Research Article Open Access
Efficacy and Durability of Cryospray Ablation for Dysplastic Barrett’s Esophagus
Jayaprakash Sreenarasimhaiah*Department of Medicine, University of Texas Southwestern Medical Center, USA
- *Corresponding Author:
- Jayaprakash Sreenarasimhaiah
Department of Medicine Division of Digestive and Liver Disease
University of Texas Southwestern Medical Center 5323 Harry Hines Blvd
MC 90 Dallas, TX 75390, USA
Tel: 9729983754
E-mail: Jayaprakash.Sreenarasimhaiah@UTSouthwestern.edu
Received date: August 21, 2014; Accepted date: September 15, 2014; Published date: September 20, 2014
Citation: Sreenarasimhaiah J (2014) Efficacy and Durability of Cryospray Ablation for Dysplastic Barrett’s Esophagus. J Gastroint Dig Syst 4:220. doi:10.4172/2161-069X.1000220
Copyright: © 2014 Sreenarasimhaiah J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Background: Dysplastic Barrett's esophagus is the precursor to esophageal adenocarcinoma. While traditional therapy has included surgical resection, several endoscopic ablation techniques have become well-established. The newest of these modalities is cryospray ablation using liquid nitrogen.
Objective: To determine efficacy of cryospray ablation therapy in eradication of Barrett's mucosa and dysplastic tissue as well as the durability of these outcomes.
Methods: A retrospective analysis of 33 consecutive patients who underwent endoscopic cryoablation therapy was performed. Patients were followed at 8 weeks, 6 months, and annually following completion of ablation. Biopsies were taken.
Results: A mean of 2.4 sessions (range 1-4) was performed with a mean follow-up of 12.5 months (range 6-36). 100% of patients with low-grade dysplasia achieved complete eradication of dysplasia at a minimum follow-up of 6 months. 95% of highgrade dysplasia was eradicated at 6 months with 5% having persistent focal low-grade dysplasia. In 9% of cases, intramucosal carcinoma was also treated successfully with cryospray ablation therapy. Overall stricture rate was 3% following ablation therapy. No perforations or other serious complications occurred
Conclusions: Cryoablation therapy offers an effective minimimally-invasive method to treat dysplastic Barrett's esophagus as well as some early intramucosal adenocarcinoma. There is minimal morbidity from this procedure and has potentially durable results at follow-up endoscopic surveillance.
Abstract
Background: Dysplastic Barrett's esophagus is the precursor to esophageal adenocarcinoma. While traditional therapy has included surgical resection, several endoscopic ablation techniques have become well-established. The newest of these modalities is cryospray ablation using liquid nitrogen.
Objective: To determine efficacy of cryospray ablation therapy in eradication of Barrett's mucosa and dysplastic tissue as well as the durability of these outcomes.
Methods: A retrospective analysis of 33 consecutive patients who underwent endoscopic cryoablation therapy was performed. Patients were followed at 8 weeks, 6 months, and annually following completion of ablation. Biopsies were taken.
Results: A mean of 2.4 sessions (range 1-4) was performed with a mean follow-up of 12.5 months (range 6-36). 100% of patients with low-grade dysplasia achieved complete eradication of dysplasia at a minimum follow-up of 6 months. 95% of highgrade dysplasia was eradicated at 6 months with 5% having persistent focal low-grade dysplasia. In 9% of cases, intramucosal carcinoma was also treated successfully with cryospray ablation therapy. Overall stricture rate was 3% following ablation therapy. No perforations or other serious complications occurred
Conclusions: Cryoablation therapy offers an effective minimimally-invasive method to treat dysplastic Barrett's esophagus as well as some early intramucosal adenocarcinoma. There is minimal morbidity from this procedure and has potentially durable results at follow-up endoscopic surveillance.
Keywords
Dysplastic Barrett's esophagus; Cryospray; High-grade dysplasia; Proton-pump inhibitor
Introduction
As more patients undergo earlier endoscopic evaluation for acid reflux symptoms, the identification of Barrett’s esophagus has been on the rise. The major concern, however, is the detection of dysplasia and the subsequent risk of progression to esophageal adenocarcinoma. Most cases of adenocarcinoma today arise from dysplastic Barrett mucosa [1]. The standard of care for several decades has been careful surveillance biopsies to detect dysplasia. Current guidelines recommend that low-grade dysplasia (LGD) should warrant repeat endoscopic surveillance at six months following high-dose proton-pump inhibitor (PPI) therapy. High-grade dysplasia (HGD) has a greater risk of cancer progression and often has concomitant early esophageal neoplasia [2]. This requires definitive therapy which has traditionally included surgical resection [3].
However, in recent years, several minimally-invasive therapies have been used to perform ablation of Barrett’s mucosa including endoscopic mucosal resection (EMR), radiofrequency ablation, and cryospray ablation. Traditional therapy such as EMR and surgical resection may have technical challenges and associated morbidity [4].
The newest of the available methods, cryospray ablation has now become an established modality for the treatment of dysplastic Barrett’s tissue with minimal morbidity and favorable clinical outcomes [5]. This method offers application of extremely frigid thermal injury to the esophageal mucosa through repeated cycles of freezing and thawing. Vascular stasis with ischemia and anoxia due to vasoconstriction is a major part of the pathologic process. This results in cell death with both immediate and delayed effects while preserving the extracellular matrix. Thus healing involves the sloughing of the treated mucosa and growth of neosquamous mucosa without significant esophageal wall injury [6-8].
The objective of this study is to determine the efficacy of achieving a complete response with cryospray ablation therapy for dysplastic Barrett’s epithelium. A durable response is also to be examined with analysis of long-term follow-up endoscopy. Furthermore, overall safety and patient comfort with cryospray ablation in dysplastic Barrett’s esophagus will be examined.
Methods
Study group
This was a retrospective analysis of 33 consecutive patients who underwent cryospray ablation therapy for dysplastic Barrett’s esophagus. The study group included 25 males and 8 females with a mean age of 65.3 years whose histology confirmed HGD (n=20), LGD (n=10), and HGD with intramucosal cancer (n=3) in the form of a nodule. All patients with intramucosal cancer had no extension of tumor into lymph nodes or distant sites as determined by computerized tomography (CT) and EUS imaging (Table 1).
| Mean age | Mean length | Male | Female | LGD | HGD | IMC | PPI twice daily |
| 65.3 years | 5.5cm | 25 | 8 | 10 | 20 | 3 | 100% |
Table 1: Study demographics.
Upper endoscopy
Upper endoscopy with narrow-band imaging (Olympus GIF1T180, Tokyo Japan), was performed initially in all patients to identify the length and nature of the Barrett’s esophagus as well as to detect nodules or strictures. Four-quadrant biopsies were taken at 1cm intervals in the esophagus using large capacity Radial Jaw-4 biopsy forceps (Boston Scientific, Natick, MA). Patients who had HGD were enrolled in the prospective study of cryospray ablation. Patients with LGD were placed on esomeprazole 40mg twice daily and followed clinically. If repeat biopsies at six months from the initial endoscopy suggested persistent dysplasia, these patients were offered study enrollment.
Endoscopic ultrasound
Patients with high-grade dysplasia or carcinoma underwent index radial endoscopic ultrasound exam (GFUE160, Olympus, Tokyo, Japan) prior to cryospray ablation therapy. Tumor and nodal staging was performed for carcinomas. The celiac axis and paraesophageal regions were examined for lymph node enlargement or abnormality. If lymph nodes were seen and measured at least 1.0cm, fine needle aspiration biopsy was performed with a 25G needle.
Cryospray therapy procedure
Endoscopy was performed under monitored anesthesia care. Ablation was accomplished using liquid nitrogen applied through a specialized spray catheter (CSA Medical, Baltimore MD, USA) which was placed through the accessory channel of the endoscope. Prior to ablation, a decompression catheter with several distal openings was positioned 1cm below the gastroesophageal junction. Constant suction was applied during the spray delivery of liquid nitrogen. Additionally, the abdomen was monitored by a nurse for over-distension. A clear cap applied to the tip of the endoscope was used to help visualize the mucosa during spray therapy. Standard protocol for ablation included 20-second freeze cycles performed twice for each site starting from the proximal end of the Barrett mucosa and proceeding distally. The patients were given acetaminophen if needed for pain control and no narcotics were required after any procedure. Twice daily dose of esomeprazole 40mg was continued for 8 weeks until the follow-up endoscopy was performed. If any mucosal breaks or ulcerations were noted, cryospray therapy was postponed until healing could be confirmed. If a nodule was found, focal EMR was performed. If EMR was performed initially, cryoablation therapy was performed at least 6 weeks later to ensure that no mucosal defects were present. If EMR was performed following cryoablation therapy, this was achieved either immediately in the same endoscopic session or anytime thereafter during follow-up endoscopy.
Ethics
The study was conducted with approval from the Institutional Review Board. Informed consent from human subjects was acquired to collect and analyze data in a confidential manner. The study methods conformed to the ethical guidelines of the "Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects.”
Statistical analysis
A descriptive analysis was performed to determine the mean length of treatment, number of sessions, and complications. This included calculation of the rate of successful resolution of dysplasia and the rate of complete eradication of Barrett’s mucosa.
Results
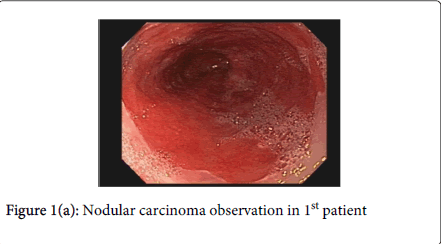
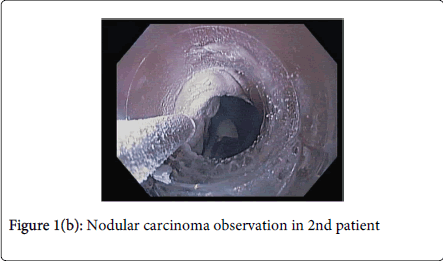
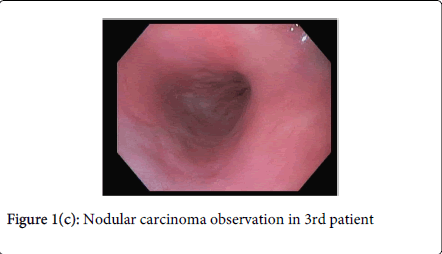
The average length of Barrett’s mucosa was 5.8cm which required a mean of 2.4 sessions (range 1-4) to achieve complete ablation of dysplastic mucosa by cryospray therapy. Complete response was defined as no visible Barrett’s mucosa and no dysplasia on subsequent surveillance biopsies. Follow-up endoscopy with biopsies was performed in all patients at 6 months following completion of therapy. The range of follow-up was between 6 and 36 months with a mean of 12.5 months. In 100% of patients with HGD, complete response was achieved at 8-week follow-up after final treatment session. At 6-month follow-up, one (5%) had a focal area of LGD but no HGD. All 10 patients with original LGD achieved complete response which was sustained at 6 month follow-up endoscopy. One patient died of unrelated causes 8 months after achieving complete ablation. One patient was lost to follow-up after 12 months due to insurance changes but had complete response at 6 month follow-up (Figure 1a, 1b, and 1c).
In all three patients with intramucosal nodular carcinoma, endoscopic therapy was successful with no findings of residual disease at six months – cryospray alone [1], EMR prior to cryotherapy [1], and EMR following ablation [1]. One patient with T1N0 esophageal carcinoma at the GE junction who was successfully treated with combined ablation and mucosal resection was seen in follow-up at 24 months and had no abnormal endoscopic or histologic findings. In addition, one patient underwent successful EMR at 8 week follow-up after cryoablation therapy for a remaining 2 cm patch of dysplastic Barrett’s mucosa (Table 2).
| Eradication of visible Barrett’s at 12 months | Eradication of LGD at 6 months | Eradication of HGD at 6 months | Eradication of IMC by cryo alone | Eradication of IMC by cryo + EMR | Eradication of LGD at 12 months | Eradication of HGD at 12 months |
| 33/33 (100%) | 10/10 (100%) | 19/20 (95%) | 1/1 | 2/2 | 9/9(100%) One died of unrelated cause | 18/18 (100%) One lost to follow-up endoscopy |
Table 2: Study results; Note: one patient with HGD at 6 months had persistent LGD which was successfully treated and was not present at 12 months; LGD- Low grade dysplasia; HGD – High grade dysplasia; IMC – Intramucosal cancer; PPI – Proton pump inhibitor
In regard to safety and complications, the procedure was well-tolerated. No patients required narcotics or hospitalization following the procedure. No perforations or bleeding episodes occurred. Transient dysphagia without odynophagia was reported in 30% for a mean of 5 days. In a total of 90 sessions of cryospray therapy, 3 (3.3%) benign strictures developed in 2 patients as a result of therapy and were treated successfully with single sessions of endoscopic balloon dilation. In the patient with two episodes of strictures, noncompliance was discovered with the prescribed daily double dose of esomeprazole (Table 3).
| Adverse events related to sedation | 0 |
| Transient dysphagia without intervention | 30% |
| Dysphagia due to stricture requiring dilation | 3.3% |
| Death related to Barrett’s or procedure | 0 |
| Unrelated death at 12 months | 3.3% |
Table 3: Complications
Discussion
The ablation of Barrett’s mucosa at the dysplastic stage can help prevent the development of esophageal cancer [9]. While Barrett’s esophagus affects only the mucosal layer, high-grade dysplasia and subsequent carcinoma may involve deeper layers of the esophagus and possibly paraesophageal lymph nodes [10]. Often, the histologic distinction of high-grade dysplasia and carcinoma cannot be made and thus EUS offers excellent visualization of the esophageal layers and paraesophageal lymph nodes to identify advanced disease [11,12] EUS has been shown to have an accuracy of 85% in staging of high-grade dysplasia and early esophageal cancer with regard to depth and invasion into layers beneath the mucosa [13,14]. Furthermore, suspicious lymph nodes can be identified. Thus, EUS imaging was performed in many of the patients with HGD prior to ablation.
Cryospray ablation therapy offers a novel approach to a not-so-novel methodology in the destruction of unwanted tissue, in this case dysplastic Barrett’s mucosa. Cryospray with liquid nitrogen has been used successfully in many other medical applications including cervical dysplasia, early skin cancers and warts, oral leukoplakia and respiratory tract papillomas and tumors [15-17]. Thus, the application in the esophagus since its introduction in the past decade has other areas of experience from which to learn.
All forms of ablation therapy including cryospray, radiofrequency ablation, argon plasma coagulation, photodynamic therapy, and laser therapy result in the destruction of the Barrett’s mucosal layer with a replacement of newly developed squamous cells known as neosquamous epithelium [18]. A major clinical concern, however, with all forms of ablation has been residual or “buried” glands of Barrett’s epithelium beneath the neosquamous epithelial layer [19].
This has been demonstrated in one study with photodynamic therapy to occur in up to 27.3% of cases [20]. The true rate of such residual glands from all forms of endoscopic ablation is unknown and difficult to determine by endoscopic mucosal biopsies. Furthermore, the clinical significance of this histologic finding particularly with recurrence of dysplasia and cancer is unclear and is still undergoing investigation. Nonetheless, in this study, biopsies in a standard 4-quadrant protocol were performed in the areas of neosquamous mucosa following complete cryoablation to help determine the durability of the treatment. As the results demonstrate at a mean follow-up of 12.5 months, a 100% rate of eradication of dysplasia was achieved by cryospray ablation. Longer follow-up will be helpful to determine further durability of this treatment and the possibility of recurrent subepithelial Barrett’s glands.
The standard of practice for nodules in the background of Barrett’s is to biopsy these separately to determine if there is advanced pathology [21]. If endoscopic ablation therapy is being planned, nodules may benefit from EMR initially. This permits complete removal of a nodule that extends into the submucosa but does not penetrate the muscularis propria. The advantage of EMR is complete removal of a nodule or even small segments of Barrett’s mucosa without concern for buried glands. However, large segments of EMR, particularly circumferential resection is technically challenging and fraught with complications such as bleeding, perforation, and esophageal stricture rates of approximately 27% [22]. The advantage of cryospray ablation in this setting is the technical ease of treatment of large areas, including circumferential disease with minimal adverse events. Often EMR is combined with endoscopic ablation therapy.
EMR has been shown to be successful following cryospray ablation therapy but not as feasible following radiofrequency ablation [9,23]. Our study demonstrated similar technical success of EMR in three cases following cryospray ablation. The delayed effect of cell death without the immediate destruction of the intracellular matrix may provide this ease of EMR therapy following cryospray ablation. Ablation procedures that cause immediate tissue destruction with intracellular disruption may potentially result in scarring that could impair effective EMR technique.
Finally, patient tolerance to any ablation procedure is an important factor to consider. In this series of patients, no significant pain was reported from cryospray ablation therapy that required admission or even the use of narcotic medications in a single patient. No serious complications were encountered such as bleeding or perforation. The stricture rate of 3% was similar to other reported studies such as a recent meta-analysis of endoscopic radiofrequency ablation in which esophageal stenosis occurred in 5% of patients [24]. In this group of cryoablation induced stricture, patients were easily treated within a single endoscopic session with balloon dilation.
Summary
Cryospray ablation therapy offers a minimally invasive method to treat dysplastic Barrett’s mucosa with low morbidity. Complete ablation can be achieved by this method as evidenced by histology specimens and endoscopic findings of neosquamous epithelium. This modality has applications for HGD, persistent LGD, and intramucosal cancers with durable response. Moreover, it is a technique that can be combined with other endoscopic therapies such as endoscopic mucosal resection. Given its superior safety profile and long-term efficacy, endoscopic cryospray ablation has emerged as a viable modality to eradicate dysplastic Barrett’s esophagus. This study demonstrates excellent durability of results after a mean follow-up of one year. Further studies of long-term durability will be helpful to confirm these findings.
References
- Sharma P (2009) Clinical practice. Barrett's esophagus.N Engl J Med 361: 2548-2556.
- Zhu W, Appelman HD, Greenson JK, Ramsburgh SR, Orringer MB, et al. (2009) A histologically defined subset of high-grade dysplasia in Barrett mucosa is predictive of associated carcinoma.Am J ClinPathol 132: 94-100.
- Wang KK, Sampliner RE; Practice Parameters Committee of the American College of Gastroenterology (2008) Updated guidelines 2008 for the diagnosis, surveillance and therapy of Barrett's esophagus.Am J Gastroenterol 103: 788-797.
- Waxman I, Konda VJ (2009) Mucosal ablation of Barrett esophagus.Nat Rev GastroenterolHepatol 6: 393-401.
- McAllaster JD, Buckles D, Al-Kasspooles M (2009) Treatment of Barrett's esophagus with high-grade dysplasia.Expert Rev Anticancer Ther 9: 303-316.
- Halsey KD, Greenwald BD (2010) Cryotherapy in the management of esophageal dysplasia and malignancy.GastrointestEndoscClin N Am 20: 75-87, vi-vii.
- Odze RD, Lauwers GY (2008) Histopathology of Barrett's esophagus after ablation and endoscopic mucosal resection therapy.Endoscopy 40: 1008-1015.
- Johnston L, Johnston MH(2005)Cryospray ablation (CSA) in the esophagus: optimization of dosimetry [abstract]. Am J Gastroenterol 101:S532.
- Dumot JA, Vargo JJ, Falk GW, Frey L, Lope R, et al. (2009) An open-label prospective trial of cryospray ablation for Barrett’s esophagus high-grade dysplasia and early esophageal cancer in high-risk patients. GastrointestEndosc70:635-644.
- Odze RD (2006) Diagnosis and grading of dysplasia in Barrett's oesophagus.J ClinPathol 59: 1029-1038.
- Larghi A, Lightdale CJ, Memeo L, Bhagat G, Okpara N, et al. (2005) EUS followed by EMR for staging of high-grade dysplasia and early cancer in Barrett's esophagus.GastrointestEndosc 62: 16-23.
- Savoy AD, Wallace MB (2005) EUS in the management of the patient with dysplasia in Barrett's esophagus.J ClinGastroenterol 39: 263-267.
- Gress F, Yeingpruksawan A, Cerulli M. Potential role of endoscopic ultrasound in evaluating dysplasia in Barrett esophagus [Abstract]. Am J Gastroenterol. 1993; 88:1487A.
- Scotiniotis IA, Kochman ML, Lewis JD, Furth EE, Rosato EF, et al. (2001) Accuracy of EUS in the evaluation of Barrett's esophagus and high-grade dysplasia or intramucosal carcinoma.GastrointestEndosc 54: 689-696.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, Landau J (2010) Liquid nitrogen: temperature control in the treatment of actinic keratosis.DermatolSurg 36: 1956-1961.
- Lin HP, Chen HM, Cheng SJ, Yu CH, Chiang CP (2012) Cryoguncryotherapy for oral leukoplakia.Head Neck 34: 1306-1311.
- Kourounis G, Iatrakis G, Diakakis I, Sakellaropoulos G, Ladopoulos I, et al. (1999) Treatment results of liquid nitrogen cryotherapy on selected pathologic changes of the uterine cervix.ClinExpObstetGynecol 26: 115.
- Berenson MM, Johnson TD, Markowitz NR, Buchi KN, Samowitz WS (1993) Restoration of squamous mucosa after ablation of Barrett's esophageal epithelium.Gastroenterology 104: 1686-1691.
- Hornick JL, Blount PL, Sanchez CA, et al. Biologic properties of columnar epithelium underneath reepithelialized squamous mucosa in Barrett’s esophagus (2005). Am J SurgPathol 29:372-380.
- Ban S, Mino M, Nishioka NS, Puricelli W, Zukerberg LR, et al. (2004) Histopathologic aspects of photodynamic therapy for dysplasia and early adenocarcinoma arising in Barrett's esophagus.Am J SurgPathol 28: 1466-1473.
- Odze RD (2006) Diagnosis and grading of dysplasia in Barrett's oesophagus.J ClinPathol 59: 1029-1038.
- Qumseya B, Panossian AM, Rizk C, Cangemi D, Wolfsen C1, et al. (2014) Predictors of esophageal stricture formation post endoscopic mucosal resection.ClinEndosc 47: 155-161.
- Pouw RE, Gondrie JJ, Sondermeijer CM, ten Kate FJ, van Gulik TM, et al. (2008) Eradication of Barrett esophagus with early neoplasia by radiofrequency ablation, with or without endoscopic resection.J GastrointestSurg 12: 1627-1636.
- Orman ES, Li N, Shaheen NJ (2013) Efficacy and durability of radiofrequency ablation for Barrett's Esophagus: systematic review and meta-analysis.ClinGastroenterolHepatol 11: 1245-1255.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 15334
- [From(publication date):
August-2014 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 10621
- PDF downloads : 4713