Research Article Open Access
Are Platelet Rich Plasma Injections More Effective in Tendinopathy or Enthesopathy?
Rocco Papalia1*, Biagio Zampogna1, Gianluca Vadala1, Alberto Di Martino1, Carolina Nobile2, Angelo Del Buono1, Guglielmo Torre1, Maria Cristina Tirindelli2, Nicola Maffulli3 and Vincenzo Denaro1
1Department of Orthopedics and Trauma Surgery, Campus Bio-medico University of Rome, Via Alvaro del Portillo Rome, Italy
2Department of Haematology, Stem Cell Transplantation, Transfusion Medicine and Cellular Therapy, Campus Bio-Medico University Hospital, Via Alvaro del Portillo, Rome, Italy
3Department of Musculoskeletal Disorders, Faculty of Medicine and Surgery, University of Salerno, Italy
- *Corresponding Author:
- Rocco Papalia
Department of Orthopedics and Trauma Surgery
Campus Bio-medico University of Rome
Via Alvaro del Portillo 200, 00128 Rome, Italy
Tel: + +393346567482
E-mail: r.papalia@unicampus.it
Received date: March 26, 2017; Accepted date: April 07, 2017; Published date: April 12, 2017
Citation: Papalia R, Zampogna B, Vadala G, Di Martino A, Nobile C, et al. (2017) Are Platelet Rich Plasma Injections More Effective in Tendinopathy or Enthesopathy? J Pain Relief 6:288. doi: 10.4172/2167-0846.1000288
Copyright: © 2017 Papalia R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Pain & Relief
Abstract
Introduction: Chronic overuse insertional tendinopathy (entesopathy) is highly prevalent, but is an extremely difficult condition that generates a high medical expense. Alternative and co-adjuvant therapies to improve the quality of life and physical function of affected patients are currently being sought.
Materials and Methods: A total of 31 patients affected by entesopathy at different anatomical sites (elbow tendinopathy, greater trochanter pain syndrome and plantar fasciopathy) were treated with three peritendinous injections of autologous platelet rich plasma (PRP) and included in the present investigation. VAS scale and segmental scores for the affected site before the first injection of PRP and at 6 months after the last injection were used. One hundred and seven patients that underwent the same injection protocol in the same time lapse were used as a control group, and were represented by patients with non-insertional tendinopathy (shoulder and at Achilles tendon).
Results: Significantly lower values between pre-treatment and follow-up pain scores at all-time points were found in the patients affected by entesopathy compared to tendinopathy (p<0.001). As a confirmative finding, the segmental scores at the shoulder and ankle did not improve overtime, differently from what occurred in patients with entesopathy.
Conclusion: At 6 months following peritendinous injections of PRP in patients with entesopathy there was a significant reduction of pain, associated to a significant improvement in recorded segmental scores. Conversely, the treatment was not effective in patients with pure tendinopathy at the shoulder and ankle level. These favorable findings point to consider PRP as a promising therapy for patients affected by entesopathy.
Keywords
Platelet rich plasma; Tendinopathy; Blood; Trochanteric pain syndrome
Introduction
Tendinopathies are very common in athletes. Traditionally, the first management is conservative, including analgesics, anti-inflammatory drugs [1], and physical therapy [2]. In the last decade, Platelet-richplasma (PRP) products have been increasingly used [3,4], but their effectiveness is still controversial. PRP is a plasma gel with high concentration of platelets, higher than that present in the blood, as clinical benefits have been shown when this concentration is at least 1,000,000/μl in 5ml of plasma [5]. There is evidence that PRP provide good excellent outcomes in patients with tendon disorders [6-10], but few studies have reported on the use of PRP in patients with insertional tendinopathies of the elbow [11,12] and patellar tendon (jumper’s knee) [13,14]. The primary endpoint of this retrospective study is to assess clinical and functional outcomes of patients with tendinopathies of the main body of the tendon and insertional tendinopathies who had undergone local injections of PRP.
Materials and Method
This study was carried out at the Department of Orthopaedic and Trauma Surgery and the Immunohematology Transfusion Medicine Unit of Campus Bio-Medico University Hospital. The Local Ethics Committee approved the study, and informed consent was obtained from all individual participants included in the study.
Subject’s selection process
Four hundred patients underwent PRP injections for management of tendon disorders at Campus Bio-Medico University from January 2011 to July 2012. We enrolled patients with tendinopathy or insertional tendinopathy at clinical examination and US, older than 18 years, hemodynamically stable, with Hb values greater than 10g/dl, count of platelets greater than 150/μl, unresponsive to traditional treatments including FANS therapy and steroid injections, with no history of rheumatic disorders neither previous surgery to the affected tendons. Patients with platelet dysfunction syndrome, hypofibrinogenemia, systemic or local infection at the site of injection, cancer history, neurological or psychiatric disorders were excluded.
Patients who had assumed local or systemic non-steroidal antiinflammatory drugs (NSAIDS) or steroids 2 weeks before the procedure were also excluded. 156 patients were considered eligible for the study. Medical records and score schedules were all examined before treatment and at an average follow up of 1 year. We included only patients who had undergone at least 3 injections of PRP for management of tendinopathy or insertional tendinopathy of all the tendons. At the end, 139 patients were enrolled in the study.
PRP preparation
Eight milliliters of whole blood of the patient were collected in a RegenLab THT tube® containing a special gel. Once the tube had been centrifuged for 9 minutes at 3100 rpm, the red blood cells were separated from the Platelet Rich Plasma. Then, the PRP is aspirated with an 18-gauge needle and injected. Adverse effects were recorded. Patients were advised not to undergo surgical procedures in the followup period (12 months); painkillers were allowed if necessary.
Patient’s recruitment and intervention modalities
Patients were divided into two groups: The Group A (31 patients) included patients with insertional tendinopathy and the Group B (108 patients) included patients with tendinopathy of the body of the tendon. Injections were undertaken in a standard manner in patients with Achilles and elbow tendinopathy, with plantar fasciopathy and greater trochanteric pain syndrome. Specifically, once the site of pain had been identified at ultrasound (US), the skin was scrubbed and a needle was introduced perpendicularly to the axis of the probe, parallel to the tendon, along its side, within the paratenon. Taking care not to enter the body of the tendon, PRP was injected [15]. Injections to rotator cuff tendons were performed through the sub acromial space with the forearm in internal rotation, and the needle perpendicular to rotator cuff tendons [15]. All patients were been assessed for pain with VAS at baseline (before the treatment), 6 months, 1 year and 3 years after treatment. Specific scores were also administered. Specifically, University of California Los Angeles (UCLA) score was used for patients with rotator cuff disorders, the Harris Hip score for patients with greater trochanteric pain syndrome, the Quick Disability of Arm Shoulder and Hand (Quick DASH) for patients with elbow tendinopathy, the VISA-A score for patients with Achilles tendinopathy, and the American Foot and Ankle Score (AOFAS) for patients with plantar fasciopathy. Data about comorbidities and concomitant therapies were also recorded. After injection, patients were instructed to apply ice over the site of injection and avoiding stresses and heavy loads.
Statistical analysis
Blinded statistical analysis was performed. A paired t-test was used for intra-group comparison of VAS and specific scores at different follow-ups; unpaired T-test was used to compare inter group VAS values. The difference was significant when p was lower than 0.05. Mean, range, and 95% confidence intervals (C.I.) were also calculated. The version 16.0.1 of the SPSS software (SPSS Inc., Chicago, Illinois) was used.
Results
Medical records of 156 patients who had undergone PRP injections for different tendinopathies were screened. Of 139 patients enrolled in the study (76 males and 63 females), treated at an average age of 54 years (range 27-65), 31 patients were allocated in the Group A, 108 were included in the Group B. In the Group A, 11 patients were managed for elbow tendinopathy, 10 for greater trochanteric pain syndrome, and 10 for plantar fasciopathy. In the Group B, 93 underwent PRP injections for management of rotator cuff disease and 15 for mid-portion tendinopathy of the Achilles tendon. Flow diagram of patient’s selection process is reported in Figure 1.
Pain
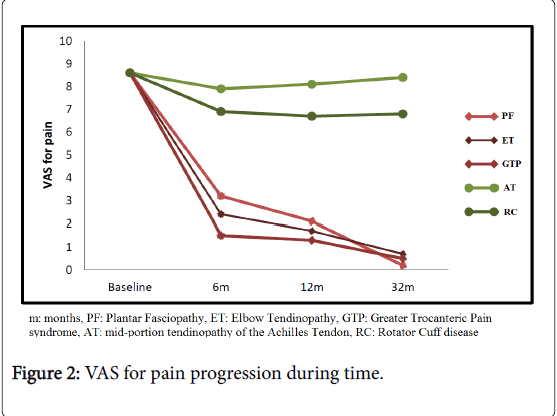
At baseline the overall average VAS was 8.6 (range 8.0-9.1). At 6 months, in the Group A, VAS values were significantly improved (p<0.05). Specifically, the average VAS was 3.2 in patients with plantar fasciopathy (range 1-4, 95% C.I. 1.5-2.4), 2.4 in those elbow tendinopathy (range 1-5, 95% C.I. 0.9-2.9), and 1.5 in those with greater trochanteric pain syndrome (range 0-2, 95% C.I. 1.7-2.2). In the Group B, at 6 months, the VAS improvement was not significant (p>0.05). The average VAS was 7.9 (range 6-9, 95% C.I. 0.2-3.7) in patients with Achilles tendinopathy, and 6.9 (range 5-8, 95% C.I. 0.7-1.8) in those with rotator cuff disease. At 1 year follow-up, in the Group A, the average VAS was 2.1 (range 0-6, 95% C.I. 1.8-2.2) in patients with plantar fasciopathy, 1.7 (range 1-3, 95% C.I. 1.3-2.7) in those with elbow tendinopathy, and 1.3 (range 1-3, 95% C.I. 1.8-2.2) in patients with greater trochanteric pain syndrome. At 1 year, in the Group B, the VAS value averaged 8.1 (range 4-10, 95% C.I. 1.3-2.6) in patients managed for Achilles tendinopathy and 6.7 (range 5-8, 95% C.I. 1.8-1.9) in those managed for rotator cuff disease. Therefore, the inter-group differences were statistically significant (p<0.001). At 3 years, in the Group A, the average VAS was 0.2 (range 0-3, 95% C.I. 1.9-2.0) in patients with plantar fasciopathy, 0.7 (range 0-2, 95% C.I. 1.8-2.1) in those with elbow tendinopathy, and 0.5 (range 0-2, 95% C.I. 1.9-2.0) in patients with greater trochanteric pain syndrome. In the Group B, the VAS value averaged 8.4 (range 6-9, 95% C.I. 0.8-3.1) in patients with Achilles tendinopathy and 6.8 (range 5-9, 95% C.I. 1.7-3.1) in those with rotator cuff disease. At 3 years, the inter-group differences were statistically significant (p<0.001; Figure 2).
Specific outcome scores
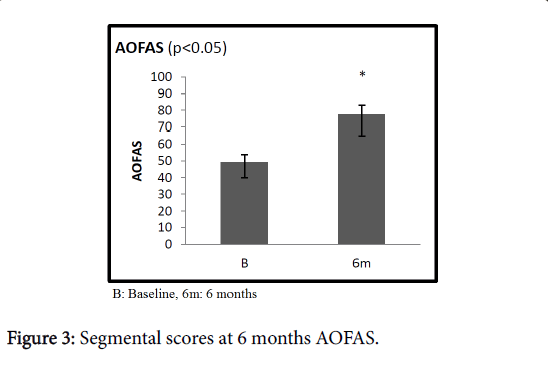
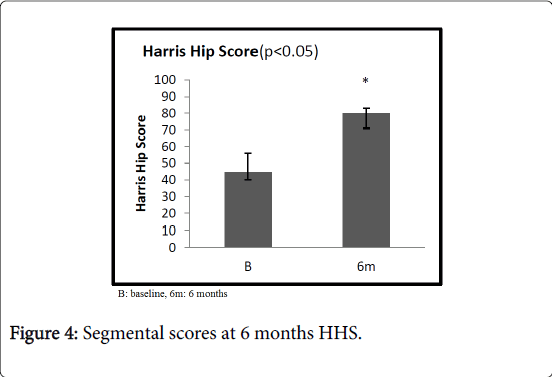
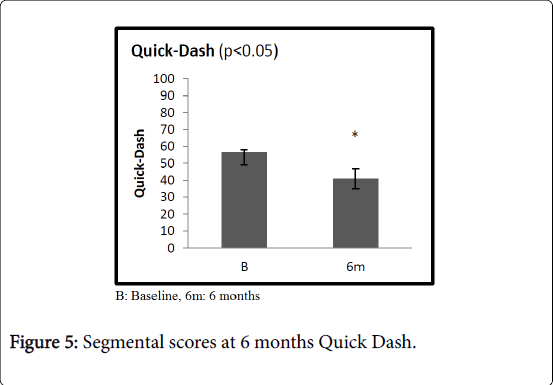
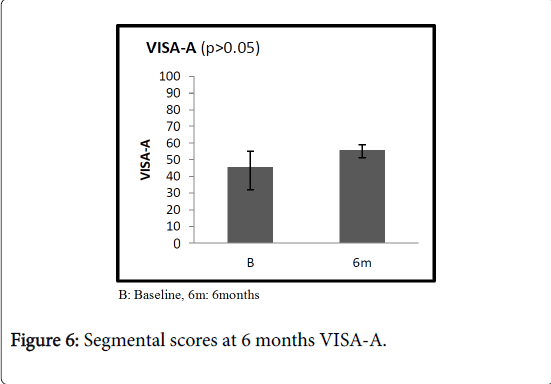
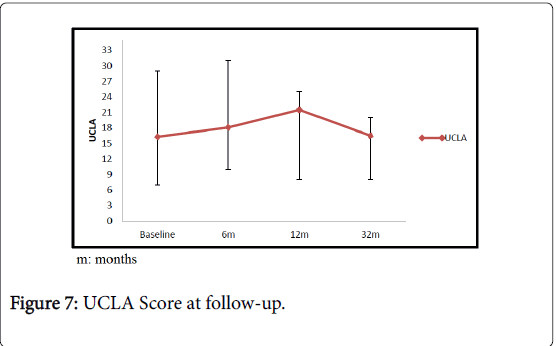
At 6 months, Specific scores were significantly better than at baseline. Specifically, in patients with plantar fasciopathy, the AOFAS score improved from an average value of 49.3 (range 40-53) to 77.4 (range 65-83, 95% C.I. 5.6-7.8) (p<0.05); the average Quick DASH improved from 56.6 (range 49-58) to 40.9 (range 35-47, 95% C.I. 6.9-12.7) (p<0.05) in patients with elbow tendinopathy; the average Harris Hip score improved from 44.7 (range 40-56) to 79.8 (range 71-83, 95% C.I. 13.2-16.8) (p<0.05) in patients with greater trochanteric pain syndrome. In the Group B, at 6 months, the average UCLA score was slightly increased compared to the average value at baseline, increasing from 16.3 (range 7-29) to 18.1 (range 10-31, 95% C.I. 4.6-16.9) (p>0.05) in patients with rotator cuff disease. Similarly, the average VISA-A score improved from 45.6 (range 32-55) at baseline to 55.6 (range 51-59, 95% C.I. 8.8-13.6) (p>0.05) at 6-month follow-up (Figures 3-6). At one year, the UCLA score averaged 21.5 (range 8-25, 95% C.I. 3.8-9.5); it decreased to 16.5 (range 8-20, 95% C.I. 3.5-7.8) at 3 years (Figure 7).
Discussion
The use of PRP has been widely shown in patients with cartilage lesions, but there is no consensus on its effectiveness in terms of cartilage repair. On the other hand, the role of PRP in the field of tendon healing as management of tendinopathy is still controversial. According to the literature, proper guidelines should be defined when referring to the application of PRP, and well-designed clinical trials are needed [3]. Even though many studies have reported outcomes after application of PRP in tendinopathy, this is the first study showing that PRP injections provide different outcomes in relationship to the tendon involved and the site injected. To the best of our knowledge, this is the first study that compares outcomes of patients who had PRP injections for tendinopathy of the body of the tendon vs insertional tendinopathy. Few studies have investigated the use of PRP for management of tendinopathies [3], focusing on patients with rotator cuff disease [9,16-19], elbow tendinopathy [12,20,21], and patellar tendinopathy [13,22,23]. Even though reported outcomes were satisfying, none of these studies clearly stated that PRP is effective, reasonably because the sample sizes were relatively small. In addition, the methods of preparation, the number of PRP injections, and the quantity of the product have not been standardized yet. Different preparations of PRP are available. Specifically, the L-PRP is used for injections, the PRP fibrin matrix is used intra-operatively, during arthroscopic procedures. The fact that different methods of preparation of the whole blood have been proposed induces many bias, making it difficult to compare available data, and perform metaanalysis on outcomes after PRP administration. The steps of administration of PRP should be standardized to allow the surgeon to define the indications to these products, the quantity of PRP to inject, and the time of injection. These injections should be performed under ultrasound guidance. In the present study, clinical and functional outcomes were significantly different between patients with tendinopathy of the body of the tendon and those with insertional tendinopathy. Pain improved at the first follow-up in both groups, not significantly in patients with tendinopathy of the body of the tendon. On the other hand, no recurrences occurred during the follow-up period, showing that PRP injections relieve pain and are a viable option of management of tendinopathy. It would be interesting to extend this study to a population of athletes, considering the return to sport. This study has some limitations: the number of patients is small to draw definitive conclusions on the effectiveness of PRP in tendinopathy, there is no control group of patients, and results are heterogeneous as tendinopathies affected different districts. In addition, US was used to inject superficial tendons, not to perform sub acromial injections, and no imaging methods were used to assess the tendon healing. On the other hand, as strengths of the study, a standardized Regen lab method produced by a specialist in hematology in a specialized Transfusion Medicine and Cellular Therapy Centre has been used to prepare PRP products, the same orthopedic surgeon performed all the injections, and an independent investigator blinded to the site of tendinopathy and number of injections collected data collection and performed the statistical analysis. As growing point for the future, given the concern on the PRP formulation, future studies should compare different formulations of PRP.
Conclusion
This is a pilot study comparing outcomes after injection of PRP for management of insertional tendinopathy and tendinopathies of the main body of the tendon, with better results in patients with insertional tendinopathy.
References
- Papalia R, Moro L, Franceschi F, Albo E, D'Adamio S, et al. (2013) Endothelial dysfunction and tendinopathy: how far have we come? MusculoskeletSurg 97:199-209.
- Notarnicola A, Maccagnano G, Tafuri S, Fiore A, Margiotta C, et al. (2015) Prognostic factors of extracorporeal shock wave therapy for tendinopathies. Musculoskeletal surgery.
- Andia I, Maffulli N (2015) Muscle and tendon injuries: the role of biological interventions to promote and assist healing and recovery. Arthroscopy 31:999-1015.
- Di Matteo B, Filardo G, Kon E, Marcacci M (2015) Platelet-rich plasma: evidence for the treatment of patellar and Achilles tendinopathy--a systematic review. MusculoskeletSurg 99:1-9.
- Lynch SE, Nixon JC, Colvin RB, Antoniades HN (1987) Role of platelet-derived growth factor in wound healing: synergistic effects with other growth factors. ProcNatlAcadSci USA 84:7696-7700.
- Anitua E, Sanchez M, Nurden AT, Zalduendo M, de la Fuente M, et al. (2006) Autologous fibrin matrices: a potential source of biological mediators that modulate tendon cell activities. J Biomed Mater Res A 77:285-293.
- de Mos M, Koevoet W, van Schie HT, Kops N, Jahr H, et al. (2009) In vitro model to study chondrogenic differentiation in tendinopathy. Am J Sports Med 37: 1214-1222.
- Papalia R, Franceschi F, Zampogna B, D'Adamio S, Maffulli N, Denaro V (2013) Augmentation techniques for rotator cuff repair. Br Med Bull 105: 107-138.
- Randelli P, Arrigoni P, Ragone V, Aliprandi A, Cabitza P (2011) Platelet rich plasma in arthroscopic rotator cuff repair: a prospective RCT study, 2-year follow-up. J Shoulder Elbow Surg 20: 518-528.
- Randelli P, Spennacchio P, Ragone V, Arrigoni P, Casella A, et al. (2012) Complications associated with arthroscopic rotator cuff repair: a literature review. MusculoskeletSurg 96:9-16.
- Mishra A, Pavelko T (2006) Treatment of chronic elbow tendinosis with buffered platelet-rich plasma. Am J Sports Med 34: 1774-1778.
- Peerbooms JC, Sluimer J, Bruijn DJ, Gosens T (2010) Positive effect of an autologous platelet concentrate in lateral epicondylitis in a double-blind randomized controlled trial: platelet-rich plasma versus corticosteroid injection with a 1-year follow-up. Am J Sports Med 38: 255-262.
- Filardo G, Kon E, Della Villa S, Vincentelli F, Fornasari PM, et al. (2010) Use of platelet-rich plasma for the treatment of refractory jumper's knee. IntOrthop 34:909-915.
- Kon E, Filardo G, Delcogliano M, Presti ML, Russo A, et al. (2009) Platelet-rich plasma: new clinical application: a pilot study for treatment of jumper's knee. Injury 40 :598-603.
- Saunders S, Longworth S (2006) Injection techniques in Ortopaedic and sports medicine. 3rd edn. Elsevier, Philadelphia.
- Barber FA, Hrnack SA, Snyder SJ, Hapa O (2011) Rotator cuff repair healing influenced by platelet-rich plasma construct augmentation. Arthroscopy 27:1029-1035.
- Bergeson AG, Tashjian RZ, Greis PE, Crim J, Stoddard GJ, et al. (2012) Effects of platelet-rich fibrin matrix on repair integrity of at-risk rotator cuff tears. Am J Sports Med 40: 286-293.
- Castricini R, Longo UG, De Benedetto M, Panfoli N, Pirani P, et al. (2011) Platelet-rich plasma augmentation for arthroscopic rotator cuff repair: a randomized controlled trial. Am J Sports Med 39:258-265.
- Jo CH, Kim JE, Yoon KS, Lee JH, Kang SB, et al. (2011) Does platelet-rich plasma accelerate recovery after rotator cuff repair? A prospective cohort study. Am J Sports Med 39:2082-2090.
- Gosens T, Peerbooms JC, van Laar W, den Oudsten BL (2011) Ongoing positive effect of platelet-rich plasma versus corticosteroid injection in lateral epicondylitis: a double-blind randomized controlled trial with 2-year follow-up. Am J Sports Med 39:1200-1208
- Krogh TP, Fredberg U, Stengaard-Pedersen K, Christensen R, Jensen P, et al. (2013) Treatment of lateral epicondylitis with platelet-rich plasma, glucocorticoid, or saline: a randomized, double-blind, placebo-controlled trial. The American journal of sports medicine 41:625-635.
- Charousset C, Zaoui A, Bellaiche L, Bouyer B (2014) Are multiple platelet-rich plasma injections useful for treatment of chronic patellar tendinopathy in athletes? a prospective study. Am J Sports Med 42:906-911.
- Dragoo JL, Wasterlain AS, Braun HJ, Nead KT (2014) Platelet-rich plasma as a treatment for patellar tendinopathy: a double-blind, randomized controlled trial. Am J Sports Med 42:610-618.
Relevant Topics
- Acupuncture
- Acute Pain
- Analgesics
- Anesthesia
- Arthroscopy
- Chronic Back Pain
- Chronic Pain
- Hypnosis
- Low Back Pain
- Meditation
- Musculoskeletal pain
- Natural Pain Relievers
- Nociceptive Pain
- Opioid
- Orthopedics
- Pain and Mental Health
- Pain killer drugs
- Pain Mechanisms and Pathophysiology
- Pain Medication
- Pain Medicine
- Pain Relief and Traditional Medicine
- Pain Sensation
- Pain Tolerance
- Post-Operative Pain
- Reaction to Pain
Recommended Journals
Article Tools
Article Usage
- Total views: 4776
- [From(publication date):
May-2017 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 3830
- PDF downloads : 946