip Sui Gak 54: 1611-1617.
- Kobayashi K, Tomonaga K, Tanaka S (1992) Identification of second immunoglobulin in the primitive shark, the frill shark (Chlamydoselachus angineus). Dev Comp Immunol 16: 295-299.
- Lim PL (1987) Isolation of specific IgM monoclonal antibodies by affinity chromatography using alkaline buffers. Mol Immunol 24: 11-15.
- Dorson M (1981) Role and characterization of fish antibody. Dev Biol Stand 49: 307-319.
- Buckmann K (2003) Antiparasitic immunity in fish. Fish Shellfish Immunol 5: 212-222.
Relevant Topics
- Acoustic Survey
- Animal Husbandry
- Aquaculture Developement
- Bioacoustics
- Biological Diversity
- Dropline
- Fisheries
- Fisheries Management
- Fishing Vessel
- Gillnet
- Jigging
- Livestock Nutrition
- Livestock Production
- Marine
- Marine Fish
- Maritime Policy
- Pelagic Fish
- Poultry
- Sustainable fishery
- Sustainable Fishing
- Trawling
Recommended Journals
Article Tools
Article Usage
- Total views: 14327
- [From(publication date):
September-2013 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 9689
- PDF downloads : 4638