Research Article Open Access
Enhancing Antioxidant Properties of Germinated Thai rice (Oryza sativa L.) cv. Kum Doi Saket with Salinity
| Kamolchanok Umnajkitikorn1,Bualuang Faiyue2*and Kobkiat Saengnil1 | ||
| 1Department of Biology, Faculty of Science, Chiang Mai University, Chiang Mai, Thailand | ||
| 2Department of Biology, Mahidol Wittayanusorn School, Salaya, Nakhon Pathom, Thailand | ||
| Corresponding Author : | Bualuang Faiyue Department of Biology Mahidol Wittayanusorn School Salaya, Nakhon Pathom, Thailand Tel: 662-849-7244 E-mail: b.faiyue@gmail.com |
|
| Received June 17, 2013; Accepted June 22, 2013; Published July 25, 2013 | ||
| Citation: Umnajkitikorn K, Faiyue B, Saengnil K (2013) Enhancing Antioxidant Properties of Germinated Thai rice (Oryza sativa L.) cv. Kum Doi Saket with Salinity. J Rice Res 1:103. doi: 10.4172/jrr.1000103 | ||
| Copyright: © 2013 Umnajkitikorn K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | ||
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
||
Visit for more related articles at Rice Research: Open Access
Abstract
The objective of this study was to investigate the possibility of enhancing the antioxidant properties of germinated rice seeds cv. Kum Doi Saket, a pigmented rice variety cultivated in northern Thailand, using salinity. Rice seeds were soaked in 0, 25, 75, 150, and 300 mM NaCl for 12 hours, sown on 0.7% agar supplemented with the same NaCl concentrations for soaking at 28 ± 2°C in the dark for 5 days. Germination percentage, growth of seedlings, antioxidant capacities, total phenolic content, and activities of Ascorbate Peroxidase (APX) and Catalase (CAT) were examined. The results showed that germination was not affected by 25 and 75 mM NaCl, but was significantly reduced by 17% and 100% under 150 and 300 mM NaCl, respectively. Fresh weight was not affected by 25 mm NaCl, whereas all NaCl concentrations significantly reduced the shoot length of germinated seeds. Root length was significantly reduced by 150 and 300 mM NaCl. Antioxidant capacities, quantified by DPPH and ABTS radical scavenging assays, and total phenolic content significantly increased in germinated seeds treated with 150 mM NaCl. A high positive correlation between antioxidant capacities and total phenolic content was found. APX activities increased with germination time, reached the highest peak on day 2 for the control, and on day 4 for salt treatments with 150 mM NaCl showing the highest activity. CAT activities of the control reached the maximum on day 2, whereas it showed different highest peaks for salt treatments; salinity reduced the activity of CAT during germination. These results suggest that antioxidant properties of Kum Doi Saket germinated rice seeds are enhanced by salinity stress of 150 mM NaCl. This is an alternative way to elevate antioxidant properties for consumption and for further pharmaceutical application of germinated Thai rice.
| Keywords | |
| Germinated seed; Phenolic content; Pigmented rice; NaCl stress; Salt stress | |
| Introduction | |
| Germinated rice has been of interest throughout Asian countries [1] as it contains high amounts of bioactive compounds such as γ-aminobutyric acid (GABA), γ-oryzanol, and dietary fiber [1-4]. Germinated rice also contains high amounts of ferulic acid [5], α-tocopherols [1], total phenolic [4], and total anthocyanin contents [6] which have potent antioxidant and free radical scavenging properties [5,7,8]. | |
| Pigmented rice (also known as colored rice) has higher antioxidant capacity which illustrates the total ability of antioxidants to scavenge free radicals [9], total phenolic and flavonoid contents than white rice [10-13]. Moreover, germinated pigmented rice has higher total anthocyanin content and antioxidant activity than those of germinated non-pigmented rice [6]. However, little attention has been given on the antioxidative property of germinated pigmented rice and data relevant to this are not readily available in Thailand [6]. | |
| Salinity promotes some antioxidant compounds and antioxidant capacity in rice. For example, Daiponmak et al. [14] reported that antioxidant capacities, total phenolic content, and cyanidin-3-glycoside content were significantly increased in leaves of 16-day-old seedlings of Thai rice cultivars KDML105 and Kum Doi Saket under salinity of 60 mM NaCl for 11 days. Chutipaijit et al. [15] found that total antioxidant capacity and the amounts of proline, anthocyanins and flavonoids of 14-day-old seedlings of pigmented and non-pigmented Thai rice cultivars KDML105, Pathumthani1, Sangyod, Khao Dang, Kulab Dang, KumSakol, Kum Khonkaen1, KumKhonkaen 2, and Black Sticky were enhanced after being grown in NB-liquid medium supplemented with 100 mM NaCl for 4 days [16]. Ghosh et al. [17] reported that a significant increase in total phenolic content was observed in 17-day old seedlings of Pokkali and Nonabokra rice varieties during exposure to salinity of 200 mM NaCl for 48 and 72 hours. | |
| Antioxidant enzymes such as Ascorbate Peroxidase (APX) and Catalase (CAT), which also have a major role in scavenging Reactive Oxygen Species (ROS), have been increased in rice under salinity. Demiral and Turkan [18,19] found that the treatment of 120 mM NaCl for 1 week significantly increased APX and CAT activity in leaves and roots of 4-week-old Pokkali seedlings. | |
| Although studies have Indicated that salinity increased the antioxidant capacity, total phenolic compounds, and antioxidant enzymes in rice, the experiments were conducted on seedlings aged 2 to 4 weeks [14-19]. Our objective was to evaluate the possibility of increasing the antioxidant properties of germinated rice cv. Kum Doi Saket, which is a local pigmented rice cultivar cultivated in northern Thailand. It has been reported to have high efficiency as a cytotoxic to cancer cells [20,21]. Salinity can add nutritional value to this crop. | |
| Materials and Methods | |
| Plant materials and treatments | |
| Seeds of rice cv. Kum Doi Saket were obtained from the Department of Plant Science and Natural Resources, Faculty of Agriculture, Chiang Mai University, Thailand. Rice seeds were dehusked with a husker (KM™ model HMF, Tokyo, Japan) and sterilized with 2.0% Clorox solution for 20 minutes. The seeds were soaked in solutions of 0 (control), 25, 75, 150, and 300 mM NaCl for 12 hours at ambient temperature (30 ± 2°C, 75 ± 3% RH) before being sown on 0.7% agar added with the same NaCl concentrations for soaking and incubated in the dark at 28 ± 1°C with a relative humidity (RH) of 80 ± 2% for 5 days. Germinated seeds were randomly sampled every day for subsequent determinations. | |
| Seed germination and growth measurement | |
| Germination percentages were recorded every day, and the germination was defined when the radicle protruded at least 1 mm [22]. Fresh weight, shoot and root lengths were recorded daily for each treatment. | |
| Determination of antioxidant capacity | |
| Antioxidant capacity was determined using DPPH and ABTS radical scavenging assays following the method of Mun’im et al. [23] and Huang et al. [24], with slight modification. Four grams of germinated seeds were homogenized in 10 ml of 80% ethanol by using a mortar and pestle, and centrifuged at 16,000Xg and 4°C (refrigerated centrifuge model IEC Multi-RF, USA) for 20 minutes. The supernatant was collected for DPPH and ABTS radical scavenging assays. | |
| For DPPH radical scavenging assay, 100 μl of supernatant were mixed with 400 μl of 0.3 M acetate buffer (pH 5.5) and 2.5 ml of 0.12 mM DPPH in 95% methanol. The mixed solution was incubated in a dark room for 30 minutes and the absorbance was measured at 517 nm with a visible spectrophotometer (Thermo Spectronic™ model Helios Epsilon, USA). The antioxidant capacity of the samples was calculated by the decreasing intensity of the purple color of DPPH• as a result of a reaction between DPPH• and the antioxidant compounds in the samples following the equation: | |
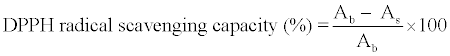 |
|
| Ab was the absorbance of the control at 30 minutes (using 80% ethanol instead of the supernatant) and As was the absorbance of the supernatant at 30 minutes. Trolox was used as a standard and the results were expressed as Trolox equivalent antioxidant capacity (TEAC; μmol Trolox/g fresh weight). | |
| For ABTS radical scavenging assay, 20 μl of supernatant were mixed with 2.0 ml of 0.08 mM ABTS•+ solution in 80% ethanol for 10 minutes at room temperature. The absorbance was measured at 734 nm with a visible spectrophotometer (Thermo Spectronic™ model Helios Epsilon, USA). The antioxidant capacity of the samples was determined by decreasing intensity of the blue-green color of ABTS•+ as a result of a reaction between ABTS•+ and the antioxidant compounds in the samples following the same equation with DPPH radical scavenging capacity. The results were also expressed as Trolox equivalent antioxidant capacity (TEAC; μmol Trolox/g fresh weight). | |
| Determination of total phenolic content | |
| The total phenolic content was determined by the method of Singleton and Rossi [25]. Four grams of germinated seeds were homogenized in 10 ml of 80% ethanol by using a mortar and pestle and then centrifuged at 16,000xg and 4°C (refrigerated centrifuge model IEC Multi-RF, USA) for 20 minutes. Four hundred microliters of supernatant were mixed with 2 ml of 10% Folin-Ciocalteu reagent (V/V) for 8 minutes. Then 1.6 ml of 7.5% sodium carbonate (W/V) was added. The tubes were incubated for 2 hours at 30°C and the absorbance was measured at 765 nm with a visible spectrophotometer (model Thermo Spectronic, USA). A standard curve of 0-0.01% gallic acid (W/V) was used to quantify the total phenolic content and the results were expressed as mg GAE/100 g fresh weight. | |
| Determination of CAT and APX activities | |
| Enzymes were extracted by the modified method of Sunohara and Matsumoto [26]. One gram of germinated seeds was ground in liquid nitrogen with a mortar and pestle. It was homogenized in 10 ml of the extracted solution containing 25 mM potassium phosphate buffer (pH 7.8), 0.4 mM EDTA, 1 mM ascorbic acid and, 2% PVPP (W/V). The homogenate was centrifuged at 15,000xg for 20 minutes at 4°C. The supernatant was filtrated through Whatman no. 1 filter paper and the filtrate was collected for CAT and APX activity assays as crude enzymes. | |
| CAT activity was assayed based on the method of Sunohara and Matsumoto [26]. The assay mixture (2 ml) contained 1.9 ml of 25 mM H2O2 in 50 mM potassium phosphate buffer (pH 7.0) and 0.1 ml of crude enzyme. The absorbance was measured at 240 nm with a UV/VIS spectrophotometer (model UV Lambda 25, USA). The enzyme activity was defined as the amount of H2O2 decomposed per minute and mg protein. | |
| APX activity was assayed based on the method of Sunohara and Matsumoto [26]. The assay mixture (2 ml)contained0.5 ml of 100 mM potassium phosphate buffer (pH 7.0),0.5 ml of 0.4 mM EDTA, 0.5 ml of 1 mM L-ascorbic acid, 0.02 ml of 10 mM H2O2, and 0.1 ml of crude enzyme. The absorbance was measured at 290 nm with a UV/VIS spectrophotometer (model UV Lambda 25, USA). The enzyme activity was defined as the amount of H2O2 decomposed per minute and mg protein. | |
| Protein levels were assayed according to Lowry et al. [27] with Folin-Ciocalteu reagent using 0-200 μg/ml Bovine Serum Albumin (BSA) as a standard. | |
| Statistical analysis | |
| The experiments were set up in a completely randomized design and repeated twice. The results were presented as the average of the repeated experiments by pooling individual data. One Way ANOVA and Tukey’s Multiple Range Tests (P<0.05) were performed to determine significant differences among the means of the treatments using SPSS version 16 (SPSS incorporation Chicago, IL, USA). Simple linear regression was used to estimate the correlation between the total phenolic content and antioxidant capacity from both DPPH and ABTS radical scavenging assays. | |
| Results | |
| Effect of NaCl on germination and growth of germinated rice seeds | |
| The germination percentage of Kum Doi Saket rice seeds was as high as 70% after being placed on agar without NaCl (control) for 1 day and reached the maximum at about 90% on day 2 (Figure 1). Seeds placed on agar supplemented with 25, 75, and 150 mM NaCl also started germinating on day 1, but the germination percentage was significantly reduced compared to the control (Figure 1). On day 5, the germination percentage of seeds placed on 25 and 75 mM NaCl was not significantly different from that of the control, ranging from 84-93%, whereas this percentage was significantly reduced by 17% in seeds germinated in 150 mM NaCl (Figure 1). The concentration of 300 mM NaCl completely inhibited the germination of Kum Doi Saket rice seeds (Figure 1). | |
| Fresh weight of rice seeds was not different between the treatments after being soaked for 12 hours (day 0) with different concentrations of NaCl (Table 1). The fresh weights of germinated rice seeds of the control and that of the seeds grown on agar with 25 and 75 mM NaCl significantly increased with the germination time from day 2 compared with that of day 0. It slightly increased from day 1 to 4, but significantly increased on day 5 for seeds germinated on 150 mM NaCl, while the fresh weight did not significantly change for seeds germinated on 300 mM NaCl (Table 1). On day 5, the fresh weight of 25 mM NaCl treatment was not significantly different from that of the control, whereas it was significantly reduced by 12, 16, and 25% for 75, 150, and 300 mM NaCl treatments, respectively (Table 1). | |
| Shoot length of germinated rice seeds of the control significantly increased from day 2 compared with that of day 0. It significantly increased from days 3, 4, and 5 for 25, 75 and 150 mM NaCl, respectively, while 300 mM NaCl completely inhibited the shoot length (Table 1). The shoot length was reduced by NaCl treatments with the reduction percentages of 39.2%, 45.3%, and 69.9% for 25, 75, and 150 mM NaCl, respectively, as compared to that of the control on day 5. Root length of germinated rice seeds was not significantly affected by 25 and 75 mM NaCl, but significantly decreased with 150 mM NaCl by 65.9% (Table 1). As with the effect on the shoot, 300 mM NaCl completely inhibited the growth of the roots (Table 1). | |
| Antioxidant capacity | |
| As shown in Figure 2, antioxidant capacities of Kum Doi Saket rice seeds determined by DPPH and ABTS radical scavenging assays were different among the treatments after soaking in different concentrations of NaCl for 12 hours (day 0). The seeds soaked in NaCl concentrations of 75, 150, and 300 mM had significantly higher antioxidant capacities than the seeds soaked in deionized water (control) and 25 mM NaCl, and 150 mM NaCl rendered the highest antioxidant capacity from both assays (Figure 2). When seeds were germinated on agar supplied with the same NaCl concentrations used for soaking, antioxidant capacities of the control gradually increased with germination time and reached the maximum at day 5 in which the antioxidant capacities were 1.45 and 1.79 times higher than those of day 0 for DPPH and ABTS radical scavenging assays, respectively (Figure 2). Similarly, the antioxidant capacities of seeds germinated on agar supplemented with NaCl increased continuously with germination time, and day 5 provided the highest antioxidant capacities in all NaCl concentrations as compared to those of day 0 (Figure 2). Among the concentrations of NaCl used in this study, 150 mM was found to be the best concentration enhancing antioxidant capacities. It produced the highest antioxidant capacity with significant differences in every germination time compared with the control, and when compared with other NaCl concentrations on days 4 and 5 (Figure 2). Although, ABTS radical scavenging assay showed higher values of antioxidant capacities than those of using DPPH radical scavenging assay, these two methods showed the same trend of results. | |
| Total phenolic content | |
| After being soaked in 25, 75, 150, and 300 mM NaCl for 12 hours (day 0), total phenolic content of Kum Doi Saket rice seeds were significantly higher than that of the control (Figure 3). Total phenolic content of the control increased with germination time and had the highest peak at day 5 (Figure 3), showing the same trend as the antioxidant capacity. The total phenolic content of germinated seeds on day 5 was 1.72 times more than day 0. Consistently, rice seeds exposed to agar supplied with 150 mM NaCl significantly increased total phenolic content when compared with the control and other NaCl concentrations on days 4 and 5 (Figure 3). The amount of total phenolic compounds increased by 1.99 times from day 0 compared with that of the control. | |
| APX and CAT activities | |
| Kum Doi Saket rice seeds soaked in 0, 25, 75, and 150 mM NaCl for 12 hours (day 0) showed the same level of APX activity, whereas the activity of APX significantly decreased in seeds soaked in 300 mM NaCl (Figure 4). During germination on agar supplimented with different NaCl concentrations for 5 days, APX activity of the control seeds (0 mM NaCl) increased with germination time and reached the maximum on day 2. APX activity of seeds germinated under NaCl stress sharply increased with germination time and NaCl concentrations, and reached the maximum on day 4 (Figure 4). A concentration of 150 mM NaCl showed the highest increase in APX activity, which is 3.37 times higher than that of the control (Figure 4). | |
| CAT activity of Kum Doi Saket rice seeds was different among the treatments after being soaked in NaCl concentrations for 12 hours (day 0) in which this activity in seeds soaked in 75 and 150 mM NaCl was significantly lower than that of other concentrations (Figure 5). The CAT activity of the control sharply increased with germination time and reached the maximum on day 2, whereas the activity of seeds germinated with different concentrations of NaCl had different highest peaks (Figure 5). The CAT activity of 25 and 75 mM NaCl reached a maximum on day 2, whereas it was on day 3 and day 5 for 150 and 300 mM NaCl, respectively (Figure 5). The activity at the highest peaks was 9.24, 8.34, 10.12, 7.04, and 3.98 times as compared to day 0 for the control, 25, 75, 150, and 300 mM NaCl, respectively. Interestingly, the CAT activity of rice seeds germinated on agar supplemented with NaCl was lower than that of the control seeds on all 5 days examined (Figure 5). | |
| Discussion | |
| Our results show that the germination percentage of rice seeds was not affected by 25 and 75 mM NaCl, and had 80% germination under high salinity of 150 mM NaCl, but an extreme salinity of 300 mM NaCl completely inhibited all germination (Figure 1). These results are consistent with the work of Shereen et al. [28] who reported that germination of rice seeds was unaffected by 50 and 75 mM NaCl. The results are also compatible with Khan et al. [29] who found that the germination of nine genotypes of rice (Kalijira, Shakkorkhora, Khaskani, Pokkali, Kalobail, Ashami, IPK37008, IPK37011, and BR11) was more than 80% in 150 mM NaCl. Similar results were reported by Islam and Karim [30] who showed that eight genotypes of Thai and Bangladesh rice (Awned-1, Rajbhog-2, Khorina, Soloi, Nonasail, Patnai 23, Sadamota and Chapali) had 80-90% germinationin 160 mM NaCl. Sohn et al. [31] reported that the germination percentage of rice cv. Kumnam was completely inhibited at 360 mM NaCl. | |
| Although Kum Doi Saket rice seeds germinated in 75 mM NaCl without a significant reduction in percentage (Figure 1), the shoot length significantly decreased with this concentration (Table 1). These findings agree with the notion that the sensitivity of rice plants to salinity depends on the developmental stage in which germination is relatively tolerant, while the early seedling stage of 1-3 weeks is salt-sensitive [32-34]. Our results agree with Khan et al. [29] who found that the average seedling height was reduced in all levels of 50, 100, 150, and 200 mM NaCl by approximately 20-80%. The inhibitory effect of salinity on growth of rice is caused by the osmotic and ionic effects of NaCl [33,35]. This osmotic effect is induced by high NaCl concentrations outside the roots, leading to the inhibitions of water uptake, cell elongation, and leaf development. The ionic effect is induced by NaCl taken up to the plants via the transpiration stream exceeding the ability of cells to compartmentalize in the vacuole [33,35,36]. Yeo et al. [37] reported that an addition of 50 mM NaCl to the culture solution stopped leaf elongation of rice genotypes IR2153 and Pokkali due to a limitation of water supply to the meristem caused by salinity. Similar results have been reported by Shereen et al. [38] who found that the shoots of six inbred rice lines of genotype IR55178 exposed to 75 mM NaCl for 2 weeks were significantly shorter than those of the control. The authors explained that this effect resulted from an inadequate ability of the rice plants to uptake water, and the toxic effect of sodium and chloride ions. Interestingly, our results show that the shoot length was significantly reduced by all concentrations of NaCl on day 5, whereas root length was significantly reduced only by 150 and 300 mM NaCl (Table 1). Similar results were reported by Lutts et al. [32] who found that the reduction of growth of rice genotypes I Kong Pao, Aiwu, Tainung 67, IR2153, IR4630, Nona Bokra, Buhra Rata, Panwell, and Pokkali under 20, 30, 40, and 50 mM NaCl for 1 week was more clearly observed in the shoot than that of the root. The explanation is that salt stress inhibits shoot growth more than root growth in order to increase water absorption and preserves water in the cells [39]. | |
| Seed germination is a complex process involving biochemical and physical activities [1,40]. Hydrolytic enzymes are activated after the imbibition of water and decompose large molecular substances of reserve compounds such as starch, non-starch polysaccharides, and proteins in the endosperm, leading to increases in small molecular compounds such as glucose, oligosaccharides and amino acids in germinated seeds [1,2,41]. Apart from nutrition level changes, germination also generates antioxidant compounds such as ferulic acid, α-tocopherol, total phenolic and total anthocyanin contents [1,2,5,6]. Our results also showed that total phenolic content and antioxidant capacity of Kum Doi Saket rice seeds increased during germination for 5 days (Figures 2 and 3). These results correlate with Maisont and Narkrugsa [4] and Sutharut and Sudarut [6]. | |
| Besides ionic and osmotic effects, which are the primary stresses of salinity, high salt concentrations additionally induce secondary stresses such as oxidative stress due to the excessive generation of ROS [40,42,43]. To prevent oxidative damage, plants process both enzymatic mechanisms such as APX and CAT, and non-enzymatic mechanisms such as phenolic compounds for ROS scavenging [7,8,43]. Our study showed that the antioxidant capacity of Kum Doi Saket rice seeds significantly increased after being soaked in 75-300 mM NaCl solutions for 12 hours (day 0) as compared to that of the control (Figure 2). This suggests that salinity stress activates the antioxidative systems after the seeds absorbed the NaCl solutions. Seeds exposed to 75 and 150 mM NaCl showed higher antioxidant capacity than that of the control during germination for 5 days (Figure 2).This Indicates that salinity stress increases antioxidant capacity in germinated rice seeds. Similar results were found by Daiponmak et al. [14] and Chutipaijit et al. [15,16]. | |
| Total phenolic content of Kum Doi Saket rice seeds increased with germination time and significantly increased in germinated seeds in 150 mM NaCl (Figure 3). This result is consistent with Daiponmak et al. [14], Chutipaijit et al. [15,16], and Ghosh et al. [17] who found that total phenolic content increased in rice seedlings under salinity stress. The antioxidant properties of phenolic compounds have been reported by donating electrons or hydrogen atoms from their hydroxyl and carboxyl groups, thus inactivating lipid free radicals and preventing decomposition of hydroperoxides into free radicals [44,45]. We found a positive correlation between total phenolic content and antioxidant capacities by DPPH (with R2=0.808) and ABTS radical scavenging assays (with R2=0.844) (Figure 6). This Indicates the role of phenolic compounds as free radical scavengers in germinated Kum Doi Saket rice seeds. Similar results were found by Sutharut and Sudarat [6] who reported that germinated black glutinous rice cvs. Niew Dam and Hom Nil had a positive correlation between total anthocyanin content and Trolox Equivalent Antioxidant Capacity (TEAC). Although the antioxidant capacity from DPPH radical scavenging assay was lower than that of the ABTS radical scavenging assay, both methods showed the same trend of results (Figure 2). This is due to the steric effect inside the DPPH• molecule, resulting in difficulty for the large reactive antioxidant compounds to react with its radical site [46]. The result is compatible with Floegel et al. [9] who compared antioxidant capacities of 50 popular antioxidant-rich US foods and found that antioxidant capacities from DPPH were lower than those from the ABTS radical scavenging assay. | |
| APX is an enzyme that catalyzes the reduction of H2O2 to water by using the reducing power of ascorbate [8]. APX activity of Kum Doi Saket seeds germinated on agar without NaCl increased and had the highest activity on day 2 of germination time (Figure 4). This agrees with Yi et al. [47] who reported that APX activity of radish seeds increased with germination time and reached the highest peak on day 3 during germination. Huang and Song [48] also found that APX activity of maize embryos from germinating seeds increased with germination time and reached the highest peak at 60 hours after imbibition. The different times for the highest peaks of APX activities depends on the species of plant. We found that APX activity of seeds germinated under NaCl stress increased with germination time with the maximum on day 4, and a concentration of 150 mM NaCl showed the highest increase in APX activity (Figure 4). These results are consistent with Lee et al. [49] who reported a significant increase in the APX activity in leaves of rice cv. Dongjin with 150 mM NaCl stress for 3 days. Similar results were reported by Demiral and Turkan [18] in leaves of 4-week-old seedlings of rice cvs. Pokkali and IR28 treated with 120 mM NaCl for 1 week. Our results show that APX activity was downregulated during day 1-2, but elevated in day 3-5 by salinity (Figure 4). The possible explanation is that germinating rice seeds under salinity required a few days to increase the APX activity. This result is compatible with the work of Lopez et al. [50] who showed that APX activity in leaves of 10-day-old radish seedlings transferred to 100 mM NaCl was lower than that of the control in day 1-2, and became slightly higher in day 3. | |
| In addition to APX, CAT is also an enzyme that catalyzes the reduction of H2O2 to water. This enzyme does not require a reducing power and has a low affinity for H2O2, thereby mainly removing the high concentration of H2O2 [8]. In our study, CAT activity of NaCl treated germinated seeds was lower than that of the control since day 1. This reduction is caused by an increasing in some metabolites generated by salinity stress that can inhibit catalase activity, such as superoxide radicals [51] and hydroxyl radicals [52]. The highest activity of CAT on day 2 of the control suggested that during the first 2 days, the germinated rice seeds produced high amounts of H2O2 which was reduced by catalase. High concentrations of NaCl at 75, 150, and 300 mM significantly inhibited the activity of this enzyme (Figure 5). The inhibitory effect of salinity on CAT activity is consistent with the results of Lee et al. [49] who also found that CAT in leaves of rice cv. Dongjin seedlings was severely inhibited during 150 mM NaCl stress for 3 days. The over-production of H2O2 under CAT deactivation caused by high salinity resulted in high activity of APX. The decrease in CAT activity with an increase in APX activity under NaCl stress occurred in our study (Figures 4 and 5) and was also reported by Demiral and Turkan [18] who studied in rice cv. IR28. | |
| Conclusions | |
| Our results showed that the antioxidant properties of Kum Doi Saket germinated rice seeds significantly increased by salinity stress with 150 mM NaCl, although germination and growth were reduced. The increase of antioxidant properties activated by salinity correlates with an increase in the total phenolic content. We proposed that salinity treatments may be an alternative way to add nutritional value for consumption and for further pharmaceutical application of Kum Doi Saket germinated rice seeds. However, further studies about the effects of NaCl on the amount of other bioactive compounds in germinated rice, such as GABA, γ-oryzanol, and dietary fiber should be conducted. | |
| Acknowledgement | |
| A part of this research was supported by Faculty of Science, Chiang Mai University, Chiang Mai, Thailand. We thank Dr. J. F. Maxwell of the Department of Biology, Faculty of Science, Chiang Mai University for proof reading this manuscript. | |
References |
|
|
|
Relevant Topics
- Basmati Rice
- Drought Tolerence
- Golden Rice
- Leaf Diseases
- Long Grain Rice
- Par Boiled Rice
- Raw Rice
- Rice
- Rice and Aquaculture
- Rice and Nutrition
- Rice Blast
- Rice Bran
- Rice Diseases
- Rice Economics
- Rice Genome
- Rice husk
- Rice production
- Rice research
- Rice Yield
- Sticky Rice
- Stress Resistant Rice
- Unpolished Rice
- White Rice
Recommended Journals
Article Tools
Article Usage
- Total views: 17013
- [From(publication date):
September-2013 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 12027
- PDF downloads : 4986
