Review Article Open Access
What the Novel H1N1 Influenza Outbreak Can Teach us about Influenza as a Bioterrorism Weapon
Matthew P Morrow*, Jian Yan, Amir S Khan, Kate E Broderick, Niranjan Y Sardesai
Inovio Pharmaceuticals
- *Corresponding Author:
- Matthew P Morrow
Inovio Pharmaceuticals
1787 Sentry Parkway West
Building 18, Suite 400
Blue Bell, PA 19422, USA
Email: mmorrow@inovio.com
Received Date: Spetember 15, 2011; Accepted Date: January 27, 2012; Published Date: February 05, 2012
Citation: Morrow MP, Yan J, Khan AS, Broderick KE, Sardesai NY (2012) What the Novel H1N1 Influenza Outbreak Can Teach us about Influenza as a Bioterrorism Weapon. J Bioterr Biodef S1:011. doi:10.4172/2157-2526.S1-011
Copyright: © 2012 Morrow MP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Bioterrorism & Biodefense
Abstract
While we often see Influenza infections as part of our daily lives during winter in the United States, it is a virus with significant potential for manipulation into an agent that may be used in the framework of Bioterrorism. The Novel H1N1 outbreak of 2009 highlighted what our response is to such an unsuspected outbreak both on a national and global scale. The responses to this outbreak allowed us to avert significant damage from the Pandemic, but retrospective analysis suggests that if the outbreak had been more virulent, greater impact would have been felt on a global scale. This short review attempts to briefly summarize our response to the Novel H1N1 outbreak in the United States and to apply the knowledge gleaned from this analysis to a hypothetical Bioterrorism attack that employs a modified Influenza virus, an event that is unlikely but not impossible.
Review
In March of 2009, a new variant of an already well known virus began a journey that would culminate with its rapid spread across the earth. In less than a year and a half, the viral outbreak had officially been termed a pandemic by the World Health Organization and had claimed over 14,000 lives [1]. This virus – Influenza – had caused pandemics before, and a new pandemic caused by Flu had been expected by experts in the field prior to this outbreak. What was not expected, however, was the genetic structure of this particular variant (an H5 virus was assumed to be the next to cause a pandemic) as well as it’s proclivity for infecting people who had been deemed otherwise healthy, as opposed to targeting the very young and very old. The Novel H1N1 pandemic of 2009 underscores a number of important issues concerning Influenza infection; including ease of transmission, viral tropism in respect to patient populations, antiviral drug resistance and the speed at which a vaccine can be made in response to an established outbreak. It may be in our best interest to seize and expand upon the positive aspects of the global response to the pandemic, especially when one considers the relative ease of acquiring the Influenza virus and its ability to be weaponized as noted by the CDC, among other groups [2,3]. These two aspects make the risk of the use of Influenza as a Bioterrorism agent (while not highly likely) a real possibility, and lessons learned from the Novel H1N1 pandemic may serve us well in the future as a response guide if such an event were to occur.
As of January of 2003, the NIAID has identified Influenza as a Category C Priority Pathogen in its Biodefense Research Agenda under the Emerging Infectious Disease category [3]. The Agenda specifically notes concerns about the use of Influenza as a biological threat agent, especially in light of the advances the scientific community has made in genetic engineering. Moreover, the Agenda notes the “devastating impact” of the 1918 Influenza Pandemic, which was estimated to have killed roughly 21 million people [3]. Had this Agenda been written more recently, one would expect the Novel H1N1 pandemic to also have been highlighted in regards to our relative lack of preparedness to a new Influenza variant on a global scale. While the advances in genetic engineering noted by the Agenda are, indeed, cause for concern in regards to the use of Influenza as a Bioterrorism agent, there are a number of additional factors to take into consideration when analyzing Influenza as a Bioterrorism threat.
It is not a single factor, but rather a culmination of multiple factors that lead one to determine that Influenza has Bioterrorism potential, limited as it may be. Among the more startling facts in this regard is the difference in required infectious dose of Influenza between direct contact (i.e. person to person) as compared with aerosol transmission. Infection by Influenza via aerosol transmission is far more efficient than person to person contact, taking an estimated 27,000 times fewer virions to induce productive infection leading to disease [2,4]. Considering that the virus is routinely aerosolized during even seasonal Influenza outbreaks, this fact requires us to take into account the possibility of genetically modified Influenza virus being released in an aerosol fashion – a method of delivery that would very likely be completely undetectable at the time of occurrence. Moreover, such a delivery method and subsequent viral outbreak would may not be immediately recognized as a Bioterrorism attack due to the fact that Influenza is not an uncommon virus, leading to a lag time between the attack and an appropriate response. While circulation and infection with Influenza is generally centered around the winter months in the Northern Hemisphere, cases do present themselves, even to the degree of requiring hospitalization, during all times of the year. Thus an increase in Influenza infections, even in the “off-season”, would not stimulate an immediate public health response to the same degree as an infectious agent that does not commonly circulate, such as Smallpox. Also of distinct possibility is that the release of such a modified virus would happen during the times of peak seasonal Influenza infection in a country such as the United States, which may allow the attack to be unrealized for a longer period of time, as most initial cases of infection would be thought to be due to poor coverage of the seasonal Influenza vaccine for the circulating strains – an event which most recently happened during 2007 in which the seasonal vaccine afforded only 30% coverage [5]. If we further compound this scenario with the fact that the incubation time for Influenza is relatively short when compared with Smallpox (1-4 days for Flu versus 10-14 days for Smallpox [2]), a situation presents itself in which a Bioterrorism agent has been released without notice due to its possible misevaluation as a standard seasonal viral infection and spreads rapidly through the population to a comparatively rapid degree. Indeed, it was only after Novel H1N1 had reached Pandemic levels that significant barriers were put into place to blunt it’s spread (such as the creation of a vaccine, government advisories to avoid crowds, closing off non-essential public buildings and, in some countries, full quarantine [6-9]). Also of note was the resistance of Novel H1N1 to oseltamivir [10], a drug resistance that is not uncommon among H1 Influenza viruses. Thankfully, Novel H1N1 retained its susceptibility to zanamivir, making it an effective treatment during the outbreak [11]. One might assume that an Influenza variant that had been engineered as a Bioterrorism agent may have been done so in a fashion that affords resistance to both of these compounds, or additionally to amantadine and rimantidine, both of which can cover both Influenza A and Influenza B viruses [12]. The concept of the possibility of a bioterror agent exhibiting resistance to some, but perhaps not all, of these drugs suggests that stockpiling all four compounds may be a key factor in blunting the spread the virus.
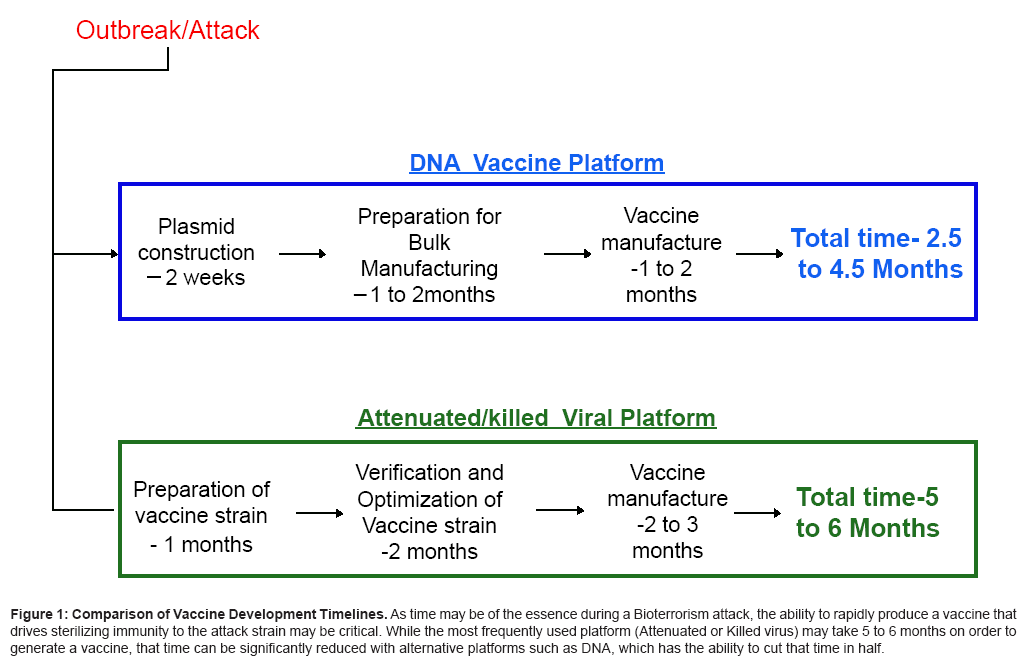
If one does assume that weaponized Influenza is released as part of a terror attack and if we additionally assume the very likely possibility that the virus will have been genetically engineered to be resistant to one or more antiviral drugs, our best and most effective response would be the rapid creation of a vaccine. In the case of the Novel H1N1 pandemic, the first vaccines that offered protective efficacy were available in early October in the United States, which was five months after the WHO had elevated the Pandemic Alert Level to Phase 6 – the highest possible [13]. Additionally, health departments across the United States ran out of the vaccine less than a month into dosing due to a shortfall of 10 million doses [14], although no declaration of a possible shortage by the CDC was officially announced once production of the vaccine began. This shortfall was due in large part to the mechanism by which a standard influenza vaccine is created; the growth of influenza virus in chicken eggs. Vaccine developers found that Novel H1N1 exhibited different growth characteristics than other H1N1 viruses within the egg embryos, and thus the ability to produce the amount of vaccine originally estimated to be delivered was hampered [15]. Thankfully, the fallout of the shortage was blunted due in large part to the fact that sterilizing immune responses were found to be generated after a single vaccination as opposed to the 2-vaccine regimen originally proposed [16]. However, should this same shortfall occur in the scope of a response to a Bioterrorism situation, the results may be far more dire. Therefore, the need for the generation of a Universal Influenza Vaccine that has the possibility of covering emerging or engineered strains and that is constructed and distributed prior to this type of event may be crucial to blunting the destructive influence of an Influenza-based Bioterrorism attack. Moreover, the possibility of the generation of new viral strains through interaction and reassortment of circulating seasonal strains with the attack strain further compounds the problem of vaccine induced coverage, thus highlighting the need not only for a vaccine that induces broad coverage, but also for a vaccine platform that allows for rapid propagation in response to both the attack strain and to any new variant that arises due to strain interactions during infection. DNA-based vaccines offer this type of increased speed of production (Figure 1) as well as the ability to induce heterosubtypic protection across flu strains [5] and studies have shown that a DNA vaccine composed of a single plasmid encoding the Hemagglutanin of H1N1 was able to generate immune responses known to correlate with protection from influenza infection after administration [5,17-19]. Moreover, studies of influenza challenges in animal models have shown that a number of DNA vaccines of differing compositions have been able to afford complete survival against lethal challenges [20,21]. Of course, the DNA platform is just one example of mechanism by which a vaccine may be made during in response to a Bioterror attack and regardless of what method is ultimately chosen the key factor in our response to such an event is clear: time.
Figure 1: Comparison of Vaccine Development Timelines. As time may be of the essence during a Bioterrorism attack, the ability to rapidly produce a vaccine that drives sterilizing immunity to the attack strain may be critical. While the most frequently used platform (Attenuated or Killed virus) may take 5 to 6 months on order to generate a vaccine, that time can be significantly reduced with alternative platforms such as DNA, which has the ability to cut that time in half.
References
- ECDC Daily Update - Pandemic H1N1 2009 (2010) European Centre for Disease Prevention and Control.
- Madjid M, Lillibridge S, Mirhaji P, Casscells W (2003) Influenza as a Bioweapon. J R Soc Med 96: 345-346.
- CDC (2003) NIAID Biodefense Research Agenda for Category B and Category C Pathogens.
- Alford RH KJ, Gerone PJ, Knight V (1966) Human Influenza resulting from aerosol inhalation. Proc Soc Exp Biol Med 122: 800-804.
- Laddy DJ, Yan J, Kutzler M, Kobasa D, Kobinger GP, et al. (2008) Heterosubtypic protection against pathogenic human and avian influenza viruses via in vivo electroporation of synthetic consensus DNA antigens. PLoS ONE 3: e2517.
- George C (2009) Schools revamp swine flu plans for fall.
- (2009) Business Planning. Flugov.
- (2009) China quarantines U.S. school group over flu concerns.
- (2009) Ship passengers cruisy in swine flu quarantine.
- Update on oseltamivir resistance to influenza H1N1 (2009) viruses.
- CDC (2009) 2008-2009 Influenza Season Week 32 ending August 15, 2009.
- Maugh TH (1976) "Amantadine: an Alternative for Prevention of Influenza". Science 192: 130-131.
- BBC (2009) WHO Declares swine flu pandemic.
- CNN (2009) Frustration looms as H1N1 vaccines run out.
- AP (2009) WHO seeks swine flu vaccine help for poor nation. USA Today.
- CDC (2009) Vaccine against 2009 H1N1 Influenza Virus.
- Laddy DJ, Yan J, Corbitt N, Kobasa D, Kobinger GP, et al. (2007) Immunogenicity of novel consensus-based DNA vaccines against avian influenza. Vaccine 25: 2984-2989.
- Ulmer JB, Donnelly JJ, Parker SE, Rhodes GH, Felgner PL, et al. (1993) Heterologous protection against influenza by injection of DNA encoding a viral protein. Science 259: 1745-1749.
- Chen J, Fang F, Li X, Chang H, Chen Z (2005) Protection against influenza virus infection in BALB/c mice immunized with a single dose of neuraminidase-expressing DNAs by electroporation. Vaccine 29: 4322-4328.
- Kutzler MA, Kraynya K, Nagle SJ, Parkinson RM, Zharikova D, et al. (2010) Plasmids encoding the mucosal chemokines CCL27 and CCL28 are effective adjuvants in eliciting antigen-specific immunity in vivo. Gene Ther 17: 72-82.
- Morrow MP, Pankhong P, Laddy DJ, Schoenly KA, Yan J, et al. (2009) Comparative ability of IL-12 and IL-28B to regulate Treg populations and enhance adaptive cellular immunity. Blood 113: 5868-5877.
Relevant Topics
- Anthrax Bioterrorism
- Bio surveilliance
- Biodefense
- Biohazards
- Biological Preparedness
- Biological Warfare
- Biological weapons
- Biorisk
- Bioterrorism
- Bioterrorism Agents
- Biothreat Agents
- Disease surveillance
- Emerging infectious disease
- Epidemiology of Breast Cancer
- Information Security
- Mass Prophylaxis
- Nuclear Terrorism
- Probabilistic risk assessment
- United States biological defense program
- Vaccines
Recommended Journals
Article Tools
Article Usage
- Total views: 14291
- [From(publication date):
specialissue-2012 - Dec 11, 2025] - Breakdown by view type
- HTML page views : 9613
- PDF downloads : 4678