Research Article Open Access
Effect of Exposure to Electromagnetic Fields (Emfs) on Monoamine Neurotransmitters of Newborn Rats
| SA Ismail, Rehab FM Ali*, Hassan HMM and Abd El-Rahman D | |
| Department of Biochemistry, Faculty of Agriculture, Cairo University, Egypt | |
| *Corresponding Author : | Rehab F.M. Ali Department of Biochemistry, Faculty of Agriculture Cairo University, 12613, Giza, Egypt Tel: +2-02- 37742600 Fax: +2-02- 37742600 E-mail: Malk_anany@yahoo.com |
| Received March 06, 2015; Accepted March 24, 2015; Published March 31, 2015 | |
| Citation: Ismail SA, Ali RFM, Hassan HMM, Abd El-Rahman D (2015) Effect of Exposure to Electromagnetic Fields (Emfs) on Monoamine Neurotransmitters of Newborn Rats. Biochem Physiol 4:156. doi:10.4172/2168-9652.1000156 | |
| Copyright: © 2015 Ismail SA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Biochemistry & Physiology: Open Access
Abstract
Background: The use of mobile phones is increasing day by day, and spending long time connected has aroused interest of possible interactions reflected on human body by radiofrequency electromagnetic radiation.
Purpose: In the current investigation pregnant female Albino rats were exposed to three mobile phone frequencies (900/1800/2100).
Materials and Methods: After delivery, the newborn rats were also subjected to the same condition of exposition to EMF. Histamine, dopamine, serotonin, melatonin, adrenaline, and noradrenaline in the brain wereassayed by highperformance liquid chromatography.
Results and Conclusions: No significant (p ≥ 0.05) changes in the level of monoamine neurotransmitters were shown for the control rats at different ages. Histamine, dopamine, adrenaline, and noradrenaline contents for the exposed animals were significantly (P ≤ 0.05) increased with increasing exposure time. However, exposure to electromagnetic field EMF caused significant decreases in the serotonin and melatonin content of rat brains.The results concluded that the exposure of new born rats to EMF may be sufficient to induce significant changes in the content of monoamine neurotransmitters of the exposed rats.
| Keywords |
| Electromagnetic field; Mobile; Neurotransmitters; Brain; Radiation |
| Introduction |
| Natural electromagnetic environment is necessary for life on the earth and, this environment has sharply changed because of introduction of the enormous and rising spectrum of manmade electromagnetic field (EMF). Radiofrequency electromagnetic radiation (RFR), a form of energy between 10 KHz-300 GHz in the electromagnetic spectrum, is used in wireless communication and emitted from antennae of mobile telephones (handys) and from cellular masts. |
| Mobile (cellular) telephony is based on two-way radio communication between a portable handset and the nearest basestation. Every base-station serves a cell, varying from hundreds of meters in extent in densely populated areas to kilometers in rural areas, and is connected both to the conventional land-line telephone network and, by tightly focused line-of-sight microwave links, to neighboring stations. As the user of a mobile phone moves from cell to cell, the call is transferred between base-stations without interruption [1]. Nowadays advances in cell phone communication have led people to take advantages of these technological achievements. It is estimated that the cell phone users in the world in 2009 is about 3 billion sets [2]. In nearly a third of the countries, the number of cell phones in use is greater than the number of people living in those countries [3]. Mobile technology has already been widely adopted around the world; its utilization is growing at a rapid rate, not just for interpersonal communication but as an important aspect of communication infrastructure for industries including finance, education, and marketing [4,5]. |
| The recent investigations indicate that the continuous using of the mobile phone for one hour caused hearing dysfunction [6]. Exposure to mobile phone radiation also decreases the preparatory slow potentials in certain regions of the brain [7], affects memory tasks [8] and increases in resting blood pressure during exposure to radio frequencies. Due to the close proximity of the mobile phone device to the head, the human brain is exposed to relatively high specific absorption rates (SARs) compared with the rest of the body [9]. |
| Mobile phones are frequently used in most parts of the world but most of us are not aware of its dangerous effects on human health. The mobile phone emits electromagnetic radiation which can affect the human brain. Therefore, the major aim of the current investigation was to evaluate the effect of exposure to EMF emitted from a mobile phone on monoamine neurotransmitters (Noradrenaline, adrenaline, histamine, serotonin, melatonin and dopamine) of Albino newborn rats. |
| Materials and Methods |
| Chemical reagents |
| Noradrenaline, adrenaline, histamine, serotonin, melatonin and dopamine were purchased from Sigma Chemical Co., Ltd (St. Louis, MO, USA). Perchloric acid was purchased from E. Merck Co. (Darmstadt, Germany). |
| Experimental animals |
| Pregnant female adult rats (Sprague Dawley strain), with an average weight of 204–236 g were used in the present investigation and were obtained from the animal house of Faculty of Veterinary Medicine, Cairo University. |
| The animal experiments were performed with the approval of the Ethics Committee forexperimental animals of the Faculty of Agriculture, Cairo University, Giza, Egypt, which followed the recommendations ofthe National Institutes of Health Guide for Care and Use of Laboratory Animals (Publication No.85-23, revised 1985). Six of pregnant female Albino rats were used for conducting the current investigation, one of them presents the control group and the remaining, five rats were exposed to three mobile (Nokia C6-01, SAR Specific Absorption Rate 1.00 W/kg, head) phone frequencies (900/1800/2100). The animals were exposed to 15 missed calls per day for pregnant period (21 day). Mobile phone was placed on the 15 cm distance of the cage of rats. The rats were housed in temperature-controlled rooms (25 ± 2°C) with constant humidity (55 ± 5%) and a 12 h/12h light/dark cycle prior to experimental protocols. The room equipped with timer for adjusting the cycle of light. All animals were allowed to drink water ad libitum. The animals were fed a basal diet. The basal diet was formulated according to (A.O.A.C., 2000). After delivery, 40 of infant rats were obtained; Newborn rats were also subjected to the same condition of exposition to EMF. Brain samples of newborn rats were taken on 1, 7, 14, 21 and 28 days after delivery. |
| Tissue preparation |
| Rats were killed by decapitation .Brains were removed within 90 seconds of death and were weighed and placed directly in a deepfreezer (−42°C) until further use. |
| Samples preparation |
| Brain samples were homogenized in 1.5 mL of 0.2 M ice-cold perchloric acid for 2 minutes and centrifuged at 14,000 rpm for 20 min at 4°C. One ml of the supernatant was added to 10 μl of the internal standard, 3, 4 dihydroxybenzyl amine (DBA). The mixture stored in a deep- freezer (−25°C) until further use. |
| Determination of monoamine neurotransmitters |
| Monoamine neurotransmitters were determined using HPLC system (1200 series). HPLC system equipped with autosapling injector, solvent degasser, quaternary HP pump and aUV detector was set at 204 nm (Hewlett-Packard, Palo Alto, CA, USA).The mobile phase consisted of (0.025MKH2 PO4 + 0.3 mM heptanes sulfonic acid in water (pH=3) andacetonitrile. The flow rate was 1.0 ml/min. Samples and mobile phases were filtrated through a 0.45 mm Millipore filter, type GV (Millipore, Bedford,MA, USA), prior to HPLC injection. Each sample was analyzed in triplicate. |
| Statistical analysis |
| Data are expressed as mean ± standard deviation (SD) of three replicates. The data were analyzed by analysis of variance (ANOVA) according to the procedures outlined by [10]. Results were processed by Excel (Microsoft Office 2007) and SPSS Version 18.0 (SPSS Inc., Chicago, IL, USA).Duncan’s multiple range test was used to determine the differences among samples. Significant levels were defined as probabilities of 0.05 or less. |
| Results and Discussion |
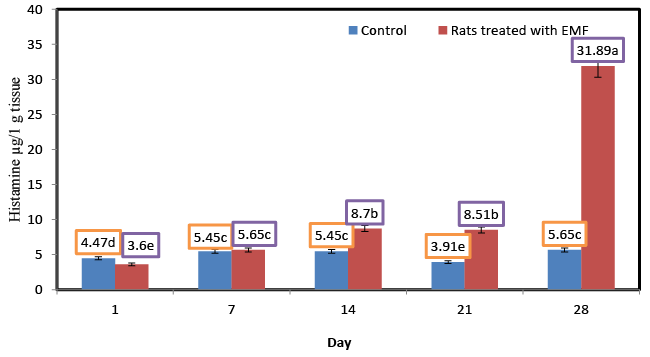
| Influence of exposure to EMF on brain histamine (μg/1 g tissue) of newborn rats |
| Brain histamine is implicated in brain homeostasis and control of several neuroendocrine functions. Histamine has an important role in the control of behavioral state, biological rhythms, body weight, energy metabolism, thermoregulation, fluid balance, stress and reproduction [11]. Figure 1 illustrates the changes in the levels of histamine for control rats and those treated with EMF. No significant (p ≥ 0.05) changes in the concentration of histamine were shown for the control ratsat different ages. Generally the levels of histamine in control rats were lower than those of rats treated with EMF. Significant (p ≤ 0.05) increases in the level of histamine were shown for rats treated with EMF. Histamine level of rats treated with electromagnetic field EMF for 14, 21 and 28 dayswere about 1.59,2.17 and 5.64 times as high as that in control rats at the same times . Histamine content of rats treated with electromagnetic field EMF increased significantly from 3.6 at the first day to 31.89 μg/1 g tissues at the end of the experiment.In facial skin samples of electrohypersensitive (EHS) persons,the most common finding is a profound increase of mastcells as monitored by various mast cell markers, such as histamine, chymase and tryptase [12]. Mast cells, when activated, release a wide range of mediators,among them histamine, which is involved in a variety ofbiological effects with clinical relevance, e.g., allergic hypersensitivity,itch, edema, local erythema, and many types of dermatoses [13]. Objective skin symptoms of EHS have been related to theincrease in mast cell counts and their degranulation, thereby causing the release of inflammatory substances such as histamine responsible for allergic hypersensitivity, sensation of itch and pain, edema, local erythema, and many kinds of dermatoses [13]. |
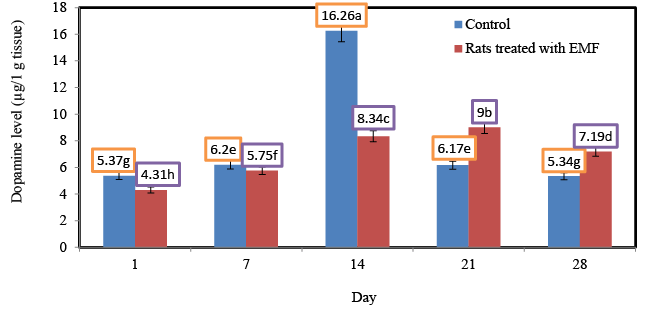
| Influence of exposure to EMF on brain dopamine (μg/1 g tissue) of newborn rats. |
| Results from lesioning and psychopharmacological experiments indicate a role of dopamine systems in behavior driven by rewards and in reward-based learning [14,15]. Figure 2 shows the changes in the levels of dopamine for control rats and rats treated with EMF. Generally the levels of dopamine in control rats werelower than those of rats treated with EMF. No significant (p ≥ 0.05) changes in the level of dopamine were shown for the control rats at different ages. Significant (p ≤ 0.05) increases in the level of dopamine were recorded for rats treated with EMF starting from the first day of the experiment and towards the end of the third week of the experiment. In the current study the highest levels (14.19; 16.72 μg/1 g tissue) of dopamine were shown for ratsexposed to electromagnetic field EMF for 14 and 21 days. Shin et al. [16,17] reported an increase in D-1 dopamine receptors and activity in the striatum of the rat after magnetic field exposure. Dopamine in the striatum is involved in Parkinson’s disease. It was reported that ELF magnetic fields potentiated morphineinduced decrease in D-2 dopamine receptors.It has been found that low-frequency electromagnetic field ELF-EMF exposure for 14 days (1 h daily) increases the rate of synthesis of dopamine and serotonin in rat frontal cortex as compared to a control [18]. These increases in the concentration of dopamine in rats exposed to EMF could be attributed to the stress-related effect of radiofrequency (RF-EMF) pulse exposure on dopaminergic MN9D cultures.The increase of intracellular nitric oxygen (NO) level after exposure was related to the activation of Ca/ CaM-dependent constitutive nitric oxide synthase. The CaM/NO/ cGMP signaling pathway activates a rapid response cascade that in turn influences EMF on Cam/NO pathways, which can modulate CNS response to inflammation and ischemia and interfere with the ability of the brain to restore post-traumatic functions [19]. Another mechanism to regulate neuronal function is calcium homeostasis, suggested to be a mediator of cell response to RF exposure [20]. In these respect, it was reported that ELF EMF-induced anxiety in the rat involved N-methyl- D-aspartate (NMDA) receptor in the brain. |
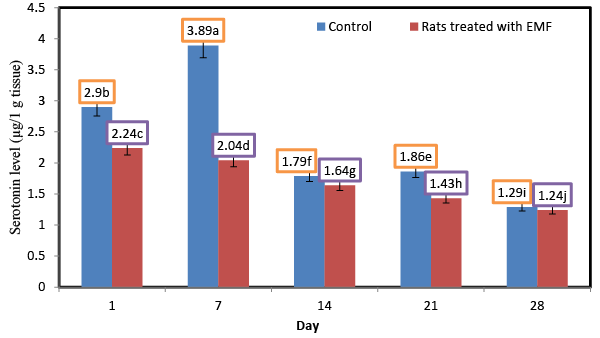
| Influence of exposure to EMF on brain serotonin (μg/1 g tissue) of newborn rats |
| Serotonin and other neurotransmitters are implicated at the dorsal root entry zone (DREZ) of the spinal cord where the afferents connect [21]. Figure 3 shows the changes in the levels of serotonin for control rats and rats treated with EMF.Serotonin values in control rats were higher than those of rats treated with EMF. No significant (p ≥ 0.05) changes in the level of serotonin were shown for the control rats at different ages. However, exposure to electromagnetic field EMF caused significant decrease in the serotonin content of rat brains.Serotonin level of rats treated with electromagnetic field EMF decreased from 2.43 at the first day to 0.88 μg/1 g tissue at the end of the end of the experiment. The lowest values (0.96; 0.88 μg/1 g tissue) of serotonin were observed for the rats whichtreated with electromagnetic field EMF for 3 and 4 weeks, respectively. The significant decrease in the level of inhibitory neurotransmitter serotonin (5-HT) recorded in the brain of irradiated rats might be attributed to the oxidant/antioxidant imbalance [22]. In this regard the decreases in serotonin levels might result from reduced synthesis resulting from radiation- induced damage to the ileal mucosa [23], and decrease in the absorption of tryptophan involved in the synthesis of serotonin. In addition the decrease of serotonin might be attributed to increased catabolism due to increased monoamine oxidase (MAO) activity [24]. These findings are in good agreement with thosereported that gamma rays exposure of ratsat doses of 2Gy/week up to a total dose of 8Gy caused significant decreases of serotonin 5-HT (P<0.001) levels compared to their respective values in the control group. |
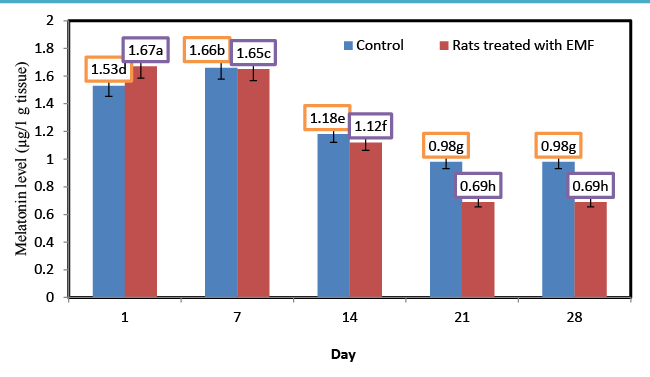
| Influence of exposure to EMF on brain melatonin (μg/1 g tissue) of newborn rats |
| Melatonin (N-acetyl-5-methoxytryptamine) is a hormone produced by the pineal gland. Melatonin, the most powerful (known) endogenous antioxidants, stimulates the GPX and can play a role in the prevention of oxidative damage [25]. Recent studies have demonstrate that melatonin is an antioxidant, which scavenges hydroxyl radicals generated in vitro by hydrogen Peroxide exposed to ultraviolet light [26,27]. Figure 4 shows the changes in the levels of melatonin for control rats and rats treated with EMF. Generally the levels of melatonin in control rats were higher than those of rats treated with EMF. |
| The lowest content (0.62 μg/1 g tissue) of melatonin was observed for the rats which treated with electromagnetic field EMF for 4 weeks. Melatonin level of rats treated with electromagnetic field EMF decreased from 1.67 at the first day to 0.69 μg/1 g tissue at the end of the experiment. There is some research indicating that low frequency EMF decreases serum and pineal melatonin levels [28,29]. The melatonin hypothesis proposed by Stevens and Davis in 1996 (24) has stood out due to a report that EMF lowered nocturnal melatonin concentrations [30] and hence its oncostatic potential (i.e., its capability to inhibit cancer cell growth and/or affect the immune system). Three mechanisms have been proposed forexplaininghow EMFs can increase cancer risk by lowering melatonin concentrations: 1) lower melatonin concentrations could lead to higher oestrogen and prolactin concentrations and eventually to higher incidence and prevalence of hormonally mediated cancers; 2) considering melatonin’s anti-oxidative properties, lowering its levels could increase DNA susceptibility to damage and eventually the overall cancer risk; and 3) considering melatonin’s ability to inhibit cell proliferation, lower concentrations could be relevant for cancer development in general [31]. |
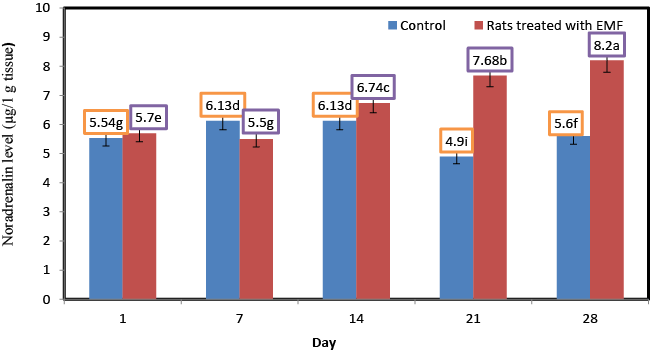
| Influence of exposure to EMF on brain noradrenalin (μg/1 g tissue) of newborn rats |
| Norepinephrine ( NE) or noradrenaline (NA or NAd) is a catecholamine with multiple roles including as a hormone and a neurotransmitter.Noradrenaline (NA) is recognized as a key modulator in the regulation of multiple central nervous system (CNS) activities, such as mood changes [32], memory formation [33], stress response [34] and cellular energy metabolism [35], among others. Figure 5 shows the changes in the levels of norepinephrine for control rats and rats treated with EMF. No significant (p ≥ 0.05) changes in the concentration of norepinephrine were shown for the rats of control group. A significant increase in the content of norepinephrine was found in those ratsexposed to electromagnetic field EMF. Norepinephrine content of rats treated with electromagnetic field EMF increased from 5.7 at the first day to 8.2 μg/1 g tissues at the end of the experiment. Zecca et al. [36] found that higher field strength exposure may raise norepinephrine levels in pineal gland of rats accompanied with key changes in brain involving opioid system in frontal cortex, parietal cortex, and hippocampus. Significantly higher levels of stress biomarkers like adrenaline, noradrenaline, and cortisol have been documented in medical staffs with RFR-EMF exposures, which could also influence B.P., heart rate (HR), and so forth [37]. |
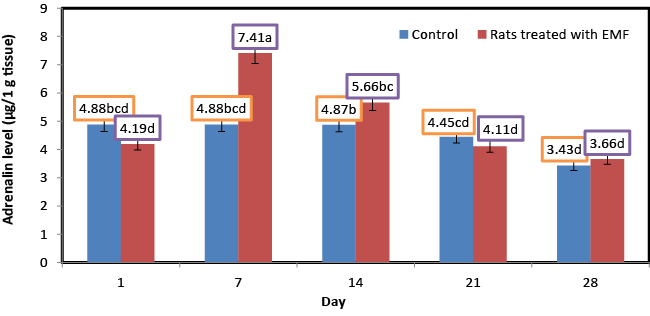
| Influence of exposure to EMF on brain adrenaline (μg/1 g tissue) of newborn rats |
| Figure 6 shows the changes in the levels of adrenaline for control rats and rats treated with EMF. No significant (p ≥ 0.05) changes in the concentration of adrenaline were shown for the rats of control group. Adrenaline content of rats treated with electromagnetic field EMF significantly increased from 4.19 at the first day to5.66 μg/1 g tissues at the end of the second week of the experiment. However the exposure to EMF caused significant (P ≤ 0.05) decreases in adrenaline content starting from the third week of the experiment and towards the fourth week of the experiment. Significantly, higher levels of the stress biomarkers adrenaline andnoradrenaline have been reported in physiotherapists [37]. Buchner and Eger [38] also assessed the catecholamines in subjects exposed to cell phone base station. They examined the acute as well as chronic effects of EMF exposure and found a significant increase in adrenaline and nor-adrenaline levels after EMF exposure, following a drop, but the normal levels were not restored even at the end of the study (about oneand a half year). Occupational studies focused on the effect of EMFs released frommagnetostrictive cavitrons used by dentists showed serum cortisol diminution [39]. In the midst of all these studies reporting either an increase or decrease in cortisol concentration, another group of researchers reported no significant change [40] reflecting the inconsistency in this field. With regard to adrenaline and noradrenaline level, significant suppression was registered in electric utility workers exposed occupationally to 50/60Hz EMFs [41]. |
| Conclusion |
| The results of the current study concluded that the exposure of new born rats to EMF may be sufficient to induce significant changes in the content of monoamine neurotransmitters of the exposed rats. |
References
|
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Analytical Biochemistry
- Applied Biochemistry
- Carbohydrate Biochemistry
- Cellular Biochemistry
- Clinical_Biochemistry
- Comparative Biochemistry
- Environmental Biochemistry
- Forensic Biochemistry
- Lipid Biochemistry
- Medical_Biochemistry
- Metabolomics
- Nutritional Biochemistry
- Pesticide Biochemistry
- Process Biochemistry
- Protein_Biochemistry
- Single-Cell Biochemistry
- Soil_Biochemistry
Recommended Journals
- Biosensor Journals
- Cellular Biology Journal
- Journal of Biochemistry and Microbial Toxicology
- Journal of Biochemistry and Cell Biology
- Journal of Biological and Medical Sciences
- Journal of Cell Biology & Immunology
- Journal of Cellular and Molecular Pharmacology
- Journal of Chemical Biology & Therapeutics
- Journal of Phytochemicistry And Biochemistry
Article Tools
Article Usage
- Total views: 18698
- [From(publication date):
June-2015 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 13918
- PDF downloads : 4780
