Research Article Open Access
Effect of Cytokinins on In vitro seed Germination and Changes in Chlorophyll and Soluble Protein Contents of Teak (Tectona grandis L.)
| Muhammad Akram* and Faheem Aftab | |
| Department of Botany, University of the Punjab, Lahore-54590, Pakistan | |
| Corresponding Author : | Muhammad Akram Department of Botany, University of the Punjab, Lahore-54590, Pakistan E-mail: m.akramks@gmail.com |
| Received May 14, 2015; Accepted June 01, 2015; Published June 08, 2015 | |
| Citation: Akram M, Aftab F (2015) Effect of Cytokinins on In vitro seed Germination and Changes in Chlorophyll and Soluble Protein Contents of Teak (Tectona grandis L.). Biochem Physiol 4:166. doi: 10.4172/2168-9652.1000166 | |
| Copyright: © 2015 Akram M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
| Related article at Pubmed, Scholar Google | |
Visit for more related articles at Biochemistry & Physiology: Open Access
Abstract
Teak (Tectona grandis L.) is amongst the precious tropical trees produces luxurious wood items and metabolites of great medicinal value. However, teak growth is hampered due to high dormancy and poor viability of the seeds under the normal field conditions. Here we investigated the effect of various cytokinins on seed germination and growth under In vitro and ex vitro conditions of seedlings. Completely randomized design was used and data were analyzed to ascertain whether or not the tested cytokinins can improve seed germination and growth of the teak plants. Seeds were cultured on MS medium supplemented with various concentrations (0.08, 0.22, 0.35, 0.80, 2.20 or 3.50 μM) of adenine sulphate (ADS), N6-benzyleadenine (BA), kinetin (KIN), thidiazuron (TDZ), and zeatin (ZEA) under In vitro conditions for 40 days. Highest (100%) germination was obtained at 0.22 μM BA with 4.41 cm shoot and 4.67 cm root lengths as well as 75.32% highest seedling’s survival was observed in glasshouse. TDZ inhibited seedling’s growth and induced hyperhydricity (16.36–35.36%). Improved growth of seedlings in both conditions may be linked with enhanced production of chlorophyll (Chl) both In vitro (1.86 Chl a, 1.30 Chl b mg/g FW) and in acclimatized seedlings (1.91 Chl a, 1.70 Chl b mg/g FW) at 0.22 μM BA. The same concentration of BA also produced highest soluble proteins in In vitro (7.52 mg/g FW) and acclimatized seedlings (8.88 mg/g FW). Under specific tissue culture conditions, seed germination and subsequent growth of teak seedlings can be improved with BA.
| Keywords |
| Chlorophyll; Hyperhydricity; In vitro seedlings; N6- benzyleadenine; Teak |
| Introduction |
| Teak (Tectona grandis L.) is a well-known timber producing deciduous tree belonging to family verbenaceae. It grows well in tropical parts of Myanmar, India, Laos and Thailand and is considered among top five tropical hardwood species in terms of area established worldwide [1]. It plays an important role in the timber industry due to its luxury applications. For example, it is used for making doors, window pans, plywood, furniture as well as water boats and ships. It provides highly demanding wood that makes it most expensive in the timber industry of the world. Teakwood is extremely resistant against termite and fungal attack due to the presence of various repellents, for example, naphthoquinones is reported to have antifungal activity [2]. Moreover, teakwood also contains lapachol [3] which has recently been demonstrated as an anticancer agent [4]. Endogenous plant hormones play fundamental role in the growth and development throughout the life cycle of plants. The level of such hormones decreases with the age of plant tissues [5]. The quiescent zone of seeds is a target site of phytohormones for germination and plant growth [6]. In vitro seed germination depends upon the physiology and viability of seeds [7]. Our previous experience on teak (Tectona grandis L.) showed 10- 15% germination under field conditions which further decreases with the passage of time during storage. This may be due to the lack of some important growth factors in the seeds; exogenous cytokinins may substitute the lost features of seeds for enhanced germination when cultured In vitro [6]. Cytokinins influence various physiological processes essential for In vitro germination, growth and development of plant [6,8]. Adenine derivative cytokinin N6-Benzyladenine (BA) promotes the growth of In vitro shoot as well as improved seed germination in different plants such as Citrus reticulata [9] and Lotus corniculatus [10] by enhancing protein and photosynthetic pigments [11]. |
| Hyperhydricity is a water logging condition in plants [12] caused by various factors including gelling agent and nitrogenous compounds. Various cytokinins and their concentrations are also responsible to induce this disorder [8,13]. Under such conditions, developing shoots losechlorophyll and soluble protein contents which may eventually prevent the plant growth [14]. Prevalence of hyperhydricity is more frequent when phenylurea derivative TDZ was used in the medium [13]. |
| Renewed interest in teak wood around the world in the recent years has received attention of researchers for its multiplication using diverse means to meet the requirements of the timber industry. However, limited number of seed production and recalcitrant nature of mature tissues are major bottlenecks for its rapid multiplication. Plant tissue culture offers a solution by direct manipulation of seeds or vegetative tissues under In vitro conditions. The use of cytokinins for both In vitro seed germination enhancement and changes in growth is an interesting research aspect [7]. There is no previous information vis-à-vis effect of cytokinins on seed germination and seedling growth of teak in In vitro developing and acclimatized seedlings as well as transplantation in glasshouse. The aim of the present study was therefore to elucidate the effect of cytokinins on In vitro seed germination and growth of teak seedlings under both In vitro and ex vitro glasshouse conditions. |
| Materials and Methods |
| Plant material |
| Mature dried drupes were collected from Changa Manga Forest, Kasur District, Punjab, Pakistan (31°05′N 73°58′E). Locular seeds were extracted out by breaking down both the outer exocarp as well as the inner hard bony endocarp by carefully tapping with a suitable iron hammer. Extracted seeds from various trees from the above location were pooled and used in germination experiments. |
| Culture conditions |
| Fleshy locular seeds were surface sterilized with 6% (v/v) solution of Sodium hypochlorite (NaOCl) containing 0.1% Tween-20 for 10 min followed by at least five rinsing with autoclaved distilled water under aseptic condition. The pH of all culture media was adjusted to 5.8 solidified with 0.8% agar (Agar Technical, Oxoid, UK) and autoclaved at 121 °C at 104 kPa for 15 min. All cultures were incubated in growth room under 16 h photoperiod (35 ± 5 μmol m-1s-1) fitted with white fluorescent tube lights (Philips, 40W, Pakistan) at 25 ± 2 °C with 44 % relative humidity of growth room. |
| In vitro seed germination and growth of seedlings: Healthy seeds were inoculated in culture vessels (25 x 150 mm, Pyrex, Germany) containing 10 ml agar-solidified MS [15] medium with 3% sucrose supplemented with Sigma-Aldrich grade adenine sulphate (ADS), N6- benzyleadenine (BA), kinetin (KIN), zeatin (ZEA), and thidiazuron (TDZ) at various levels (0.08,0.22,0.35,0.8,2.2 or 3.5μM). Medium without cytokinins was considered as control. The data for percent seed germination were recorded after 40 days of initial culture. Shoot as well as root length (cm) was also recorded on the same day of harvesting. |
| Hyperhydricity |
| The hyperhydricity was calculated according to Kadota and Niimi (2003) by using the following formula: |
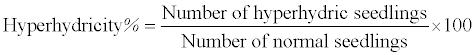 |
| The percentage of hyperhydricity (vitreous, chlorophyll deficient water logging shoots) was recorded on the same day (40) of harvesting. Green shoot with normal growth was considered as morphologically normal seedlings. |
| Acclimatization and survival of seedlings |
| Seedlings were immersed in 1% (v/v) fungicide (Dithane M-45, Dow AgroSciences, USA) for 30 sec and planted in poly-cups filled with peat moss + sand + soil (1:1:1) in glasshouse at natural day/night low light conditions (67 μmol m-1s-1) at 27 ± 2 °C with 60 ± 5% relative humidity. The survival percentage of seedlings was calculated after 40 days of transfer in the glasshouse by the following formula. |
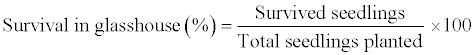 |
| Biochemical analysis (determination of soluble proteins) |
| A sample of 0.1 g leaves from 40-day old In vitro and acclimatized seedlings grown on different concentrations of each cytokinin were collected for extraction and determination of total soluble proteins (TSP) following the method of Premkumar et al. [16]. Leaf tissues were crushed in pestle and mortar in liquid nitrogen to a fine powder and homogenized in extraction buffer and centrifuged at 13,000 rpm for 15 minutes. The supernatant was separated and TSP were determined spectrophotometrically (HITACHI U-1100) at 750 nm. After the extraction, quantitative analysis of TSP was performed according to the method of Bradford [17]. |
| Physiological analysis (determination of chlorophyll contents) |
| For determination of the chlorophyll a (Chl a) and chlorophyll b (Chl b), plant material as described above was collected and ground in pestle and mortar to a fine pulp in 80% acetone and centrifuged (Sorvall® RC-5B) at 5,000 rpm for 15 min. The supernatant was collected and concentration of Chl contents was determined with spectrophotometer on the basis of mg/g fresh weight (FW) of tissue according to Premkumar et al. [16]. |
| Experimental design and data analysis |
| Completely randomized design was used of 5 cytokinins with 7 different concentrations for In vitro seed germination and biochemical analyses. Thirty replicates per treatment were used and the experiment was repeated thrice. Data were transformed where necessary and statistical analyses were carried out by Analysis of Variance (ANOVA). Standard error (±SE) of the means was calculated and Duncan’s multiple range test (DMRT) at p<0.05 was performed to find out mean difference by using SPSS release 16.0. |
| Results |
| In vitro seed germination and growth of seedlings |
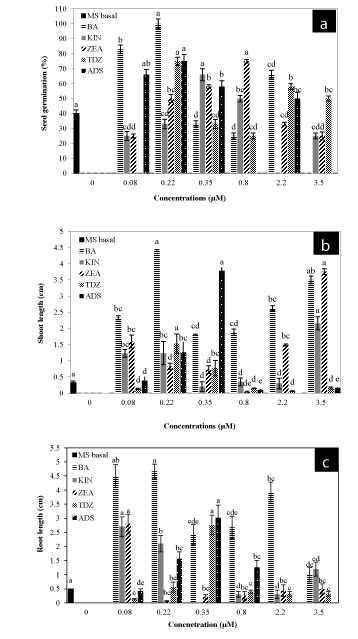
| Swelling on seeds started after 5 days and protrusion of radical commenced at day 10. The frequency of seed germination was highest with different cytokinins than control. Figure 1a demonstrated that BA significantly improved germination than other cytokinins. Low concentrations (0.08, 0.22 or 0.35 μM) of all cytokinins were more effective as compared to their higher levels (2.20 or 3.50 μM) for improved growth of seedlings. |
| Highest (100%) germination was obtained at 0.22 μM BA followed by 75% with ADS (0.22 μM) after 40 days of culture (Figure 1a). Similar germination frequency (75%) was also obtained with TDZ (0.22 μM) as well as with ZEA at 0.8 μM than control (40%). The non-germinable seeds (if any) became necrotic. |
| Generally, cytokinins also improved shoot and root lengths of seedlings (Figure 1b, c). BA at 0.22 μM produced highest shoot length (4.41 cm) and root length (4.67 cm) (Figure 2a). This was followed by 3.78 cm shoot length and 3.02 cm root length at 0.35 μM ADS after the same culture period. KIN, TDZ and ZEA were least effective in terms of shoot-root growth. |
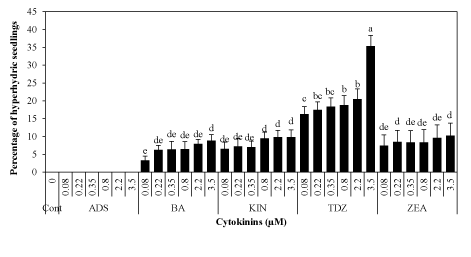
| Hyperhydricity and survival of plantlets |
| Hyperhydric seedlings were observed on each cytokinin of different levels (Figure 3). Morphologically such seedlings looked normal, but growth was stunted due to complete loss of chlorophyll (Figure 2b). Hyperhydricity occurred when seedlings acquired favorable root-toshoot growth with BA and ZEA (Figure 2b). However, TDZ induced hyperhydricity at early stages of seedling development (15 days) and strongly inhibited the growth of hypocotyl to epicotyl. The rate of hyperhydricity was highest (35.36%) at 3.50 μM TDZ whereas frequency of such anomaly was reduced to 10.25% at the same level of ZEA. Two to three hyperhydric seedlings were observed on each treatment of BA and KIN. However, hyperhydricity was not observed with ADS. |
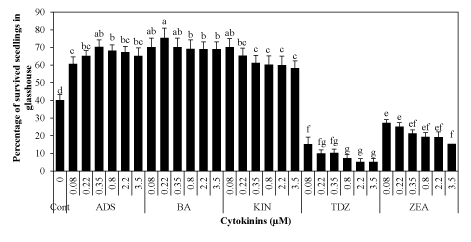
| Seedling’s survival was highest (75.32%) pre-cultured at 0.22 μM BA followed by 70.30% at 0.35 μM ADS and 70.22% at 0.08 μM KIN as compared to control (40.21%) after 40 days in glasshouse (Figure 4). Generally, survival percentage of acclimatized seedlings was low with high concentrations of cytokinins. Seedlings grown on BA were healthy and vigorous with large leaves and stout stems irrespective of seedlings on TDZ hardly survived or very low survival frequency (5.21 to 10.22%) was obtained (Figure 2c). |
| Chlorophyll and TSP contents |
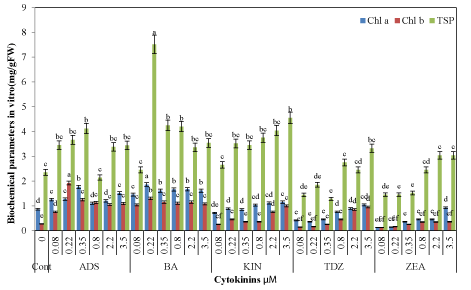
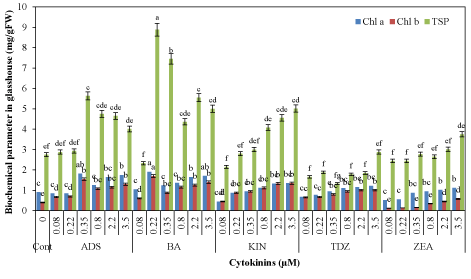
| Generally, the amount of photosynthetic pigments was significantly improved by increasing the concentration of each cytokinin In vitro (Figure 5) as well as in glasshouse growing plants (Figure 6). Both Chl a (1.86 mg/g FW) and Chl b (1.30 mg/g FW) were highest at 0.22 μM BA from In vitro seedlings as compared to control (Chl a 0.86, Chl b 0.27 mg/g FW). Seedlings on TDZ and ZEA had least amount of photosynthetic pigments. Similarly, acclimated seedlings for 40 days in the glasshouse also had more chlorophyll pre-cultured on all cytokinins especially BA and ADS (Figure 4). Highest amount of Chl a (1.91 mg/g FW) and Chl b (1.70 mg/g FW) were recorded from plantlets previously grown at 0.22 μM BA as compared to control (Chl a 0.91 and Chl b 0.40 mg/g FW). However, TDZ and ZEA were least effective produced chlorophyll contents in the acclimatized seedlings. |
| The amount of TSP was highest in acclimatized as compared to In vitro seedlings (Figure 6). Generally, amount of TSP was significantly improved by increasing the concentration of all cytokinins. In case of In vitro seedlings, BA at 0.22 μM produced highest TSP contents (7.52 mg/g FW) as compared to other cytokinins and control (2.35 mg/g FW) followed by 4.12 mg/g FW TSP with ADS (0.35 μM). TSP contents in acclimatized plantlets were also highest at 0.22 μM BA (8.88 mg/g FW) followed by 5.63 mg/g FW at 0.35 μM ADS. Thiourea derivative TDZ and ZEA at 3.50 μM were least effective for production of TSP, i.e., 2.88 and 3.75 mg/g FW, respectively than other cytokinins, albeit higher than control (2.75 mg/g FW). |
| Discussion |
| In vitro seed germination is an important aspect for axenic seedling production under the influence of various plant growth regulators (PGRs) for subsequent micropropagation and organogenesis of woody and non-woody plants. The present investigation clearly showed that germination of teak seeds was significantly improved with different cytokinins under In vitro conditions. Our previous report demonstrated the likelihood of teak seed germination on MS basal medium [18]. In the present study, we achieved improved rate of germination with cytokinins. |
| Significant relationship exists between endogenous hormones and their action on the targeted developmental seed germination loci. Exogenous application of cytokinins plays significant role by stimulating specific metabolic activity for enhanced germination and seedling growth [6,19]. In the present study, BA was most effective as compared to ADS, ZEA, KIN, and TDZ for In vitro seed germination. Existing information vis-à-vis the use of cytokinins added in tissue culture medium for In vitro seed germination of trees is scanty. However, Bhattacharya and Khuspe [20] obtained 70% In vitro seed germination with BA in Carica papaya. Stewart and Kane [21] reported 47.2% In vitroseed germination in Habenaria macroceratitis with KIN (1 μM). Dutra et al. [22] and Samuel et al. [23] also demonstrated the similar results. In contrast, sole use of BA (10 μM) accelerated germination of Digitalis purpurea up to 65.5%, however, when indole-3-butyric acid at 10 μM was used along with BA (10 μM) germination was significantly improved to 81% [18]. Similarly, sole use of BA was also less effective for In vitro seed germination of Campanulla species [24]. Such contrasting reports demonstrated that germination depends upon the genotype and uptake of BA in different plants [23]. In the present study, 100% germination was achieved with BA however its positive role for In vitro seed germination is not well understood. It is assumed that being derivative of nucleic acids, BA enhances germination by the stimulation of lipid metabolism [25]. Amongst the multiple activities of BA, germination and further development of seedlings is one of its key roles. Various analytical methods demonstrated the effectiveness of cytokinins and BA in plant metabolism for optimum seedling growth [25], and KIN is known to alleviate some endogenous growth stresses [6,26]. |
| Hyperhydricity causes stunted growth, brittle and translucent appearance of In vitro grown plants [13]. In the present study, hyperhydricity occurred on all treatments lacking chlorophyll and stunted growth suggested that survival of seedlings was greatly influenced by hyperhydricity. Cytokinins have been reported to occur hyperhydricity during In vitro culture system [8,12,13]. Furthermore, hyperhyrdicity is significantly influenced by using different cytokinins and their concentrations similar to the observations of Ivanova and Van Staden [8] in Aloe spp. We found TDZ that triggered extensive hyperhydricity than adenine based cytokinins (ADS,BA,KIN) consistent with Kadota and Niimi [13] in pear cultivars. TDZ mediated hyperhydricity occurred having high cytokinin activity which has previously been reported in many other plants [8,12,27]. |
| It is well known fact that chlorophyll is responsible for synthesis of food for optimum growth and development of plants. In the present study, growth of In vitro as well as acclimatized seedlings in glasshouse was more vigorous with BA as compared to other cytokinins. We observed high amount of both photosynthetic pigments and TSP in In vitro (Ch a1.86,Chl b 1.30,TSP 7.52 mg/gFW) and acclimatized seedlings (Chl a1.91,Chl b 1.70,TSP 8.80 mg/gFW) by BA that might have been the reason for vigorous growth. During the acclimatization phase, most of the teak seedlings survived as affected by BA with the production of long and expanded leaves after 10 days of transplantation. Our results support the findings of Ron’zhina [28] who also reported BA as an extremely active cytokinin that may improve efficiency of photosynthetic machinery In vitro for better growth and acclimatization in Faba beans. High photosynthetic potential of In vitro cultured plants and production of high dry matter in acclimatized seedlings were favoured by KIN and BA [29]. Furthermore, BA stimulated the production of RNA and proteins. Within 2 days of initial culture, BA inhibited the amount and subsequently increased chlorophyll during seedlings growth period [30]. Vigorously growing plants maintain the optimum nutrient levels that may favour high chlorophyll activity in plants which may be linked with high amount of protein contents. Arigita et al. [31] reported that BA accumulates itself at the basal regions of developing shoots of Kiwi fruit and Spathiphyllum floribundum plants [32] confirming its retention site in shoot making the plantlet stout to withstand low water contents and other ex vitro stresses [33]. The present study clearly demonstrated that cytokinins improved the germination of teak seeds under In vitro conditions and their growth and survival in glasshouse was more pronounced with BA especially at 0.22 μM. TDZ was responsible for stunted growth and prevalence of hyperhydricity at all concentrations. Chl a, b and TSP were highest in individuals taken from BA that demonstrated the effectiveness of BA for improved growth during the acclimatization phase which may favor speedy production of planting stock in a short period of time. |
References
|
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Analytical Biochemistry
- Applied Biochemistry
- Carbohydrate Biochemistry
- Cellular Biochemistry
- Clinical_Biochemistry
- Comparative Biochemistry
- Environmental Biochemistry
- Forensic Biochemistry
- Lipid Biochemistry
- Medical_Biochemistry
- Metabolomics
- Nutritional Biochemistry
- Pesticide Biochemistry
- Process Biochemistry
- Protein_Biochemistry
- Single-Cell Biochemistry
- Soil_Biochemistry
Recommended Journals
- Biosensor Journals
- Cellular Biology Journal
- Journal of Biochemistry and Microbial Toxicology
- Journal of Biochemistry and Cell Biology
- Journal of Biological and Medical Sciences
- Journal of Cell Biology & Immunology
- Journal of Cellular and Molecular Pharmacology
- Journal of Chemical Biology & Therapeutics
- Journal of Phytochemicistry And Biochemistry
Article Tools
Article Usage
- Total views: 17067
- [From(publication date):
September-2015 - Aug 18, 2025] - Breakdown by view type
- HTML page views : 12133
- PDF downloads : 4934
