Review Article Open Access
The Role of Platelets in Viral Hemorrhagic Fevers
Dermot Cox1*, Maria S Salvato2 and Juan Carlos Zapata2
1Molecular and Cellular Therapeutics, Royal College of Surgeons in Ireland, Ireland
2Institute of Human Virology, University of Maryland School of Medicine, Baltimore, Maryland, 21201, USA
- *Corresponding Author:
- Dermot Cox
Molecular & Cellular Therapeutics
Royal College of Surgeons in Ireland
123 St. Stephens Green, Dublin 2, Ireland
Tel: +353-1-402-2152
Fax: +353-1-402-2453
E-mail: dcox@rcsi.ie
Received Date: February 02, 2013; Accepted Date: April 04, 2013; Published Date: April 08, 2013
Citation: Cox D, Salvato M, Zapata JC (2013) The Role of Platelets in Viral Hemorrhagic Fevers. J Bioterr Biodef S12:003. doi: 10.4172/2157-2526.S12-003
Copyright: © 2013 Cox D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors and source are credited.
Visit for more related articles at Journal of Bioterrorism & Biodefense
Abstract
Arenaviridae, Bunyaviridae, Filoviridae and Flaviviridae are all RNA viruses that can induce a coagulopathy in infected patients. While there is a paucity of information on the cause of this coagulopathy, these viruses all produce a severe thrombocytopenia. Here, we review the literature on viral hemorrhagic fever and discuss the role played by platelets in the coagulopathy. We also discuss the possibility of using anti-platelet agents to moderate the coagulopathy, a strategy that might have a role to play in treating patients infected by these viruses.
Keywords
Platelets; Arenaviridae; Bunyaviridae; Filoviridae; Flaviviridae; Dengue; Viral hemorrhagic fever; Disseminated intravascular coagulation; Anti-platelet agents; Biodefense
Hemorrhagic Fever Viruses
Viral hemorrhagic fevers (VHF) are a group of illnesses that are characterized by a loss of homeostasis leading to increased bleeding and shock. VHFs are caused by enveloped RNA viruses from four different families: Arenaviridae, Bunyaviridae, Filoviridae, and Flaviviridae [1]. The bleeding signs of VHF are more frequently associated with Marburg and Ebola (Filoviruses), Crimean Congo Hemorrhagic Fever (Bunyavirus), and South American hemorrhagic fever (Arenaviruses), but less frequently associated with Dengue and Yellow Fever (Flaviviruses), Rift Valley Fever (Bunyavirus) or Lassa fever (Arenavirus) [2]. A common characteristic of all hemorrhagic fever viruses is that they infect vascular endothelial cells and diminish platelet numbers or function. However, these viruses differ greatly in their replication strategies, extent of cytopathic effects, and extent of immunopathology [3]. Here, we review information on the effects of VHF on platelet functions, with emphasis on dengue hemorrhagic fever pathogenesis.
Biothreat of Viral Hemorrhagic Fevers (VHF)
VHFs have a number of properties that make them potential bioweapons, whereas the arenaviruses and filoviruses are aerosolizable; such high doses are needed for infection that they are unlikely to be transmitted that way. Outbreaks of VHF generally center on bleeding patients, as a source of nosocomial infection for health care personnel. Also, the effects of VHF can be sufficiently dramatic to instill terror in a civilian population. Even if many of these diseases can be medically managed, this often requires treatment in ICU units. Thus, even if only a small number of patients develop the disease, as a result of an attack the image of infected patients bleeding from several orifices, the risk of spread of infection from blood and the pressure placed on ICU resources would all serve to terrorize a civilian population.
One of the biggest problems in dealing with a VHF outbreak due to a bioterrorist attack is the ability to identify the causative agent. Outbreaks occurring in their endemic regions are easier to identify than those occurring in new regions. Thus, a patient presenting with hemorrhagic fever in Malaysia is most likely to have dengue fever, while in Eastern Europe, it would be CCHF. Doctors are aware of these endemic diseases and look out for them in patients. If there were an imported case or a covert release of a VHF agent in a US city, it would take doctors a long time to appreciate that the patients had VHF, as they would not have any of the typical risk factors. Furthermore, it would be very difficult to identify the causative agent, as most VHFs have similar presentations. Even if there were anti-viral agents available, they would be unlikely to be effective against all agents, as the viruses are from different families. However, a common factor with all of these viruses is their ability to evade the innate immune system and compromise platelet function. Thus, platelets are a potential drug target in the treatment of VHFs. Therefore, the study of the role of platelets in the VHF pathogenesis may lead to the identification of new drug targets that will not only inhibit viral replication, but will also improve the antiviral immune response, preventing the onset of the hemorrhagic disease. While targeting the platelet may not inhibit viral replication, it may provide time for the immune system to clear the virus, and thereby, preventing the onset of hemorrhagic fever.
Flaviviridae
Epidemiology of flavivirus hemorrhagic fevers: Dengue fever (DF) is caused by Dengue virus (DENV), a positive-strand RNA virus with four recognized serotypes [4]. It is transmitted by the Aedes aegypti mosquito, and is endemic in Asia and South America, and is expanding to areas that were considered DENV free [5,6]. Worldwide, there are around 100 million infections per year, with case fatality rates between 1-15% [7,8], depending on whether a person gets simple DF or the more severe Dengue hemorrhagic fever (DHF). Each DENV serotype is capable of inducing severe disease, but if a patient was infected with DENV serotype 1 and acquires serotype 2 or 3, there is a higher probability of developing hematopathology, characterized by a massive thrombocytopenia and disseminated intravascular coagulation (DIC). There are approximately 500,000 cases annually of severe DHF [7-10]. In some cases, this progresses to complete circulatory collapse, which is known as dengue shock syndrome (DSS) [9,11].
Yellow fever (YF) is another member of Flaviviridae (transmitted by mosquitos: Aedes albopictus, A. africanus, A. hemagogus or A. sabethes). YF is endemic in tropical and subtropical areas of South America and Africa, where it causes around 200,000 cases per year, with around 30,000 deaths [12]. After initial infection, there is an “acute” phase with flu-like symptoms that can be resolved in 3 or 4 days. However, 15% of YF patients enter a phase of intoxication, which is characterized by hepatic and renal failure and hemorrhage. Up to 30% of patients in the intoxication stage will die. Yellow fever hemorrhagic fever occurs just as antibodies appear in the circulation, suggesting a potential role for antibodies in the thrombocytopenia [13].
Other flaviviruses include Omsk hemorrhagic fever virus [14], and Kyanasur Forest virus [15], which are transmitted by ticks, and also cause hemorrhagic fever.
Biothreat of Dengue virus: Due to its significant public health impact, the absence of a vaccine, and a well-characterized pathogenesis, we will describe in more detail the known facts surrounding DHF.
Dengue virus structural features: All four of the DENV serotypes (DENV-1, 2, 3 and 4) are believed to arise from a common ancestor, and homology between the viruses can be as low as 60%, and diversity is further enhanced by the existence of a number of genotypes within each serotype [16]. The mature virus is a 50 nm diameter sphere, consisting of a central electron dense core surrounded by a lipid bilayer and an external, icosahedral protein scaffold [17]. The core contains the 11 kb RNA genome encapsulated by multiple 11 kDa capsid (C) proteins, while the protein scaffold consists of 180 copies each of a 53 kDa envelope (E) protein, and an approximately 8 kDa membrane (M) protein; both of which are anchored in the lipid bilayer. In the mature virus, the membrane protein lies completely beneath the envelope protein [17].
Dengue virus replication: The virus enters host cells by receptormediated endocytosis [18,19], where low-pH conditions in the endosome induce uncoating of the viral genome, followed by translation and replication of the virus [20,21]. The genomic RNA is translated as a single polyprotein, which is co- and post- translationally processed by virus encoded and host proteases, into the 3 structural (envelope (E), pre-membrane (prM) and capsid proteins), and 7 non-structural (NS1, NS2A, NS2B, NS3, NS4A, NS4B and NS5) proteins [22,23]. The non-structural proteins initiate the replication of the viral genome, in close association with the endoplasmic reticulum (ER), and the newly synthesized RNA is packaged by the C protein, forming a nucleocapsid complex. The nucleocapsids bud from the ER and become enveloped by a lipid membrane, carrying with it associated E and the prM proteins. These form prM/E heterodimers that subsequently associate into trimers [17,23], which on the newly formed immature virus project from the surface as 60 trimeric spikes, with a diameter of approximately 600 Angstroms [24,25]. After budding from the ER, the immature viruses transit the secretory pathway, where the prM protein is cleaved by the cellular furin protease, after a major conformational change induced by the acidic environment of the trans-Golgi environment [24,25]. The cleaved “pr” domain of the prM protein remains in association with the essentially mature virus particle, preventing premature membrane fusion inside the cell, until exposure to a neutral pH in the extracellular milieu induces dissociation of the “pr” peptide in the final step of virion maturation [25]. Due to inefficient furin cleavage, as much as 30% of virions released are immature virions [19]. While these immature virions are not infectious, they do bind to Fc bearing cells in the presence of anti-prM antibodies [26].
Expression of the prM and E proteins (in the absence of the capsid protein) leads to the formation of slightly smaller coreless particles [27]. These recombinant subviral particles (RSPs) are similar in immunogenicity to native virions [28], and as such represent an easily manipulable system, as mutation of the furin cleavage site can generate either predominantly “mature” or “immature” RSPs [29]. These RSPs, therefore, represent an ideal tool to investigate the interaction of the dengue E or prM/E proteins with cells in a non-infectious system.
While the majority of the non-structural proteins are solely intracellular during infection, there is one notable exception, the 50 kDa NS1 protein. Numerous studies have shown that the NS1 glycoprotein is exported through the secretory pathway to the cell surface, and that it can dissociate from the plasma membrane and accumulate in the extracellular medium as a hexamer [30]. In vivo, high levels of NS1 are found in the sera of dengue infected patients, where it may have an immuno-modulatory role through its binding to the complement regulatory factor H [31].
Dengue clinical characteristics: Dengue has a wide spectrum of clinical presentations. While most patients recover following a self-limiting non-severe clinical course, a small proportion progress to severe disease, characterized by plasma leakage, with or without hemorrhage. Symptomatic DENV infections are grouped into three categories: undifferentiated fever, DF and DHF. DHF is further classified into four severity grades, with grades III and IV being defined as DSS [8]. Infection with any of the four serotypes causes a similar clinical course that may vary in severity, depending on a number of risk factors. Important risk factors influencing the proportion of patients who have severe disease during epidemic transmission include the serotype of the infecting virus and the immune status, age, previous exposure to different serotypes and genetic background of the patient [32].
Manifestations of DF depend on the age of the patient. Infants and children younger than 15-years may have an undifferentiated febrile disease, accompanied by a maculopapular rash. After an incubation period of 4 to 7 days, older children and adults will seek treatment with either a mild febrile illness or an incapacitating disease, characterized by the rapid onset of high fever, in combination with severe headache, retro-ocular pain, muscle and joint pain, rash, nausea and vomiting, diarrhea, abdominal pain, malaise and lymphadenopathy. Mild signs of pistaxis, petechiae, gingival bleeding, hematuria and menorrhagia are common. Leukopenia (commonly caused by a neutropenia) and lymphocytosis are usually seen (atypical lymphocytes), and a moderate thrombocytopenia may be observed in DF, especially in those with hemorrhagic signs [33,34].
DHF is primarily a disease of children under the age of 15 years, although it may also occur in adults. It has sudden onset of fever, and a variety of non-specific signs. The major pathophysiologic hallmarks that determine disease severity and distinguish DHF from DF are (i) hemorrhagic manifestations (positive tourniquet test or spontaneous bleeding), (ii) increased vascular permeability, resulting in loss of plasma from the vascular compartment, leading to hemoconcentration or fluid effusion in chest or abdominal cavities and shock, and (iii) a disorder of homeostasis involving vascular changes, thrombocytopenia, and coagulopathy. Towards the end of febrile phase, total leukocytes and neutrophils are reduced, just prior to an increase in both typical and atypical lymphocytes [32,35]. The liver is usually palpable early in the febrile phase, with mildly elevated liver enzymes [36], and clotting factor abnormalities [37-39]. Pleural effusion and ascites may also be detected, and may predict the development of DSS. Intense abdominal pain is a frequent complaint that appears shortly before the onset of shock.
The critical stage of the disease is reached at the end of the febrile phase, accompanying or shortly after rapid drop in temperature, with varying degrees of circulatory disturbance [8,32]. The patient is often in a sweat, restless with cool extremities. In mild DHF cases (WHO grades I and II), the changes in vital signs are minimal and transient. Patients recover spontaneously or shortly after a brief period of treatment. The life-threatening DSS stage occurs at the time of or shortly after defervescence, which is characterized by a rapid, weak pulse with narrow pulse pressure or hypotension, and evidence of the early stage of shock (grade III). Although the course of shock is short, it is life-threatening for the patients. If patients do not receive prompt and appropriate treatment, a stage of profound shock may set in, in which pulse and blood pressure become undetectable (grade IV), resulting in death within 12 to 36 h after onset [8,32]. Prognosis depends on early recognition of DSS cases and adequate treatment of shock. Uncorrected shock will result in development of metabolic acidosis, severe bleeding from the gastrointestinal tract and other organs. Recovery from adequately treated DSS is short and uneventful, occurring within 2 to 5 days, although pleural effusions and ascites may be detected for a little longer. The course of DHF/DSS is approximately 7 to 10 days, and usually shows no prolonged sequelae [32,37].
Dengue pathogenesis: Little is known about the pathogenesis of DHF or DSS. It is assumed that antibodies generated during the first exposure enhance infection with the second serotype [40-42], but as this occurs with other viral infections, it does not explain why hemorrhagic fever is not seen with other infections. The presence of antibodies to DENV enhances replication, rather than inhibiting it in a process known as antibody-dependent enhancement (ADE) [42,43]. This occurs through an interaction with Fc receptors [26,42,44,45], although it can also involve other receptors [46]. This process has also been shown to be regulated by complement generation [47,48], and to alter free radical and cytokine production [49].
Role of genetics in dengue fever pathogenesis: Epidemiological studies have shown that particular ethnic groups have increased tendency to developing severe DF. Past outbreaks of DHF and DSS in Cuba have implicated ethnicity as a strong predictor of disease severity. Epidemiological studies from outbreaks in 1981, 1997 and 2001 have all consistently shown individuals of European ancestry to be over-represented, in terms of morbidity and mortality compared to individuals of African ancestry [50,51]. Strikingly, the same studies showed individuals of mixed African/European descent to carry an intermediate risk. A recent study of Dengue in Brazilian populations, showed Afro-Brazilian and African ancestry (determined genetically through ancestry informative markers, and after controlling for income), as protective for DHF [52]. In Asia, there have been reports of higher incidences of DHF among Han Chinese compared to Malaysians [53,54]. These reports all suggest that populations indigenous to Dengue regions have developed a degree of immunity to DHF/DSS, and this immunity may have a genetic basis.
Numerous studies have been reported in the literature, seeking to identify host genetic predictors of Dengue severity. All have followed a candidate gene design, focusing principally on the HLA and various other immune related genes (for summary of studies see [55]). However, none have identified consistent, reproducible genetic predictors of disease outcome.
Filoviridae
The most severe VHF is caused by the Filoviruses, Marburg and Ebola. Marburg was discovered after an outbreak in laboratory workers in Marburg, Germany in 1967, and the Ebola virus was first identified in an outbreak in Democratic Republic of the Congo in 1976. They are thought to be carried by fruit bats in the endemic area of central Africa. Both are of interest as biological warfare agents due to their high case fatality (up to 90%) [56]. Both have a similar clinical presentation with an incubation period of around 6-days, followed by a flu-like syndrome, often accompanied by vomiting, diarrhea and abdominal pain. Over the following week, the patient deteriorates developing hypotension, bruising, bleeding and evidence of hemorrhagic shock. After 10 days, some patients begin to show signs of recovery, while the remaining patients eventually lapse into coma and die. A high virus titer is a predictor of poor outcome [57].
There is very little data on the nature of the Ebola hemorrhagic fever in humans, and much of the information comes from animal studies. Platelet activation was found in Ebola infected rhesus monkeys [58,59]. A common finding is a significant immune suppression, despite the presence of high levels of antigen. This manifests as severe suppression of the adaptive immune system, including a failure of antigen presenting cells to respond, extensive apoptosis of lymphocytes, and the generation of high levels of cytokines [60]. High levels of IFN have been associated with alteration in megakaryocyte development and platelet production in other VHF [61]. In patients infected with Ebola, there is a markedly increase in interferon (IFN)-gamma, IFN-α, IL-2, IL-10, and TNF-α, that could contribute not only to a high immune activation, but also to megakaryocyte malfunction [62].
Bunyaviridae
This family of RNA viruses is associated with a number of hemorrhagic fevers. In particular, Phleboviruses, such as Rift Valley fever virus, Nairoviruses like Crimean-Congo Hemorrhagic fever and Hantaan virus from the Hantavirus genus.
Crimean-Congo Hemorrhagic fever: Crimean-Congo Hemorrhagic fever (CCHF) is a tick-borne zoonosis first described in 12th century Tajikistan, but not identified as a virus until it emerged in the Crimea in 1944, and was subsequently shown to be the same virus that caused a disease outbreak in the Congo in animals in 1956 [63]. Humans can be exposed to the virus through the bite of an infected tick, contact with an infected patient, or infected livestock. CCHF can have a case fatality rate as high as 50%. The incubation period for CCHF is around 1 week, and then the patient enters the first phase, characterised by non-specific symptoms such as fever, headache, and muscle aches. During this phase, the platelet count begins to fall, the liver enzyme levels begin to rise, and viral load declines. By week 2, severe cases can enter into a second phase, which is associated with disseminated intravascular coagulation (DIC), with accompanying hemorrhage. Within 2-3 days, the patient either dies or recovers. The extent of thrombocytopenia is a good predictor of outcome in infected patients [64,65]. The CCHF virus has three genomic RNA segments (L, M and S). The segment L encodes the RNA polymerase. Recent structural studies show that CCHFV (bunyavirus) and Lassa virus (arenavirus) nucleocapsid proteins are more similar than N proteins of CCHFV and other bunyaviruses [66].
Other bunyavirus hemorrhagic fevers: Other significant members of the Bunyaviridae associated with hemorrhagic disease are members of Hantavirus and phleboviruses, such as Rift Valley fever virus [67]. Hantaan virus produces a hemorrhagic fever renal syndrome (HFRS), with around 100,000 cases each year and 3,000 deaths in China alone, and other Hantaviruses cause pulmonary syndromes in the Americas [68]. Thrombocytopenia is a common clinical finding in infections caused by bunyaviruses, and direct binding to platelets was demonstrated with the phlebovirus SFTS virus [69].
Arenaviridae
The arenaviruses including Old World Lassa fever virus and the New World arenaviruses Junin, Guanarito, Machupo and Sabia are also capable of causing hemorrhagic fever [70]. They are carried by a variety of rodent species, and the infection is usually acquired through close contact with excrement or urine from infected animals. Although 80% of the infections are asymptomatic, some cases (for example during pregnancy, during nosocomial exposure, or after transplantation with infected blood) evolve into a more severe syndrome, with hepatic and pulmonary complications and hemorrhage. In West Africa, Lassa fever virus is responsible for about 300,000 cases per year with 5,000 deaths. Interestingly, platelet numbers during Lassa fever disease can be within the normal range, but arenaviral infection appear to induce a soluble factor that inhibits platelet aggregation [71,72]. Soluble P-selectin serum levels increase during arenavirus infection of mice, indicating that platelets can be activated during infection [73]. In vitro culture of human hematopoietic progenitors with Junin virus induces IFNα/ß, which causes the release of P-selectin from platelet granules [74]. Thus arenaviruses can indirectly have negative effects on platelet function and production.
Role of Platelets in VHF Pathogenesis
Platelet biology
Platelets are anucleate derivatives of megakaryocytes, and are the smallest and most numerous corpuscular component of blood [75]. The typical shape of resting platelets is discoid, and upon activation, they undergo a shape change to a globular form with pseudopodia. As platelets lack nuclei, their ability to synthesize proteins is limited, although they can translate stable megakaryocyte mRNA [76]. Platelets contain granules (α-granules, dense granules and lysosomes) for storage of important bioactive molecules, such as ADP, serotonin, calcium, and proteins such as fibrinogen and von Willebrand factor [77].
Platelets play a crucial role in thrombus formation. First, single platelets bind to a site of damaged endothelium, where collagen in the underlying extracellular matrix has been exposed. Platelet surface receptors for collagen α2β;1 and GPVI), fibrinogen (GPIIb/IIIa), and von Willebrand factor (GPIb/IX/V), help to bind platelets to the exposed endothelium, resulting in platelet activation [78]. Upon activation, GPIIb/IIIa undergoes a conformational change that allows it to bind soluble fibrinogen [79]. At the same time, cytoplasmic granule contents are released, triggering activation of other platelets that are then recruited into the growing thrombus, plugging the endothelial damage.
There are two general pathways to platelet activation. The first is mediated by “weak agonists” such as low-dose ADP, and involves activation of phospholipase A2 releasing arachidonic acid to be converted to thromboxane A2 by cyclooxygenase (COX). The second pathway is activated by “strong agonists”, and is mediated by phospholipase C.
Recently there has been a growing awareness that platelets also play an important role in the innate immune system [80,81]. Platelet granules contain platelet microbicidal peptides (PMP) and proteins which act to sterilize the wound [82]. However, the ability to activate platelets has been exploited by some pathogens to promote colonization and avoidance of the host immune responses [83]. Thus, strains of S. aureus that are susceptible to PMPs in vitro, are less likely to cause endovascular infections in humans, and are more rapidly cleared from the bloodstream than bacteria that are PMP-resistant [84-86]. Similarly, in experimental endocarditis, S. aureus that is resistant to PMPs in vitro is more virulent [87].
Platelet function
The primary role of platelets is in hemostasis; this is achieved through three distinct properties of platelets. First, platelets have the ability to adhere to the site of injury (adhesion). Secondly, they become activated and clump together (aggregation), and also secrete the contents of their granules (secretion). Adhesion is usually the critical initial stage in the process and acts to anchor the thrombus to the site of injury, and trigger the signaling events that lead to aggregation of the activated platelets. Secretion is also a significant step in the process, as the granules contain many bioactive substances, such as ADP, which helps activate other platelets.
As platelets are in very high concentration in the blood and are the first responders to an injury, they play a major role in preventing infection. They do this by adhering to the pathogen and secreting antimicrobial peptides and chemokines that attract immune cells.
Platelet pharmacology
Due to the significant role played by platelets in the pathogenesis of cardiovascular disease, there has been a lot of effort to develop anti-platelet agents. The first and most widely used anti-platelet agent is aspirin, which acts to inhibit cyclooxygenase, and thus, platelet activation by all weak agonists. The second class of anti-platelet agents is the P2Y12 antagonists, such as clopidogrel and prasugrel. P2Y12 is the ADP receptor on platelets, and is critical in the amplification of thrombosis by ADP secreted from activated platelets. The third class of anti-platelet agent is the GPIIb/IIIa antagonists (abciximab, eptifibatide and tirofiban), which prevent fibrinogen binding and thus aggregation, but do not effect platelet activation or secretion. These drugs all block platelet function by a range of different agonists, and are often used together. Thus, clopidogrel and aspirin are usually administered together, as are aspirin and GPIIb/IIIa antagonists. Combination therapy provides robust inhibition of platelet function, however; it is also associated with increased bleeding risk.
Platelets, hemorrhagic fever and sepsis
At first glance there is much in common between VHF, sepsis and systemic inflammatory response syndrome (SIRS). Cytokine storm, characterized by massive increases in cytokine levels, may be involved in all of these disorders. Platelet dysfunction is certainly associated with SIRS [88,89], and sepsis [90-94]. Thrombocytopenia can be due to platelet activation alone [91], and can be independent of thrombin generation [95]. However, the precise role played by platelets is unclear. It is also unclear whether the pathogens are acting only indirectly through platelet-activating cytokines, or whether their direct interactions with platelets have further affects on platelet trafficking or platelet functions.
Platelets play an important role in septic shock [81,90,96-102]. Activation of platelets leads to degranulation and adhesion to leukocytes and endothelial cells. They have also been shown to bind to neutrophils, forming neutrophil traps [103]. As platelets contain many vasoactive substances, such as serotonin, platelet factor 4 and histamine, their degranulation can down-regulate platelet production [104], and disrupt endothelial function. This platelet-endothelial interaction is also important in the pathogenesis of shock [98,102,105- 108]. In sepsis, neutrophil binding to platelets leads to platelet sequestration in the liver [93], and adhered platelets support neutrophil adhesion [109]. Endothelial cell infection can lead to activation and subsequent adhesion of platelets [110]. Thus, endothelial cell activation has been suggested to drive HIV-associated thrombocytopenia [111], and DENV-infected endothelial cells support platelet adhesion [112]. The presence of activated platelets or platelet-leucocyte aggregates has also been shown to lead to activation of the endothelium [113]. Preliminary evidence suggests that the use of aspirin, and/or clopidogrel, may reduce mortality in sepsis [114,115]. However, while anti-platelet agents preserve platelet numbers, they do so by inhibiting platelet function, which may exacerbate hemorrhagic complications in thrombocytopenic patients.
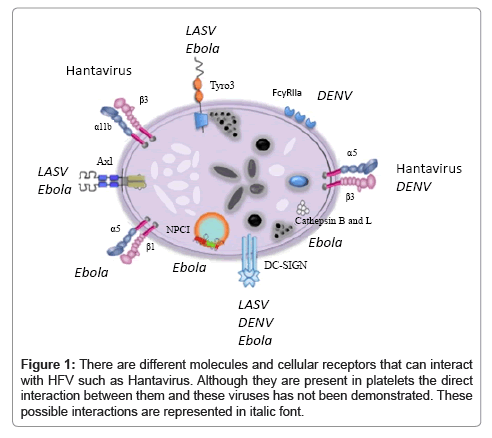
The homeostatic control of coagulation depends on thrombin balancing pro- and anti-coagulant forces, as well as pro- and antifibrinolytic consequences. In most VHF infections, this balance is lost, triggering excessive and dysregulated thrombin generation in vivo, leading to disseminated intravascular coagulation (DIC) [116]. The predominant functional consequence of thrombin generation is influenced by the nature of the inciting agent. In bacterial infections, this is typically microvascular thrombosis leading to organ failure. In DHF, there is hyperfibrinolysis, where thrombin activation of protease activated receptors induces vascular hyper-permeability resulting in DSS. Linked to the hypothesis of the central role played by DENVplatelet interactions (Figure 1), platelet activation would lead to exposure of anionic phospholipid surfaces that can catalyse thrombin generation by 250,000 fold [117]. These surfaces also assemble and catalyse fibrinolytic reactions and secrete plasmin [118], the major fibrinolytic enzyme that lyses fibrin clot. Platelet-bound plasmin is also resistant to inhibition, and high plasmin levels can in turn induce platelet dysfunction, whilst also interfering with fibrinogen interactions with GPIIb/IIIa [118].
Virus-mediated platelet activation can happen through direct interaction with platelets, or indirect interactions. In the case of a direct interaction, this can be due to virus directly binding to platelets (Figure 1), or can be mediated by a plasma protein that binds to the virus, that can in turn bind to the platelet. Indirect activation of platelets can occur through a number of different mechanisms. Viruses can activate neutrophils, which then bind to platelets, clearing them from the circulation (Table 1). Alternatively, virus can activate the immune system leading to increased levels of circulating cytokines, which could play a role in activating platelets (Table 1). Direct virus binding to platelets could affect platelet function in coagulation and innate immune recognition (Table 1). Finally, infection of megakaryocytes could lead to reduced platelet production (Table 1).
| Mechanisms of HFV-induced thrombocytopenia | |||
|---|---|---|---|
| Direct Platelet Destructiona | Immune destruction of plateletb | Megakaryocyte impairmentc | Inhibition of platelet functiond |
| Hantavirus [166] | SFTS [69] | Hantavirus [139] | Hantavirus [166] |
| DENV [112,167,168] | DENV [169] | LASV [71] | |
| JUNV [74,170] | JUNV [72] | ||
| Ebola [62] | |||
aDestruction of platelets by direct interaction: SomeHFV can bind platelets directly
causing activation and granules release.
bImmunological destruction of platelet-virus complexes: Thrombocytopenia can be
mediated by macrophages sequestration of platelet-virus complexes at the local
site or/and in the spleen, platelets-virus-leukocyte aggregation and subsequent
phagocytosis by macrophages or destruction mediated by platelet-virus associated
antibodies.
cMegakaryocytes or Megakaryocyte precursors impairment: Some HFV can
infect megakaryocytes or their precursor causing reduction in platelets number or
impairment in their function.
dInhibition of platelet function: Some unidentified soluble factors present in plasma
from infected patients can inhibit aggregation of platelets from healthy individuals.
Table 1: Four main mechanisms by which Haemorrhagic fever viruses (HFV) can induce thrombocytopenia.
Although for years, little was known about the interaction between platelets and viruses, recently there is an increased interest on this issue. Not only do hemorrhagic fever viruses subvert platelet production and hemostatic function, they are also able to subvert the platelet’s immunological role in pathogen recognition and control. Viruses are not unique in their ability to interact with platelets and experience with other pathogens that interact with platelets can provide some insight into the potential viral interactions.
Platelet activation by direct interactions with microbes: There is abundant evidence of platelet activation induced by bacteria through a direct interaction mechanism, resulting in thrombosis, and/or thrombocytopenia [119]. Thrombosis probably occurs when platelets and pathogen interact in a localized environment. Such is the case with infective endocarditis, where a bacteria-platelet thrombus occurs on the surface of a damaged cardiac valve. However, in the case of disseminated intravascular coagulation (DIC), the platelet activation occurs throughout the circulation. This results in clearance of the activated platelets. We have previously shown that Streptococcus sanguinis interacts with platelet GPIbα via HSA on its surface [120]. This interaction is sufficient to induce platelet activation and aggregation. Many bacteria bind a plasma protein, and then interact with platelets. We have previously shown that clumping factor A (Clf A) [121,122], and fibronectin-binding protein (FnBP) [123], induce platelet aggregation by binding either fibrinogen or fibronectin, which in turn, interacts with GPIIb/IIIa and Helicobacter pylori binds von Willebrand factor, which in turn interacts with GPIbα on the platelet surface [124]. Complement assembly on the bacterial surface interacts with a complement receptor on the platelet surface, leading to platelet activation [125-127].
Much less is known about the interaction of viruses with platelets, although a number of receptors known to be present on platelets have been shown to bind to different viruses. These include β3 integrins (Hantavirus [128]), α5β1 (Adenovirus [129]), Clec-2 (HIV [130]), α2β1 (Rotavirus [131]), and DC-SIGN (HIV [130], DENV [132], arenaviruses [133], Ebola virus [134,135], and Lentiviruses [136]). Axl and Tyro3 are Ebola virus [135,137], and LASV receptors [133]. Ebola virus requires the cholesterol transporter Niemann-Pick C1 (NPC1) for cell entry [138] [Figure 1].
Platelet activation by indirect interactions with microbes: Indirect platelet-virus interactions have not been widely described so far. However, there is evidence of pathogens that can indirectly activate platelets, primarily through their interaction with immune cells. One example of this is seen with lipopolysaccharide (LPS) from Gramnegative bacteria. LPS can stimulate platelet activation; however, it would appear that this is primarily due to activation of neutrophils, and subsequent platelet binding [103]. The other possibility is that activation of the immune system leads to increased cytokine levels, which may activate platelets. Certainly platelet dysfunction is also associated with SIRS [88,89]. Hemorrhagic fever viruses not only interact with platelets, but can also infect megakaryocytes. This is the case for some strains of Hantavirus associated with hemorrhagic fever, where megakaryocyte infection is thought to induce their destruction by T-cells [139]. Similarly, cytomegalovirus infection of megakaryocytes triggers caspase-mediated apoptosis resulting in thrombocytopenia [140]. Recent studies suggest that DENV also infects megakaryocytes, and inhibits megakaryopoiesis by inducing apoptosis in vitro [141,142].
Platelet FcγRIIa: The role of platelets in DHF/DSS is unclear. Certainly a severe thrombocytopenia occurs and is implicated in poor prognosis, however, is this secondary to DHF/DSS, or is it a key player in the pathogenesis? Anti-platelet antibodies or platelet-associated antibodies have been detected in some patients [108,111,143-145], and this may be due to molecular mimicry between a platelet antigen and a DENV antigen [144], and molecular mimicry is known to occur with other microorganisms [146-148]. However, this does not explain why a repeat infection is required for thrombocytopenia, as the platelet antigen should drive the immune response. One possible explanation is that anti-viral antibodies cross-react with an activation-dependent antigen. This is not unusual, and is the mechanism involved in heparininduced thrombocytopenia [98]. Heparin mildly activates platelets, causing them to express platelet factor 4 (PF4) on their surface, which is not found on the surface of a resting platelet. Antibodies are directed against the PF4-heparin complex on the platelet surface, thereby bringing them into contact with platelet FcγRIIa. Thus, virus may partially activate platelets, causing increased binding to FcγRIIa, and generating thrombocytopenia. In the absence of virus, there is no platelet activation, and no thrombocytopenia. There is some evidence to suggest that this may be the case, as it has previously been shown that DENV is capable of binding to platelets, and that antibody can exacerbate the subsequent thrombocytopenia [108].
Another possibility is the role of antibody-dependent enhancement [43]. This describes a process where non-neutralizing anti-Dengue antibodies [40,149] bind to the virus particle, and can interact with Fc receptors on cells aiding the infection process, a process that can also involve complement generation [47,150]. This is a process that is mechanistically similar to bacterial-induced platelet activation [119], where bacteria-bound, non-neutralizing antibodies can interact with platelet FcγRIIa receptor [123,125,126,151,152]. We have shown that complement plays a role in platelet activation as well [125-127]. Several publications point to similar pathogen-platelet interactions, with regard to Dengue virus. Wang et al. [153] showed that antibody-bound DENV-2 virus can bind to platelets, and was not inhibited by the anti- FcγRIIa antibody IV.3. This, however, is not unusual as IV.3 inhibits S. sanguinis-induced platelet aggregation, but not S. sanguinis mediated adhesion to platelets [152]. The reason for this is that the interaction with FcγRIIa is a weak interaction and cannot support adhesion, which is usually mediated by a specific adhesion molecule. With bacteria, at least, the effect of the FcγRIIa interaction is to trigger platelet activation and usually that occurs in the presence of a secondary adhesive interaction [119]. While these studies suggest that a direct virus-platelet interaction may take place, and that this can be enhanced by antibody, neither the platelet nor virus antigens involved have been identified nor has the role for antibody been clarified.
Targeting the platelet in VHF
While vaccination is potentially beneficial in preventing people from developing VHF, as shown by YF17D and Candid#1 vaccination, there are no approved vaccines for other VHF. Thus, treating the patient with VHF is the only possible option at present. There are two potential strategies for treating VHF. The first is to inhibit viral replication, and the second is to inhibit the host response to infection. Currently, no drugs have been approved to inhibit viral replication in VHF. However, there is some evidence to suggest that ribavirin may be beneficial in some types of VHF, although its mechanism of action is unknown [154,67]. The second approach is to prevent development of the hemorrhagic fever. This could potentially be achieved with conventional anti-platelet agents. For instance, there is evidence that aspirin may be beneficial in sepsis [155,156], and other critically ill patients associated with SIRS [115], and this is also true for clopidogrel [157]. There is a paucity of studies of the use of anti-platelet agents in VHF, although a 1979 paper found that while the combination of aspirin and dipyridamole did not affect mortality, it did reduce the progression to DHF [158].
The biggest concern with using conventional anti-platelet agents is that they preserve platelet number, but at the expense of inhibiting platelet function. In the case of patients with hemorrhagic fever, this may increase the risk of bleeding. To make matters worse, many are irreversible inhibitors, and thus, if the patient develops a bleeding problem, there is little that can be done. The obvious anti-platelet agent to use is a P2Y12 antagonist, as it is effective at inhibiting platelet activation by many different agents. Recently, reversible P2Y12 antagonists, such as cangrelor have been developed [159]. While these agents will completely inhibit platelet function, their effects can be rapidly reversed in the case of bleeding complications.
A more effective strategy would be to prevent the interaction of the viruses with platelet in the first place. There are a relatively small number of receptors on the platelet surface that could interact with viruses, and most of these have been the subjects of drug discovery programmes [160]. While few have been approved, this is primarily due to the lack of a suitable indication, rather than failure to discover inhibitors. The dominant receptor is GPIIb/IIIa, the fibrinogen receptor, and there are already 3 approved inhibitors of this receptor [161]. The next most abundant receptor is GPIbα, the von Willebrand receptor, and inhibitors have been discovered and used in animal models of thrombosis [162]. Other receptors include the collagen receptors α2β1 and GPVI [163], and PAR-1, the thrombin receptor [164]. As discussed above, FcRIIa is an important receptor for the interaction of DENV with platelets, and small molecule inhibitors of this receptor have been discovered [165]. Selectively inhibiting the interaction of viruses with platelets has the advantage of preventing virus-mediated platelet activation and subsequent thrombocytopenia, while preserving platelet function.
Conclusion
In conclusion, a detailed description of the hemorrhagic fever virus-platelet interaction, and the role of platelets in VHF pathogenesis is important not only for unraveling the mysteries of this group of diseases, but also for developing therapies that prevent the appearance of hemorrhagic signs.
References
- Gowen BB, Holbrook MR (2008) Animal models of highly pathogenic RNA viral infections: hemorrhagic fever viruses. Antiviral Res 78: 79-90.
- Zapata JC, Pauza CD, Djavani MM, Rodas JD, Moshkoff D, et al. (2011) Lymphocytic choriomeningitis virus (LCMV) infection of macaques: a model for Lassa fever. Antiviral Res 92: 125-138.
- Peters CJ, Zaki SR (2002) Role of the endothelium in viral hemorrhagic fevers. Crit Care Med 30: S268-S273.
- Leyssen P, De Clercq E, Neyts J (2000) Perspectives for the treatment of infections with Flaviviridae. Clin Microbiol Rev 13: 67-82.
- Streit JA, Yang M, Cavanaugh JE, Polgreen PM (2011) Upward trend in dengue incidence among hospitalized patients, United States. Emerg Infect Dis 17: 914-916.
- Moi ML, Lim CK, Kotaki A, Takasaki T, Kurane I (2010) Development of an antibody-dependent enhancement assay for dengue virus using stable BHK-21 cell lines expressing Fc gammaRIIA. J Virol Methods 163: 205-209.
- WHO (2006) Scientific working group report on dengue. WHO, Geneva, Switzerland.
- WHO Library Cataloguing-in-Publication Data (2009) Dengue-Guidelines for diagnosis, treatment, prevention and control. World Health Organization, Geneva, Switzerland.
- Gibbons RV, Vaughn DW (2002) Dengue: an escalating problem. BMJ 324: 1563-1566.
- Guzman MG, Kouri G (2003) Dengue and dengue hemorrhagic fever in the Americas: lessons and challenges. J Clin Virol 27: 1-13.
- Simmons CP, Farrar JJ, Nguyen vV, Wills B (2012) Dengue. N Engl J Med 366: 1423-1432.
- WHO. Yellow Fever.
- Monath TP (2001) Yellow fever: an update. Lancet Infect Dis 1: 11-20.
- Ružek D, Yakimenko VV, Karan LS, Tkachev SE (2010) Omsk haemorrhagic fever. Lancet 376: 2104-2113.
- CDC (2012) Kyasanur Forest Disease. CDC, USA.
- Westaway EG, Blok J (1997) Taxonomy and evolutionary relationships of flaviviruses. In: Gubler DJ, Kuno G (Eds.), Dengue and Dengue Hemorrhagic Fever. CAB International, Cambridge, UK 147–173.
- Kuhn RJ, Zhang W, Rossmann MG, Pletnev SV, Corver J, et al. (2002) Structure of dengue virus: implications for flavivirus organization, maturation, and fusion. Cell 108: 717-725.
- Krishnan MN, Sukumaran B, Pal U, Agaisse H, Murray JL, et al. (2007) Rab 5 is required for the cellular entry of dengue and West Nile viruses. J Virol 81: 4881-4885.
- van der Schaar HM, Rust MJ, Waarts BL, van der Ende-Metselaar H, Kuhn RJ, et al. (2007) Characterization of the early events in dengue virus cell entry by biochemical assays and single-virus tracking. J Virol 81: 12019-12028.
- Modis Y, Ogata S, Clements D, Harrison SC (2004) Structure of the dengue virus envelope protein after membrane fusion. Nature 427: 313-319.
- Mukhopadhyay S, Kuhn RJ, Rossmann MG (2005) A structural perspective of the flavivirus life cycle. Nat Rev Microbiol 3: 13-22.
- Henchal EA, Putnak JR (1990) The dengue viruses. Clin Microbiol Rev 3: 376-396.
- Perera R, Kuhn RJ (2008) Structural proteomics of dengue virus. Curr Opin Microbiol 11: 369-377.
- Li L, Lok SM, Yu IM, Zhang Y, Kuhn RJ, et al. (2008) The flavivirus precursor membrane-envelope protein complex: structure and maturation. Science 319: 1830-1834.
- Yu IM, Holdaway HA, Chipman PR, Kuhn RJ, Rossmann MG, et al. (2009) Association of the pr peptides with dengue virus at acidic pH blocks membrane fusion. J Virol 83: 12101-12107.
- Rodenhuis-Zybert IA, van der Schaar HM, da Silva Voorham JM, van der Ende-Metselaar H, Lei HY, et al. (2010) Immature dengue virus: a veiled pathogen? PLoS Pathog 6: e1000718.
- Ferlenghi I, Clarke M, Ruttan T, Allison SL, Schalich J, et al. (2001) Molecular organization of a recombinant subviral particle from tick-borne encephalitis virus. Mol Cell 7: 593-602.
- Kuwahara M, Konishi E (2010) Evaluation of extracellular subviral particles of dengue virus type 2 and Japanese encephalitis virus produced by Spodoptera frugiperda cells for use as vaccine and diagnostic antigens. Clin Vaccine Immunol 17: 1560-1566.
- Junjhon J, Lausumpao M, Supasa S, Noisakran S, Songjaeng A, et al. (2008) Differential modulation of prM cleavage, extracellular particle distribution, and virus infectivity by conserved residues at nonfurin consensus positions of the dengue virus pr-M junction. J Virol 82: 10776-10791.
- Flamand M, Megret F, Mathieu M, Lepault J, Rey FA, et al. (1999) Dengue virus type 1 nonstructural glycoprotein NS1 is secreted from mammalian cells as a soluble hexamer in a glycosylation-dependent fashion. J Virol 73: 6104-6110.
- Chung KM, Liszewski MK, Nybakken G, Davis AE, Townsend RR, et al. (2006) West Nile virus nonstructural protein NS1 inhibits complement activation by binding the regulatory protein factor H. Proc Natl Acad Sci U S A 103: 19111-19116.
- Nimmannitya S (2002) Dengue hemorrhagic fever: current issues and future research. Asian-Oceanian J Pediar Child Health 1: 1-20.
- Guilarde AO, Turchi MD, Siqueira JB Jr, Feres VC, Rocha B, et al. (2008) Dengue and dengue hemorrhagic fever among adults: clinical outcomes related to viremia, serotypes, and antibody response. J Infect Dis 197: 817-824.
- Kittigul L, Pitakarnjanakul P, Sujirarat D, Siripanichgon K (2007) The differences of clinical manifestations and laboratory findings in children and adults with dengue virus infection. J Clin Virol 39: 76-81.
- Srichaikul T, Nimmannitya S (2000) Haematology in dengue and dengue haemorrhagic fever. Baillieres Best Pract Res Clin Haematol 13: 261-276.
- Kalayanarooj S, Vaughn DW, Nimmannitya S, Green S, Suntayakorn S, et al. (1997) Early clinical and laboratory indicators of acute dengue illness. J Infect Dis 176: 313-321.
- Bhamarapravati N (1989) Hemostatic defects in dengue hemorrhagic fever. Rev Infect Dis 11: S826-S829.
- Hathirat P, Isarangkura P, Srichaikul T, Suvatte V, Mitrakul C (1993) Abnormal hemostasis in dengue hemorrhagic fever. Southeast Asian J Trop Med Public Health 24: 80-85.
- Chua MN, Molanida R, de Guzman M, Laberiza F (1993) Prothrombin time and partial thromboplastin time as a predictor of bleeding in patients with dengue hemorrhagic fever. Southeast Asian J Trop Med Public Health 24: 141-143.
- Halstead SB, O'Rourke EJ (1977) Dengue viruses and mononuclear phagocytes. I. Infection enhancement by non-neutralizing antibody. J Exp Med 146: 201-217.
- Alvarez M, Rodriguez-Roche R, Bernardo L, Vázquez S, Morier L, et al. (2006) Dengue hemorrhagic Fever caused by sequential dengue 1-3 virus infections over a long time interval: Havana epidemic, 2001-2002. Am J Trop Med Hyg 75: 1113-1117.
- Schmidt AC (2010) Response to dengue fever--the good, the bad, and the ugly? N Engl J Med 363: 484-487.
- Cummings DA, Schwartz IB, Billings L, Shaw LB, Burke DS (2005) Dynamic effects of antibody-dependent enhancement on the fitness of viruses. Proc Natl Acad Sci U S A 102: 15259-15264.
- Goncalvez AP, Engle RE, St Claire M, Purcell RH, Lai CJ (2007) Monoclonal antibody-mediated enhancement of dengue virus infection in vitro and in vivo and strategies for prevention. Proc Natl Acad Sci U S A 104: 9422-9427.
- Moi ML, Lim CK, Takasaki T, Kurane I (2010) Involvement of the Fc gamma receptor IIA cytoplasmic domain in antibody-dependent enhancement of dengue virus infection. J Gen Virol 91: 103-111.
- Mady BJ, Erbe DV, Kurane I, Fanger MW, Ennis FA (1991) Antibody-dependent enhancement of dengue virus infection mediated by bispecific antibodies against cell surface molecules other than Fc gamma receptors. J Immunol 147: 3139-3144.
- Mehlhop E, Ansarah-Sobrinho C, Johnson S, Engle M, Fremont DH, et al. (2007) Complement protein C1q inhibits antibody-dependent enhancement of flavivirus infection in an IgG subclass-specific manner. Cell Host Microbe 2: 417-426.
- Yamanaka A, Kosugi S, Konishi E (2008) Infection-enhancing and -neutralizing activities of mouse monoclonal antibodies against dengue type 2 and 4 viruses are controlled by complement levels. J Virol 82: 927-937.
- Chareonsirisuthigul T, Kalayanarooj S, Ubol S (2007) Dengue virus (DENV) antibody-dependent enhancement of infection upregulates the production of anti-inflammatory cytokines, but suppresses anti-DENV free radical and pro-inflammatory cytokine production, in THP-1 cells. J Gen Virol 88: 365-375.
- de la C Sierra B, Kourí G, Guzmán MG (2007) Race: a risk factor for dengue hemorrhagic fever. Arch Virol 152: 533-542.
- Guzman MG, Kouri G (2008) Dengue haemorrhagic fever integral hypothesis: confirming observations, 1987-2007. Trans R Soc Trop Med Hyg 102: 522-523.
- Blanton RE, Silva LK, Morato VG, Parrado AR, Dias JP, et al. (2008) Genetic ancestry and income are associated with dengue hemorrhagic fever in a highly admixed population. Eur J Hum Genet 16: 762-765.
- Shekhar KC, Huat OL (1992) Epidemiology of dengue/dengue hemorrhagic fever in Malaysia--a retrospective epidemiological study 1973-1987. Part I: Dengue hemorrhagic fever (DHF). Asia Pac J Public Health 6: 15-25.
- Shekhar KC, Huat OL (1992) Epidemiology of dengue/dengue hemorrhagic fever in Malaysia--a retrospective epidemiological study. 1973-1987. Part II: Dengue fever (DF). Asia Pac J Public Health 6: 126-133.
- Coffey LL, Mertens E, Brehin AC, Fernandez-Garcia MD, Amara A, et al. (2009) Human genetic determinants of dengue virus susceptibility. Microbes Infect 11: 143-156.
- Borio L, Inglesby T, Peters CJ, Schmaljohn AL, Hughes JM, et al. (2002) Hemorrhagic fever viruses as biological weapons: medical and public health management. JAMA 287: 2391-2405.
- Sanchez A, Lukwiya M, Bausch D, Mahanty S, Sanchez AJ, et al. (2004) Analysis of human peripheral blood samples from fatal and nonfatal cases of Ebola (Sudan) hemorrhagic fever: cellular responses, virus load, and nitric oxide levels. J Virol 78: 10370-10377.
- Fisher-Hoch SP, Platt GS, Lloyd G, Simpson DI, Neild GH, et al. (1983) Haematological and biochemical monitoring of Ebola infection in rhesus monkeys: implications for patient management. Lancet 2: 1055-1058.
- Fisher-Hoch SP, Platt GS, Neild GH, Southee T, Baskerville A, et al. (1985) Pathophysiology of shock and hemorrhage in a fulminating viral infection (Ebola). J Infect Dis 152: 887-894.
- Zampieri CA, Sullivan NJ, Nabel GJ (2007) Immunopathology of highly virulent pathogens: Insights from Ebola virus. Nat Immunol 8: 1159-1164.
- Iannacone M, Sitia G, Isogawa M, Whitmire JK, Marchese P, et al. (2008) Platelets prevent IFN-alpha/beta-induced lethal hemorrhage promoting CTL-dependent clearance of lymphocytic choriomeningitis virus. Proc Natl Acad Sci U S A 105: 629-634.
- Villinger F, Rollin PE, Brar SS, Chikkala NF, Winter J, et al. (1999) Markedly elevated levels of interferon (IFN)-gamma, IFN-alpha, interleukin (IL)-2, IL-10, and tumor necrosis factor-alpha associated with fatal Ebola virus infection. J Infect Dis 179: S188-S191.
- Ergönül O (2006) Crimean-Congo haemorrhagic fever. Lancet Infect Dis 6: 203-214.
- Cevik MA, Erbay A, Bodur H, Gülderen E, Bastug A, et al. (2008) Clinical and laboratory features of Crimean-Congo hemorrhagic fever: predictors of fatality. Int J Infect Dis 12: 374-379.
- Swanepoel R, Gill DE, Shepherd AJ, Leman PA, Mynhardt JH, et al. (1989) The clinical pathology of Crimean-Congo hemorrhagic fever. Rev Infect Dis 11: S794-S800.
- Carter SD, Surtees R, Walter CT, Ariza A, Bergeron É, et al. (2012) Structure, function, and evolution of the Crimean-Congo hemorrhagic fever virus nucleocapsid protein. J Virol 86: 10914-10923.
- Bossi P, Tegnell A, Baka A, Van Loock F, Hendriks J, et al. (2004) Bichat guidelines for the clinical management of haemorrhagic fever viruses and bioterrorism-related haemorrhagic fever viruses. Euro Surveill 9.
- Johnson K (1998) Introduction. In: Lee H, Calisher C, Schmaljohn C (Eds.), Manual of hemorrhagic fever with renal syndrome and hantavirus pulmonary syndrome, WHO Collaborating Center for Virus Reference and Research (Hantaviruses) 1-6.
- Jin C, Liang M, Ning J, Gu W, Jiang H, et al. (2012) Pathogenesis of emerging severe fever with thrombocytopenia syndrome virus in C57/BL6 mouse model. Proc Natl Acad Sci U S A 109: 10053-10058.
- Cummins D (1991) Arenaviral haemorrhagic fevers. Blood Rev 5: 129-137.
- Cummins D, Fisher-Hoch SP, Walshe KJ, Mackie IJ, McCormick JB, et al. (1989) A plasma inhibitor of platelet aggregation in patients with Lassa fever. Br J Haematol 72: 543-548
- Cummins D, Molinas FC, Lerer G, Maiztegui JI, Faint R, et al. (1990) A plasma inhibitor of platelet aggregation in patients with Argentine hemorrhagic fever. Am J Trop Med Hyg 42: 470-475.
- Lang PA, Contaldo C, Georgiev P, El-Badry AM, Recher M, et al. (2008) Aggravation of viral hepatitis by platelet-derived serotonin. Nat Med 14: 756-761.
- Pozner RG, Ure AE, Jaquenod de Giusti C, D'Atri LP, Italiano JE, et al. (2010) Junin virus infection of human hematopoietic progenitors impairs in vitro proplatelet formation and platelet release via a bystander effect involving type I IFN signaling. PLoS Pathog 6: e1000847.
- Hartwig JH (2002) Platelet structure. In: Michelson AD (Ed.) Platelets, Academic Press, USA.
- Weyrich AS, Schwertz H, Kraiss LW, Zimmerman GA (2009) Protein synthesis by platelets: historical and new perspectives. J Thromb Haemost 7: 241-246.
- Reed G (2002) Platelet secretion. In: Michelson AD (Ed.) Platelets: Academic Press, USA.
- Furie B, Furie BC (2005) Thrombus formation in vivo. J Clin Invest 115: 3355-3362.
- Shattil SJ, Newman PJ (2004) Integrins: dynamic scaffolds for adhesion and signaling in platelets. Blood 104: 1606-1615.
- Cox D, Kerrigan SW, Watson SP (2011) Platelets and the innate immune system: mechanisms of bacterial-induced platelet activation. J Thromb Haemost 9: 1097-1107.
- Li Z, Yang F, Dunn S, Gross AK, Smyth SS (2011) Platelets as immune mediators: their role in host defense responses and sepsis. Thromb Res 127: 184-188.
- Yeaman MR (2010) Platelets in defense against bacterial pathogens. Cell Mol Life Sci 67: 525-544.
- Yeaman MR (2010) Bacterial-platelet interactions: virulence meets host defense. Future Microbiol 5: 471-506.
- Bayer AS, Cheng D, Yeaman MR, Corey GR, McClelland RS, et al. (1998) In vitro resistance to thrombin-induced platelet microbicidal protein among clinical bacteremic isolates of Staphylococcus aureus correlates with an endovascular infectious source. Antimicrob Agents Chemother 42: 3169-3172.
- Fowler VG Jr, McIntyre LM, Yeaman MR, Peterson GE, Barth Reller L, et al. (2000) In vitro resistance to thrombin-induced platelet microbicidal protein in isolates of Staphylococcus aureus from endocarditis patients correlates with an intravascular device source. J Infect Dis 182: 1251-1254.
- Fowler VG Jr, Sakoulas G, McIntyre LM, Meka VG, Arbeit RD, et al. (2004) Persistent bacteremia due to methicillin-resistant Staphylococcus aureus infection is associated with agr dysfunction and low-level in vitro resistance to thrombin-induced platelet microbicidal protein. J Infect Dis 190: 1140-1149.
- Dhawan VK, Yeaman MR, Cheung AL, Kim E, Sullam PM, et al. (1997) Phenotypic resistance to thrombin-induced platelet microbicidal protein in vitro is correlated with enhanced virulence in experimental endocarditis due to Staphylococcus aureus. Infect Immun 65: 3293-3299.
- Iba T, Gando S, Murata A, Kushimoto S, Saitoh D, et al. (2007) Predicting the severity of systemic inflammatory response syndrome (SIRS)-associated coagulopathy with hemostatic molecular markers and vascular endothelial injury markers. J Trauma 63: 1093-1098.
- Ogura H, Gando S, Iba T, Eguchi Y, Ohtomo Y, et al. (2007) SIRS-associated coagulopathy and organ dysfunction in critically ill patients with thrombocytopenia. Shock 28: 411-417.
- Alt E, Amann-Vesti BR, Madl C, Funk G, Koppensteiner R (2004) Platelet aggregation and blood rheology in severe sepsis/septic shock: relation to the Sepsis-related Organ Failure Assessment (SOFA) score. Clin Hemorheol Microcirc 30: 107-115.
- Gawaz M, Dickfeld T, Bogner C, Fateh-Moghadam S, Neumann FJ (1997) Platelet function in septic multiple organ dysfunction syndrome. Intensive Care Med 23: 379-385.
- Larsson A, Eriksson M, Lundahl T, Lundkvist K, Lindahl T (1998) Impaired platelet function in endotoxemic pigs analyzed by flow cytometry. Platelets 9: 115-119.
- Singer G, Urakami H, Specian RD, Stokes KY, Granger DN (2006) Platelet recruitment in the murine hepatic microvasculature during experimental sepsis: role of neutrophils. Microcirculation 13: 89-97.
- Yaguchi A, Lobo FL, Vincent JL, Pradier O (2004) Platelet function in sepsis. J Thromb Haemost 2: 2096-2102.
- Camerer E, Cornelissen I, Kataoka H, Duong DN, Zheng YW, et al. (2006) Roles of protease-activated receptors in a mouse model of endotoxemia. Blood 107: 3912-3921.
- Taylor SM, Reilly MP, Schreiber AD, Chien P, Tuckosh JR, et al. (2000) Thrombosis and shock induced by activating antiplatelet antibodies in human Fc gamma RIIA transgenic mice: the interplay among antibody, spleen, and Fc receptor. Blood 96: 4254-4260.
- Shenkman B, Rubinstein E, Dardik R, Tamarin I, Savion N, et al. (1999) Activated platelets mediate Staphylococcus aureus deposition on subendothelial extracellular matrix: the role of glycoprotein Ib. Platelets 10: 36-44.
- Warkentin TE, Aird WC, Rand JH (2003) Platelet-endothelial interactions: sepsis, HIT, and antiphospholipid syndrome. Hematology Am Soc Hematol Educ Program 497-519.
- Kasperska-Zajaç A, Rogala B (2006) Platelet function in anaphylaxis. J Investig Allergol Clin Immunol 16: 1-4.
- Ohba M, Shibazaki M, Sasano T, Inoue M, Takada H, et al. (2004) Platelet responses and anaphylaxis-like shock induced in mice by intravenous injection of whole cells of oral streptococci. Oral Microbiol Immunol 19: 26-30.
- Sharma B, Sharma M, Majumder M, Steier W, Sangal A, et al. (2007) Thrombocytopenia in septic shock patients--a prospective observational study of incidence, risk factors and correlation with clinical outcome. Anaesth Intensive Care 35: 874-880.
- Tweardy DJ, Khoshnevis MR, Yu B, Mastrangelo MA, Hardison EG, et al. (2006) Essential role for platelets in organ injury and inflammation in resuscitated hemorrhagic shock. Shock 26: 386-390.
- Clark SR, Ma AC, Tavener SA, McDonald B, Goodarzi Z, et al. (2007) Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood. Nat Med 13: 463-469.
- Lambert MP, Wang Y, Bdeir KH, Nguyen Y, Kowalska MA, et al. (2009) Platelet factor 4 regulates megakaryopoiesis through low-density lipoprotein receptor-related protein 1 (LRP1) on megakaryocytes. Blood 114: 2290-2298.
- Johnson JR, Johnston B, Clabots C, Kuskowski MA, Castanheira M (2010) Escherichia coli sequence type ST131 as the major cause of serious multidrug-resistant E. coli infections in the United States. Clin Infect Dis 51: 286-294.
- Lei HY, Yeh TM, Liu HS, Lin YS, Chen SH, et al. (2001) Immunopathogenesis of dengue virus infection. J Biomed Sci 8: 377-388.
- Rahbar A, Söderberg-Nauclér C (2005) Human cytomegalovirus infection of endothelial cells triggers platelet adhesion and aggregation. J Virol 79: 2211-2220.
- Funahara Y, Ogawa K, Fujita N, Okuno Y (1987) Three possible triggers to induce thrombocytopenia in dengue virus infection. Southeast Asian J Trop Med Public Health 18: 351-355.
- Kulkarni S, Woollard KJ, Thomas S, Oxley D, Jackson SP (2007) Conversion of platelets from a proaggregatory to a proinflammatory adhesive phenotype: role of PAF in spatially regulating neutrophil adhesion and spreading. Blood 110: 1879-1886.
- Sahni SK (2007) Endothelial cell infection and hemostasis. Thromb Res 119: 531-549.
- Ahmed S, Siddiqui RK, Siddiqui AK, Zaidi SA, Cervia J (2002) HIV associated thrombotic microangiopathy. Postgrad Med J 78: 520-525.
- Krishnamurti C, Peat RA, Cutting MA, Rothwell SW (2002) Platelet adhesion to dengue-2 virus-infected endothelial cells. Am J Trop Med Hyg 66: 435-441.
- Dole VS, Bergmeier W, Mitchell HA, Eichenberger SC, Wagner DD (2005) Activated platelets induce Weibel-Palade-body secretion and leukocyte rolling in vivo: role of P-selectin. Blood 106: 2334-2339.
- Otto GP, Sossdorf M, Boettel J, Kabisch B, Breuel H, et al. (2012) Effects of low-dose acetylsalicylic acid and atherosclerotic vascular diseases on the outcome in patients with severe sepsis or septic shock. Platelets.
- Lösche W, Boettel J, Kabisch B, Winning J, Claus RA, et al. (2012) Do aspirin and other antiplatelet drugs reduce the mortality in critically ill patients? Thrombosis 2012: 720254.
- Levi M, Toh CH, Thachil J, Watson HG (2009) Guidelines for the diagnosis and management of disseminated intravascular coagulation. British Committee for Standards in Haematology. Br J Haematol 145: 24-33.
- Mann KG, Nesheim ME, Church WR, Haley P, Krishnaswamy S (1990) Surface-dependent reactions of the vitamin K-dependent enzyme complexes. Blood 76: 1-16.
- Loscalzo J, Pasche B, Ouimet H, Freedman JE (1995) Platelets and plasminogen activation. Thromb Haemost 74: 291-293.
- Fitzgerald JR, Foster TJ, Cox D (2006) The interaction of bacterial pathogens with platelets. Nat Rev Microbiol 4: 445-457.
- Plummer C, Wu H, Kerrigan SW, Meade G, Cox D, et al. (2005) A serine-rich glycoprotein of Streptococcus sanguis mediates adhesion to platelets via GPIb. Br J Haematol 129: 101-109.
- O'Brien L, Kerrigan SW, Kaw G, Hogan M, Penadés J, et al. (2002) Multiple mechanisms for the activation of human platelet aggregation by Staphylococcus aureus: roles for the clumping factors ClfA and ClfB, the serine-aspartate repeat protein SdrE and protein A. Mol Microbiol 44: 1033-1044.
- Kerrigan SW, Clarke N, Loughman A, Meade G, Foster TJ, et al. (2008) Molecular basis for Staphylococcus aureus-mediated platelet aggregate formation under arterial shear in vitro. Arterioscler Thromb Vasc Biol 28: 335-340.
- Fitzgerald JR, Loughman A, Keane F, Brennan M, Knobel M, et al. (2006) Fibronectin-binding proteins of Staphylococcus aureus mediate activation of human platelets via fibrinogen and fibronectin bridges to integrin GPIIb/IIIa and IgG binding to the FcgRIIa receptor. Mol Microbiol 59: 212-230.
- Byrne MF, Murphy JF, Corcoran PA, Atherton JC, Sheehan KM, et al. (2003) Helicobacter pylori induces cyclooxygenase-1 and cyclooxygenase-2 expression in vascular endothelial cells. Scand J Gastroenterol 38: 1023-1030.
- Ford I, Douglas CW, Cox D, Rees DG, Heath J, et al. (1997) The role of immunoglobulin G and fibrinogen in platelet aggregation by Streptococcus sanguis. Br J Haematol 97: 737-746.
- Loughman A, Fitzgerald JR, Brennan MP, Higgins J, Downer R, et al. (2005) Roles for fibrinogen, immunoglobulin and complement in platelet activation promoted by Staphylococcus aureus clumping factor A. Mol Microbiol 57: 804-818.
- Miajlovic H, Loughman A, Brennan M, Cox D, Foster TJ (2007) Both complement- and fibrinogen-dependent mechanisms contribute to platelet aggregation mediated by Staphylococcus aureus clumping factor B. Infect Immun 75: 3335-3343.
- Gavrilovskaya IN, Brown EJ, Ginsberg MH, Mackow ER (1999) Cellular entry of hantaviruses which cause hemorrhagic fever with renal syndrome is mediated by beta3 integrins. J Virol 73: 3951-3959.
- Asokan A, Hamra JB, Govindasamy L, Agbandje-McKenna M, Samulski RJ (2006) Adeno-associated virus type 2 contains an integrin alpha5beta1 binding domain essential for viral cell entry. J Virol 80: 8961-8969.
- Chaipan C, Soilleux EJ, Simpson P, Hofmann H, Gramberg T, et al. (2006) DC-SIGN and CLEC-2 mediate human immunodeficiency virus type 1 capture by platelets. J Virol 80: 8951-8960.
- Fleming FE, Graham KL, Takada Y, Coulson BS (2011) Determinants of the specificity of rotavirus interactions with the alpha2beta1 integrin. J Biol Chem 286: 6165-6174.
- Boonnak K, Slike BM, Burgess TH, Mason RM, Wu SJ, et al. (2008) Role of dendritic cells in antibody-dependent enhancement of dengue virus infection. J Virol 82: 3939-3951.
- Shimojima M, Ströher U, Ebihara H, Feldmann H, Kawaoka Y (2012) Identification of cell surface molecules involved in dystroglycan-independent Lassa virus cell entry. J Virol 86: 2067-2078.
- Alvarez CP, Lasala F, Carrillo J, Muñiz O, Corbí AL, et al. (2002) C-type lectins DC-SIGN and L-SIGN mediate cellular entry by Ebola virus in cis and in trans. J Virol 76: 6841-6844.
- Shimojima M, Ikeda Y, Kawaoka Y (2007) The mechanism of Axl-mediated Ebola virus infection. J Infect Dis 196: S259-S263.
- Flaujac C, Boukour S, Cramer-Bordé E (2010) Platelets and viruses: an ambivalent relationship. Cell Mol Life Sci 67: 545-556.
- Hunt CL, Kolokoltsov AA, Davey RA, Maury W (2011) The Tyro3 receptor kinase Axl enhances macropinocytosis of Zaire ebolavirus. J Virol 85: 334-347.
- Carette JE, Raaben M, Wong AC, Herbert AS, Obernosterer G, et al. (2011) Ebola virus entry requires the cholesterol transporter Niemann-Pick C1. Nature 477: 340-343.
- Lütteke N, Raftery MJ, Lalwani P, Lee MH, Giese T, et al. (2010) Switch to high-level virus replication and HLA class I upregulation in differentiating megakaryocytic cells after infection with pathogenic hantavirus. Virology 405: 70-80.
- Dou J, Li X, Cai Y, Chen H, Zhu S, et al. (2010) Human cytomegalovirus induces caspase-dependent apoptosis of megakaryocytic CHRF-288-11 cells by activating the JNK pathway. Int J Hematol 91: 620-629.
- Basu A, Jain P, Gangodkar SV, Shetty S, Ghosh K, et al. (2008) Dengue 2 virus inhibits in vitro megakaryocytic colony formation and induces apoptosis in thrombopoietin-inducible megakaryocytic differentiation from cord blood CD34+ cells. FEMS Immunol Med Microbiol 53: 46-51.
- Noisakran S, Onlamoon N, Pattanapanyasat K, Hsiao HM, Songprakhon P, et al. (2012) Role of CD61+ cells in thrombocytopenia of dengue patients. Int J Hematol 96: 600-610.
- Oishi K, Inoue S, Cinco MT, Dimaano EM, Alera MT, et al. (2003) Correlation between increased platelet-associated IgG and thrombocytopenia in secondary dengue virus infections. J Med Virol 71: 259-264.
- Akkerhuis KM, van Den Brand MJ, van Der Zwaan C, Peels HO, Suryapranata H, et al. (2001) Pharmacodynamics and safety of lefradafiban, an oral platelet glycoprotein IIb/IIIa receptor antagonist, in patients with stable coronary artery disease undergoing elective angioplasty. Heart 85: 444-450.
- Anders R, Kleiman J, Nicholson N, Wazowicz B, Burns D (2001) Xemilofiban/orbofiban: insight into drug development. Cardiovasc Drug Rev 19: 116-132.
- Bachmaier K, Neu N, de la Maza LM, Pal S, Hessel A, et al. (1999) Chlamydia infections and heart disease linked through antigenic mimicry. Science 283: 1335-1339.
- Bachmaier K, Le J, Penninger JM (2000) "Catching heart disease": antigenic mimicry and bacterial infections. Nat Med 6: 841-842.
- Stanley RG, Ngaiza JR, Atieno E, Jell G, Francklow K, et al. (2003) Immune-dependent thrombocytopaenia in mice infected with Schistosoma mansoni. Parasitology 126: 225-229.
- Beltramello M, Williams KL, Simmons CP, Macagno A, Simonelli L, et al. (2010) The human immune response to dengue virus is dominated by highly cross-reactive antibodies endowed with neutralizing and enhancing activity. Cell Host Microbe 8: 271-283.
- Avirutnan P, Punyadee N, Noisakran S, Komoltri C, Thiemmeca S, et al. (2006) Vascular leakage in severe dengue virus infections: a potential role for the nonstructural viral protein NS1 and complement. J Infect Dis 193: 1078-1088.
- Byrne MF, Kerrigan SW, Corcoran PA, Atherton JC, Murray FE, et al. (2003) Helicobacter pylori binds von Willebrand factor and interacts with GPIb to induce platelet aggregation. Gastroenterology 124: 1846-1854.
- Kerrigan SW, Douglas I, Wray A, Heath J, Byrne MF, et al. (2002) A role for glycoprotein Ib in Streptococcus sanguis-induced platelet aggregation. Blood 100: 509-516.
- Wang S, He R, Patarapotikul J, Innis BL, Anderson R (1995) Antibody-enhanced binding of dengue-2 virus to human platelets. Virology 213: 254-257.
- Ergonul O (2012) Crimean-Congo hemorrhagic fever virus: new outbreaks, new discoveries. Curr Opin Virol 2: 215-220.
- Sossdorf M, Otto GP, Boettel J, Winning J, Lösche W (2013) Benefit of low-dose aspirin and non-steroidal anti-inflammatory drugs in septic patients. Crit Care 17: 402.
- Eisen DP, Reid D, McBryde ES (2012) Acetyl salicylic acid usage and mortality in critically ill patients with the systemic inflammatory response syndrome and sepsis. Crit Care Med 40: 1761-1767.
- Seidel M, Winning J, Claus RA, Bauer M, Lösche W (2009) Beneficial effect of clopidogrel in a mouse model of polymicrobial sepsis. J Thromb Haemost 7: 1030-1032.
- Kho LK, Wulur H, Himawan T (1979) Dipyridamole in the treatment of dengue haemorrhagic fever. Southeast Asian J Trop Med Public Health 10: 385-388.
- Bhatt DL, Stone GW, Mahaffey KW, Gibson CM, Steg PG, et al. (2013) Effect of platelet inhibition with cangrelor during PCI on ischemic events. N Engl J Med 368: 1303-1313.
- Siller-Matula JM, Krumphuber J, Jilma B (2010) Pharmacokinetic, pharmacodynamic and clinical profile of novel antiplatelet drugs targeting vascular diseases. Br J Pharmacol 159: 502-517.
- Cox D, Brennan M, Moran N (2010) Integrins as therapeutic targets: lessons and opportunities. Nat Rev Drug Discov 9: 804-820.
- Vanhoorelbeke K, Ulrichts H, Van de Walle G, Fontayne A, Deckmyn H (2007) Inhibition of platelet glycoprotein Ib and its antithrombotic potential. Curr Pharm Des 13: 2684-2697.
- Clemetson KJ, Clemetson JM (2007) Collagen receptors as potential targets for novel anti-platelet agents. Curr Pharm Des 13: 2673-2683.
- Wiisanen ME, Moliterno DJ (2012) Platelet protease-activated receptor antagonism in cardiovascular medicine. Coron Artery Dis 23: 375-379.
- Pietersz GA, Mottram PL, van de Velde NC, Sardjono CT, Esparon S, et al. (2009) Inhibition of destructive autoimmune arthritis in FcgammaRIIa transgenic mice by small chemical entities. Immunol Cell Biol 87: 3-12.
- Gavrilovskaya IN, Gorbunova EE, Mackow ER (2010) Pathogenic hantaviruses direct the adherence of quiescent platelets to infected endothelial cells. J Virol 84: 4832-4839.
- Lin CF, Lei HY, Liu CC, Liu HS, Yeh TM, et al. (2001) Generation of IgM anti-platelet autoantibody in dengue patients. J Med Virol 63: 143-149.
- Saito M, Oishi K, Inoue S, Dimaano EM, Alera MT, et al. (2004) Association of increased platelet-associated immunoglobulins with thrombocytopenia and the severity of disease in secondary dengue virus infections. Clin Exp Immunol 138: 299-303.
- Noisakran S, Onlamoon N, Hsiao HM, Clark KB, Villinger F, et al. (2012) Infection of bone marrow cells by dengue virus in vivo. Exp Hematol 40: 250-259.
- Gomez RM, Pozner RG, Lazzari MA, D'Atri LP, Negrotto S, et al. (2003) Endothelial cell function alteration after Junin virus infection. Thromb Haemost 90: 326-333.
Relevant Topics
- Anthrax Bioterrorism
- Bio surveilliance
- Biodefense
- Biohazards
- Biological Preparedness
- Biological Warfare
- Biological weapons
- Biorisk
- Bioterrorism
- Bioterrorism Agents
- Biothreat Agents
- Disease surveillance
- Emerging infectious disease
- Epidemiology of Breast Cancer
- Information Security
- Mass Prophylaxis
- Nuclear Terrorism
- Probabilistic risk assessment
- United States biological defense program
- Vaccines
Recommended Journals
Article Tools
Article Usage
- Total views: 22512
- [From(publication date):
specialissue-2013 - Dec 06, 2025] - Breakdown by view type
- HTML page views : 17630
- PDF downloads : 4882