Review Article Open Access
Virus-Like Particle-Based vaccines for Rift Valley Fever Virus
Robert B. Mandell and Ramon Flick*
BioProtection Systems, a subsidiary of NewLink Genetics Corporation, 2901 S. Loop Drive, Ames, IA 50010
- *Corresponding Author:
- Ramon Flick
BioProtection Systems,
a subsidiary of NewLink Genetics Corporation
2901 S. Loop Drive, Ames, IA 50010
Tel: 515-598-5017
E-mail: rflick@bpsys.net
Received Date: July 22, 2011; Accepted Date: September 29, 2011; Published Date: November 04, 2011
Citation: Mandell RB, Flick R (2011) Virus-Like Particle-Based Vaccines for Rift Valley Fever Virus. J Bioterr Biodef S1:008. doi: 10.4172/2157-2526.S1-008
Copyright: © 2011 Mandell RB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Bioterrorism & Biodefense
Abstract
There is a clear need for a vaccine to protect humans and livestock against the devastating consequences of Rift Valley fever (RVF) virus infections. Virus-like particles (VLPs), readily generated for many viruses by expression of their structural proteins, are a safe and immunogenic vaccine platform that has been approved for use as human vaccines. Pre-clinical studies have shown that RVF VLPs are highly immunogenic and efficacious in rodent models, and thus present a promising vaccine candidate for Rift Valley fever virus.
Introduction
Rift Valley fever virus (RVFV) causes a devastating zoonotic disease that can lead to high morbidity and mortality in both humans and livestock [1-6]. RVFV is recognized as a significant threat in places where it is endemic, and further research is required to understand the potential of this arbovirus (family Bunyaviridae, genus phlebovirus) to emerge in the future, invade new geographic areas such as the United States or Europe [7,8], and become critical public and veterinary health problems [9,10].
While some vaccines for RVFV are available on a limited basis, there is still no widely accepted safe and efficacious vaccine for protecting humans and livestock against the devastating consequences of this virus. Unfortunately, the development of RVFV vaccines has proven to be challenging, as detailed in several recent reviews [1,3-6,9].
Virus-like particles as a vaccine platform
Virus-like particles (VLPs) can be generated for many viruses and virus families by the expression of their structural proteins [11- 14]. VLPs can be generated in many different production systems including insect cells [15-19], mammalian cells [14,20], yeast [16,21], and plants [22,23]. VLPs have proven to be useful tools for the study of virus assembly and morphogenesis, genome packaging, budding, receptor analysis and virus entry [24-31]. VLPs can also allow for the study of high containment viruses under lower biosafety conditions [32-34]. VLPs present an important vaccine platform because they retain the antigenicity of virus particles without the risks associated with other types of antiviral vaccine platforms [11,35]. VLP-based vaccines are considered to be inherently safer than attenuated virus vaccines because they are usually free of viral genetic material and therefore are not encumbered by the possible safety-related drawbacks such as genomic reversion, recombination and reassortment [11,36]. VLPs can be inherently more immunogenic than purified proteins because a) the structural proteins are often displayed in a more authentic conformation [11-13] and b) they exhibit a morphology that is usually very similar to the wild type virus from which they are derived and mimic the functional interactions of the live virus with cellular receptors [13,14,37]. Additionally, VLPs have been shown to stimulate B cell-mediated and CD4 proliferative and cytotoxic T lymphocyte responses [38-40], and can overcome potential preexisting immunity and be re-used as an antigen carrier [41]. VLPs have been successfully employed as vehicles to display unrelated antigens and/or immune stimulators [42-46] and to deliver drugs and imaging reagents [47]. Recent developments in the VLP-based vaccine development field allow for large-scale and cost-effective manufacturing [48]. Finally, there are precedents for VLPs as efficacious vaccines, since Cervarix (GlaxoSmithKline) and Gardasil (Merck) vaccines for human papilloma virus and Recombivax HB (Merck) for hepatitis B have been approved for human use by the US Food and Drug Administration (FDA) [15,16,49-51]. Promising preclinical and clinical data have also been obtained for VLP-based vaccine candidates for emerging viral pathogens including Chikungunya virus [52], Ebola and Marburg viruses [53,54], H1N1 and H5N1 influenza viruses[22,55-59], Hantavirus [60], arena viruses [61,62] and Nipah virus [63,64]. VLPs also show promise as treatments of chronic diseases such as inflammatory hyperalgesia, hypertension, Alzheimer’s disease and certain antibody-mediated hypersensitivities [65-68]. Agricultural applications include promising vaccine candidates for livestock including bluetongue [11,69,70], Newcastle disease [44] and foot-andmouth viruses [71,72].
VLPs are ideally suited for the generation of vaccines that follow the DIVA (Differentiating Infected from Vaccinated Animals) concept because they usually contain no viral genetic material and thus can be distinguished from an infection with, for example, RVFV genomespecific RT-PCR [73-75]. This is especially important in agricultural sectors [9,76]. Additionally, because VLPs usually do not contain all of the protein components of their wildtype virus counterparts, DIVAconforming VLP-based vaccines can be analyzed with ELISA-based diagnostic tests for specific viral antigens. Using DIVA vaccines in previously unaffected areas is extremely valuable as it enables close monitoring of virus spread within a vaccinated population, allows for the adaptation of vaccination strategies during outbreaks, and could possibly reduce economic damage due to trade restrictions with disease-free countries [76].
VLP-based Vaccines for RVFV
Even though several promising RVFV vaccine candidates (for example, the live attenuated recombinant RVFV, MP-12 and a naturally attenuated RVFV, Clone 13), are currently being developed (reviewed in [1,3-5]), VLP-based vaccines provide a potent alternative vaccine platform, especially for humans and in locations where RVFV has not yet emerged [76]. A VLP vaccine platform presents an important option for the development of a safe and efficient RVFV vaccine because VLPs are unlikely to face serious safety issues regarding risk for the environment, since these vaccines are incapable of autonomous amplification and spread (as stated in a 2011 report by the Food and Agriculture Organization of the United Nations (FAO))[76].
Insect cell-derived RVF VLPs
The recombinant baculovirus-driven insect cell system presents an important platform for the production of VLPs [77-83], as illustrated by the successful employment for the generation of the FDA-approved VLP-based vaccine Cervarix for human papilloma virus types 16 and 18 [15,51].
It is likely that the first examples of RVF VLPs produced using an insect expression system were not originally characterized as VLPs [84,85]. Liu et al. [17] generated RVF VLPs in Spodoptera frugiperda insect cells using a single recombinant baculovirus encoding the RVFV nucleoprotein (N)and the glycoprotein precursor, which is post-translationally cleaved into the two mature glycoproteins GN and GC [17] (Figure 1). RVF VLPs were concentrated and purified by ultracentrifugation through potassium tartrate-glycerol gradients. The presence of RVFV proteins in the VLPs was confirmed by Western analysis with specific antibodies against GN and N, and a polyclonal antisera against RVFV particles. Transmission electron microscopic (TEM) analysis revealed 90–120 nm diameter particulate structures with a spiky outer layer that resembled the structure of RVFV. However, immunological and efficacy studies are required in suitable animal models to assess their utility as vaccine candidates against RVFV.
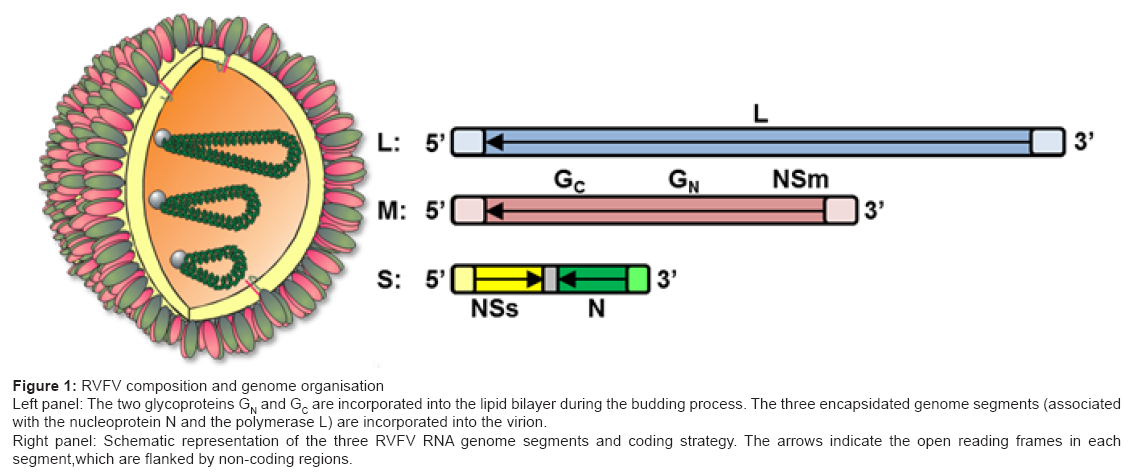
Figure 1: RVFV composition and genome organisation
Left panel: The two glycoproteins GN and GC are incorporated into the lipid bilayer during the budding process. The three encapsidated genome segments (associated
with the nucleoprotein N and the polymerase L) are incorporated into the virion.
Right panel: Schematic representation of the three RVFV RNA genome segments and coding strategy. The arrows indicate the open reading frames in each
segment,which are flanked by non-coding regions.
Further, Mandell et al. [34] generated RVF VLPs comprised of RVFV GN, GC and N proteins in Spodoptera frugiperda insect suspension cells using two separate recombinant baculovirus encoding the RVFV glycoprotein precursor and RVFV N, respectively [20]. The VLP composition was confirmed by Western analysis using anti- RVFV GN, GC and N antibodies. A strong humoral response was detected in vaccinated mice and rats using ELISAs to detect GN and N-specific antibodies. Furthermore, these RVF VLPs were shown to be 100% protective in mice and rats infected with a lethal dose of RVFV wild type virus, even with a single subcutaneous vaccination ([20] and unpublished data). Based on published studies, this was the first time that a single dose of a non-live attenuated RVFV vaccine has been 100%efficacious in a rat challenge model. Importantly, the RVFV ZH501 challenge was performed with a 2 log10 higher dose than is often reported in the literature to demonstrate vaccine potency. Taken together, the results of Liu et al. [17] and Mandell et al. [34] demonstrate that RVF VLPs can be generated with insect cells [17,20] and can be protective against lethal challenge in rodent models [20].
De Boer et al. [86] then described the generation of RVF VLPs using Drosophila insect cells that were stably transfected with an inducible expression plasmid containing the RVFV GN and GC envelope glycoprotein genes [86]. Morphological analysis by TEM confirmed the generation of particles resembling RVFV, and Western analysis of purified RVF VLPs confirmed the presence of both glycoproteins. A single intraperitoneal vaccination of mice with RVF VLPs was sufficient to induce GN and GC antibodies. Additionally, virus neutralization tests with sera from vaccinated mice confirmed the generation of neutralizing antibodies, even after a single vaccination. Finally, two intraperitoneal vaccinations with RVF VLPs were 100% protective in a mouse model.
Mammalian Cell-derived RVF VLPs
Efficient generation of transcriptionally-active mammalian cell-derived RVF VLPs has been reported by Habjan et al. [87] by transfection of 293T cells with plasmid DNAs encoding recombinant RVFV polymerase (L), nucleocapsid protein (N), a mini genome encoding GFP and an M segment expression construct (encoding GN, GC, NSm, and the 78 kDa protein) [87]. The VLPs were shown to contain RVFV GN, the 78kD protein and N by Western blot analysis, shown to resemble authentic virus particles by TEM, and were able to infect new cells as indicated by expression of a mini genome reporter gene (after infection, VLP-associated nucleocapsids autonomously performed primary transcription of the mini genome).Additionally, infection by VLPs was neutralized by RVFV-specific antisera, indicating that infection was RVFV protein-dependent.
Naslund et al. [88] then showed that three intraperitoneal injections with 1×106 of the transcriptionally-active VLPs resulted in the generation of a strong humoral immune response as indicated by high titers of RVFV-specific antibodies in a mouse model. The RVF VLPs were highly protective (92% vs 8% survival in vaccinated and control groups, respectively) against lethal challenge with RVFV [88]. RVFV RNA was quantified by qRT-PCR in blood samples of VLP-vaccinated and control animals after challenge. Results demonstrated that RVF VLP vaccination suppressed replication of the challenge virus, whereas high RNA levels were observed in the control animals.
Building on this successful platform, Pichlmair et al. [89] showed that a single inoculation of 1x106transcriptionally-active RVF VLPs that are able to facilitate expression of the RVFV nucleocapsid (N) gene(instead of the previously employed mini genome-driven reporter gene) in VLP-infected cells was sufficient to confer complete protection of mice from a lethal RVFV challenge [89]. Taken together, these results demonstrate that transcriptionally-active RVF VLPs are highly immunogenic and confer protection against RVFV infection in mice.
Mandell et al. [20,34] developed a simpler VLP-based vaccine candidate by generating RVF VLPs in HEK-293 cells by co-expressing RVFV glycoproteins and the nucleoprotein [20,34], and “chimeric” RVF VLPs by co-expression of the Moloney Murine leukemia virus matrix protein (gag) [34]. These RVF VLPs contain no viral genetic material and thus present a platform that should facilitate movement through the vaccine licensure process. Another important step was the generation of an HEK-293 suspension cell-based RVF VLP production system [20] that allows for scale-up and cost effective production amenable to commercial applications. Consistent with the analysis of previously described RVF VLPs, TEM analysis of RVFV G- and N-transfected 293 cells illustrated budding RVF VLPs on intracellular membranes and the appearance of putative RVFV glycoprotein spikes protruding from the VLP membranes [34].
RVF VLPs were also successfully generated without the nucleoprotein, which affords a simple means for antigen-based DIVA analyses [90,91]. Interestingly, results suggest that RVF VLP production is actually more efficient without N [20], and recent studies show that RVF VLPs without N are protective (unpublished data). However, contrary to these findings, a recent study by Piper et al. [31] showed that the incorporation of N leads to more efficient production of viral particles (with the inclusion of genomic RNA) [31].
Analysis of neutralizing antibody titers generated by vaccinated mice (Bal b/C) suggests that both chimeric RVF VLPs and RVF VLPs (containing RVFV N) can induce long-lasting humoral immune responses. RVFV-specific cytokine secretion profiles of spleenocytes from chimeric RVF VLP-vaccinated mice are consistent with the induction of both humoral and cellular immunity[34]. However, immune correlates alone are not always predictive of efficacy in a live challenge model. Therefore, efficacy studies were performed in two different rodent models. First, three vaccinations of either of the RVF VLPs were shown to be up to 100% protective in mice against a 103 pfu challenge with RVFV strain ZH501 ([34] and unpublished data). Second, chimeric RVF VLPs (containing RVFV N) were shown to be 100% protective in a rat (Wistar-Furth) model against a 105 pfu challenge with RVFV strain ZH501 [20], and no clinical signs of disease (e.g., weight loss) were observed during the course of the experiment. Further optimization and characterization of the RVF VLP vaccine candidate led to a recent study resulting in full protection of mice even after a single immunization (unpublished data).
Conclusion
Although the impact of RVFV to agriculture and human health would be severe [9], there are currently no US Department of Agriculture (USDA) or FDA-approved prophylactic or therapeutic countermeasures for this viral pathogen. VLPs clearly present an important platform for the generation of urgently needed vaccines to prevent the spread of RVFV. RVF VLPs have been prepared with several different cell-based systems and, as vaccine candidates, have yielded very promising immunological and efficacy data in two rodent models. The results described above suggest that an efficacious, DIVA-conforming VLP-based vaccine is a safe and practical strategy for developing a broadly applicable vaccine candidate against RVFV. However, while a viable alternative human vaccine candidate, more improvements are needed before this VLP-based vaccine platform would be practical in the poorest countries of Africa where RVF is endemic in livestock (i.e., cost, storage conditions, scale-up improvements).
References
- Bouloy M, R Flick (2009) Reverse genetics technology for Rift Valley fever virus: current and future applications for the development of therapeutics and vaccines. Antiviral Res 84: 101-118.
- Bouloy M, F Weber (2009) Molecular biology of rift valley Fever virus. Open Virol J 4: 8-14.
- Ikegami T, S Makino (2009) Rift valley fever vaccines. Vaccine 27 Suppl 4: D69-72.
- Bird B H, et al. (2009)Rift Valley fever virus. J Am Vet Med Assoc 234: 883-893.
- LaBeaud AD, JW Kazura, CH King (2010) Advances in Rift Valley fever research: insights for disease prevention. Curr Opin Infect Dis 23: 403-408.
- Pepin M, Bouloy M, Bird BH, Kemp A, Paweska J (2010) Rift Valley fever virus(Bunyaviridae: Phlebovirus): an update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. Vet Res 41: 61.
- Kasari TR, Carr DA, Lynn TV, Weaver JT (2008) Evaluation of pathways for release of Rift Valley fever virus into domestic ruminant livestock, ruminant wildlife, and human populations in the continental United States. J Am Vet Med Assoc 232: 514-529.
- Chevalier V, Pepin M, Plee L, Lancelot R (2010) Rift Valley fever-a threat for Europe?. Euro Surveill 15: 19506.
- Mandell RB, R Flick (2010) Rift Valley fever virus: An unrecognized emerging threat? Hum Vaccin 6: 597-601.
- Mandell RB, R Flick (2011) Rift Valley Fever Virus: A Real Bioterror Threat. J Bioterr Biodef 2: 108.
- Noad R, P Roy (2003) Virus-like particles as immunogens. Trends Microbiol 11: 438-444.
- Garcea RL, L Gissmann (2004) Virus-like particles as vaccines and vessels for the delivery of small molecules. Curr Opin Biotechnol 15: 513-517.
- Grgacic EV, DA Anderson (2006) Virus-like particles: passport to immune recognition. Methods 40: 60-65.
- Roldao A, Mellado MC, Castilho LR, Carrondo MJ, Alves PM (2010) Virus-like particles in vaccine development. Expert Rev Vaccines 9: 1149-1176.
- Monie A, Hung CF, Roden R, Wu TC (2008) Cervarix: a vaccine for the prevention of HPV 16, 18-associated cervical cancer. Biologics 2: 97-105
- Harper DM (2009) Currently approved prophylactic HPV vaccines. Expert Rev Vaccines 8: 1663-1679.
- Liu L, CC Celma, P Roy (2008) Rift Valley fever virus structural proteins: expression, characterization and assembly of recombinant proteins. Virol J 5: 82.
- Metz SW, GP Pijlman (2011) Arbovirus vaccines; opportunities for the baculovirus-insect cell expression system. J Invertebr Pathol 107 Suppl: S16-S30.
- Vicente T, Roldao A, Peixoto C, Carrondo MJ, Alves PM (2011) Large-scale production and purification of VLP-based vaccines. J Invertebr Pathol 107 Suppl: S42-S48.
- Mandell RB, Koukuntla R, Mogler LJ, Carzoli AK, Holbrook MR et al. (2010) Novel suspension cell-based vaccine production systems for Rift Valley fever virus-like particles. J Virol Methods 169: 259-268.
- Govan VA (2008) A novel vaccine for cervical cancer: quadrivalent human papillomavirus (types 6, 11, 16 and 18) recombinant vaccine (Gardasil). Ther Clin Risk Manag 4: 65-70.
- Landry N, Ward BJ, Trepanier S, Montomoli E, Dargis M et al. (2010) Preclinical and clinical development of plant-made virus-like particle vaccine against avian H5N1 influenza. PLoS One 5: e15559.
- Soria-Guerra RE, L Moreno-Fierros, S Rosales-Mendoza (2011) Two decades of plant-based candidate vaccines: a review of the chimeric protein approaches. Plant Cell Rep 30: 1367-1382.
- Bos EC, W Luytjes, WJ Spaan (1997) The function of the spike protein of mouse hepatitis virus strain A59 can be studied on virus-like particles: cleavage is not required for infectivity. J Virol 71: 9427-9433.
- Johnson JE, W Chiu (2000) Structures of virus and virus-like particles. Curr Opin Struct Biol 10: 229-235.
- Li PP, Naknanishi A, Tran MA, Ishizu K, Kawano M et al. (2003) Importance of Vp1 calcium-binding residues in assembly, cell entry, and nuclear entry of simian virus 40. J Virol 77: 7527-7538.
- Licata JM, Johnson RF, Han Z, Harty RN (2004) Contribution of ebola virus glycoprotein, nucleoprotein, and VP24 to budding of VP40 virus-like particles. J Virol 78: 7344-7351.
- Schmitt AP, Leser GP, Waning DL, Lamb RA (2002) Requirements for budding of paramyxovirus simian virus 5 virus-like particles. J Virol 76: 3952-3964.
- Overby AK, Popov V, Neve EP, Petterson RF (2006) Generation and analysis of infectious virus-like particles of uukuniemi virus (bunyaviridae): a useful system for studying bunyaviral packaging and budding. J Virol 80: 10428-10435
- Ye L, Lin J, Sun Y, Bennouna S, Lo M et al. (2006) Ebola virus-like particles produced in insect cells exhibit dendritic cell stimulating activity and induce neutralizing antibodies. Virology 351: 260-270.
- Piper ME, DR Sorenson, SR Gerrard (2011) Efficient cellular release of Rift Valley fever virus requires genomic RNA. PLoS One 6: e18070.
- Watanabe S, Watanabe T, Noda T, Takada A, Feldmann H et al.(2004) Production of novel ebola virus-like particles from cDNAs: an alternative to ebola virus generation by reverse genetics. J Virol 78: 999-1005.
- Warfield KL, Swenson DL, Negley DL, Schmaljohn AL, Aman MJ et al. (2004) Marburg virus-like particles protect guinea pigs from lethal Marburg virus infection. Vaccine 22: 3495-3502.
- Mandell RB, Koukuntla R, Mogler LJ, Carzoli AK, Freiberg AN et al. (2010)A replication-incompetent Rift Valley fever vaccine: chimeric virus-like particles protect mice and rats against lethal challenge. Virology 397: 187-198.
- Chackerian B (2007) Virus-like particles: flexible platforms for vaccine development. Expert Rev Vaccines 6: 381-390.
- Grgacic EV, Anderson DA (2006) Virus-like particles; passport to immune recognition. Methods 40: 60-65.
- Noad R, Roy P (2003) Virus-like particles as immunogens. Trends Microbiol 11: 438–444.
- Schirmbeck R, W Bohm, J Reimann (1996) Virus-like particles induce MHC class I-restricted T-cell responses. Lessons learned from the hepatitis B small surface antigen. Intervirology 39: 111-119.
- Paliard X, Liu Y, Wagner R, Wolf H, Baenziger J et al. (2000) Priming of strong, broad, and long-lived HIV type 1 p55gag-specific CD8+ cytotoxic T cells after administration of a virus-like particle vaccine in rhesus macaques. AIDS Res Hum Retroviruses 16: 273-282.
- Murata K, Lechmann M, Qiao M, Gunji T, Alter HJ et al. (2003) Immunization with hepatitis C virus-like particles protects mice from recombinant hepatitis C virus-vaccinia infection. Proc Natl Acad Sci U S A 100: 6753-6758.
- Jegerlehner A, Wiesel M, Dietmeier K, Zabel F, Gatto D et al. (2010) Carrier induced epitopic suppression of antibody responses induced by virus-like particles is a dynamic phenomenon caused by carrier-specific antibodies. Vaccine 28: 5503-5512.
- Caldeira JC, DS Peabody (2011) Thermal stability of RNA phage virus-like particles displaying foreign peptides. J Nanobiotechnology 9: 22.
- Tissot AC, Renhofa R, Schmitz N, Cielens I, Meijerink E et al.(2010) Versatile virus-like particle carrier for epitope based vaccines. PLoS One 5: e9809.
- McGinnes LW, Pantua H, Laliberte JP, Gravel KA, Jain S et al. (2010) Assembly and biological and immunological properties of Newcastle disease virus-like particles. J Virol 84: 4513-4523.
- Patel KG, JR Swartz (2011) Surface functionalization of virus-like particles by direct conjugation using azide-alkyne click chemistry. Bioconjug Chem 22: 376-387.
- Pejawar-Gaddy S, Rajawat Y, Hilioti Z, Xue J, Gaddy DF et al. (2010) Generation of a tumor vaccine candidate based on conjugation of a MUC1 peptide to polyionic papillomavirus virus-like particles. Cancer Immunol Immunother 59: 1685-1696.
- Yildiz I, S Shukla, NF Steinmetz (2011) Applications of viral nanoparticles in medicine. Curr Opin Biotechnol.
- Middelberg AP, Rivera-Hernandez T, Wibowo N, Lua LH, Fan Y et al. (2011) A microbial platform for rapid and low-cost virus-like particle and capsomere vaccines. Vaccine 29: 7154-7162.
- Lu B, Kumar A, Castellsaque X, Giuliano AR (2011) Efficacy and safety of prophylactic vaccines against cervical HPV infection and diseases among women: a systematic review & meta-analysis. BMC Infect Dis 11: 13.
- Sitrin RD, DE Wampler, RW Ellis (1993) Survey of licensed hepatitis B vaccines and their production processes, in Hepatitis B vaccines in clinical practice RW Ellis Editor Marcel Dekker Inc New York. 83-102.
- FDA licensure of bivalent human papillomavirus vaccine (HPV2, Cervarix) for use in females and updated HPV vaccination recommendations from the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep, 2010. 59: 626-629.
- Akahata W, Yang ZY, Andersen H, Sun S, Holdaway H et al. (2010) A virus-like particle vaccine for epidemic Chikungunya virus protects nonhuman primates against infection. Nat Med 16: 334-338.
- Warfield KL, Posten NA, Swenson DL, Olinger GG, Esposito D et al. (2007) Filovirus-like particles produced in insect cells: immunogenicity and protection in rodents. J Infect Dis 196 Suppl 2: S421-S429.
- Warfield KL, Swenson DL, Demmin G, Bavari S (2005) Filovirus-like particles as vaccines and discovery tools. Expert Rev Vaccines 4: 429-440.
- Pushko P, Kort T, Nathan M, Pearce MB, Smith G et al. (2010) Recombinant H1N1 virus-like particle vaccine elicits protective immunity in ferrets against the 2009 pandemic H1N1 influenza virus. Vaccine 28: 4771-4776.
- Giles BM, TM Ross (2011) A computationally optimized broadly reactive antigen (COBRA) based H5N1 VLP vaccine elicits broadly reactive antibodies in mice and ferrets. Vaccine 29: 3043-3054.
- Kang SM,Yoo DG, Lipatov AS, Song JM, Davis CT et al. (2009) Induction of long-term protective immune responses by influenza H5N1 virus-like particles. PLoS One 4: e4667.
- Ding H, Tsai C, Zhou F, Buchy P, Deubel V et al. (2011) Heterosubtypic antibody response elicited with seasonal influenza vaccine correlates partial protection against highly pathogenic H5N1 virus. PLoS One 6: e17821.
- Ding H, Tsai C, Gutierrez RA, Zhou F, Buchy P et al. (2011) Superior neutralizing antibody response and protection in mice vaccinated with heterologous DNA prime and virus like particle boost against HPAI H5N1 virus. PLoS One 6: e16563.
- Li C, Liu F, Liang M, Zhang Q, Wang X et al. (2010) Hantavirus-like particles generated in CHO cells induce specific immune responses in C57BL/6 mice. Vaccine 28: 4294-4300.
- Casabona JC, Levingston Macleod JM, Loureiro ME, Gomez GA, Lopez N et al. (2009) The RING domain and the L79 residue of Z protein are involved in both the rescue of nucleocapsids and the incorporation of glycoproteins into infectious chimeric arenavirus-like particles. J Virol 83: 7029-7039.
- Branco LM, Grove JN, Geske FJ, Boisen ML, Muncy IJ et al. (2010) Lassa virus-like particles displaying all major immunological determinants as a vaccine candidate for Lassa hemorrhagic fever. Virol J 7: 279.
- Walpita P, Barr J, Sherman M, Basler CF, Wang L (2011) Vaccine potential of Nipah virus-like particles. PLoS One 6: 10.
- Patch JR, Crameri G, Wang LF, Eaton BT, Broder CC (2007) Quantitative analysis of Nipah virus proteins released as virus-like particles reveals central role for the matrix protein. Virol J 4: 1.
- Rohn TA, Ralvenius WT, Paul J, Borter P, Hernandez M et al. (2011) A virus-like particle-based anti-nerve growth factor vaccine reduces inflammatory hyperalgesia: potential long-term therapy for chronic pain. J Immunol 186: 1769-1780.
- Jennings GT, MF Bachmann (2009) Immunodrugs: therapeutic VLP-based vaccines for chronic diseases. Annu Rev Pharmacol Toxicol 49: 303-326.
- Ambuhl PM, Tissot AC, Fulurija A, Maurer P, Nussberger J et al. (2007) A vaccine for hypertension based on virus-like particles: preclinical efficacy and phase I safety and immunogenicity. J Hypertens 25: 63-72.
- Chackerian B, Rangel M, Hunter Z, Peabody DS (2006) Virus and virus-like particle-based immunogens for Alzheimer's disease induce antibody responses against amyloid-beta without concomitant T cell responses. Vaccine 24: 6321-6331.
- Roy P, M Boyce, R Noad (2009) Prospects for improved bluetongue vaccines. Nat Rev Microbiol 7: 120-128.
- Roy P, R Noad (2008) Virus-like particles as a vaccine delivery system: myths and facts. Hum Vaccin 4: 5-12.
- Remond M, Da Costa B, Riffault S, Parida S, Breard E et al. (2009) Infectious bursal disease subviral particles displaying the foot-and-mouth disease virus major antigenic site. Vaccine 27: 93-98.
- Li Z, Yi Y, Yin X, Zhang Z, Liu J (2008) Expression of foot-and-mouth disease virus capsid proteins in silkworm-baculovirus expression system and its utilization as a subunit vaccine. PLoS One 3: e2273.
- Capua I, G Cattoli, S Marangon (2004) DIVA--a vaccination strategy enabling the detection of field exposure to avian influenza. Dev Biol (Basel) 119: 229-233.
- Bird BH, Albarino CG, Hartman AL, Erickson BR, Ksiazek TG et al. (2008) Rift valley fever virus lacking the NSs and NSm genes is highly attenuated, confers protective immunity from virulent virus challenge, and allows for differential identification of infected and vaccinated animals. J Virol 82: 2681-2691.
- Lee DH, Park JK, Lee YN, Song JM, Kang SM et al. (2011) H9N2 avian influenza virus-like particle vaccine provides protective immunity and a strategy for the differentiation of infected from vaccinated animals. Vaccine 29: 4003-4007.
- FAO, Rift Valley fever vaccine development, progress and constraints. In Proceedings of the GF-TADs meeting. 2011, FAO Animal Production and Health Proceedings: Rome, Italy.
- Latham T, JM Galarza (2001) Formation of wild-type and chimeric influenza virus-like particles following simultaneous expression of only four structural proteins. J Virol 75: 6154-6165.
- Gheysen D, Jacobs E, de Foresta F, Thiriart C, Francotte M et al. (1989) Assembly and release of HIV-1 precursor Pr55gag virus-like particles from recombinant baculovirus-infected insect cells. Cell 59: 103-112.
- Loudon PT, P Roy (1991) Assembly of five bluetongue virus proteins expressed by recombinant baculoviruses: inclusion of the largest protein VP1 in the core and virus-like proteins. Virology 180: 798-802.
- Nermut MV, Hockley DJ, Jowett JB, Jones IM, Garreau M et al. (1994) Fullerene-like organization of HIV gag-protein shell in virus-like particles produced by recombinant baculovirus. Virology 198: 288-296.
- Rose RC, Bonnez W, Reichman RC, Garcea RL (1993) Expression of human papillomavirus type 11 L1 protein in insect cells: in vivo and in vitro assembly of viruslike particles. J Virol 67: 1936-1944.
- Yamshchikov GV, Ritter GD, Vey M, Compans RW (1995) Assembly of SIV virus-like particles containing envelope proteins using a baculovirus expression system. Virology 214: 50-58.
- Zeng CQ, Wentz MJ, Cohen J, Estes MK, Ramig RF (1996) Characterization and replicase activity of double-layered and single-layered rotavirus-like particles expressed from baculovirus recombinants. J Virol 70: 2736-2742.
- Takehara K, S Morikawa, DH Bishop (1990) Characterization of baculovirus-expressed Rift Valley fever virus glycoproteins synthesized in insect cells. Virus Res 17: 173-190.
- Schmaljohn CS, Parker MD, Ennis WH, Dalrymple JM, Collett MS et al. (1989) Baculovirus expression of the M genome segment of Rift Valley fever virus and examination of antigenic and immunogenic properties of the expressed proteins. Virology 170: 184-192.
- de Boer SM, Kortekaas J, Antonis AF, Kant J, van Oploo JL et al. (2010) Rift Valley fever virus subunit vaccines confer complete protection against a lethal virus challenge. Vaccine 28: 2330-2339.
- Habjan M, Penski N, Wagner V, Spiegel M, Overby AK et al. (2009) Efficient production of Rift Valley fever virus-like particles: The antiviral protein MxA can inhibit primary transcription of bunyaviruses. Virology 385: 400-408.
- Naslund J, Lagergvist N, Habjan M, Lundkvist A, Evander M et al. (2009) Vaccination with virus-like particles protects mice from lethal infection of Rift Valley Fever Virus. Virology 385: 409-415.
- Pichlmair A, Habjan M, Unger H, Weber F (2010) Virus-like particles expressing the nucleocapsid gene as an efficient vaccine against Rift Valley fever virus. Vector Borne Zoonotic Dis 10: 701-703.
- McElroy AK, CG Albarino, ST Nichol (2009) Development of a RVFV ELISA that can distinguish infected from vaccinated animals. Virol J 6: 125.
- van Vuren PJ, JT Paweska (2010) Comparison of enzyme-linked immunosorbent assay-based techniques for the detection of antibody to Rift Valley fever virus in thermochemically inactivated sheep sera. Vector Borne Zoonotic Dis 10: 697-699.
Relevant Topics
- Anthrax Bioterrorism
- Bio surveilliance
- Biodefense
- Biohazards
- Biological Preparedness
- Biological Warfare
- Biological weapons
- Biorisk
- Bioterrorism
- Bioterrorism Agents
- Biothreat Agents
- Disease surveillance
- Emerging infectious disease
- Epidemiology of Breast Cancer
- Information Security
- Mass Prophylaxis
- Nuclear Terrorism
- Probabilistic risk assessment
- United States biological defense program
- Vaccines
Recommended Journals
Article Tools
Article Usage
- Total views: 17101
- [From(publication date):
specialissue-2012 - Dec 20, 2025] - Breakdown by view type
- HTML page views : 12234
- PDF downloads : 4867