Review Article Open Access
Important Points to Attain Reproducible Sterility Assurance
| Hideharu Shintani* | |
| Faculty of Science and Engineering, Chuo University, Tokyo, Japan | |
| *Corresponding Author : | Hideharu Shintani Faculty of Science and Engineering Chuo University, 1-13-27, Kasuga, Bunkyo, 112-8551, Tokyo, Japan Tel: +81425922336 Fax: +81425922336 E-mail: shintani@mail.hinocatv.ne.jp |
| Received May 06, 2014; Accepted May 31, 2014; Published June 04, 2014 | |
| Citation: Shintani H (2014) Important Points to Attain Reproducible Sterility Assurance. Biochem Physiol 3:135. doi:10.4172/2168-9652.1000135 | |
| Copyright: © 2014 Shintani H. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Biochemistry & Physiology: Open Access
Abstract
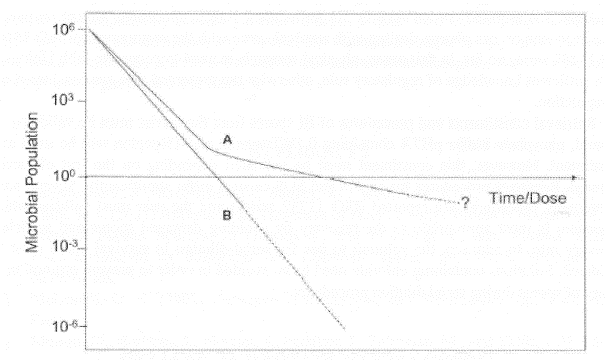
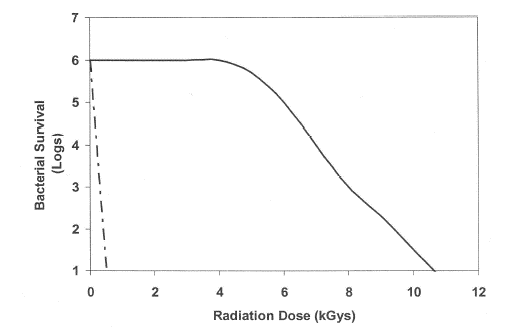
Many papers published and conducted so far by physical researchers on sterilization using gas plasma exposure. I as microbiologist consider that they overlooked the important points to avoid clumps (overlay of microorganisms, they called clumps as stacking, which is incorrect) in biological indicator (BI) and the importance considering statistical analysis. As a characteristic, microorganisms have themselves significant variation, so number of samples utilized must be necessary to be able to conduct statistical analysis, which concept differed from concept of physical science. The aim of gas plasma sterilization is the death of bioburden and the distribution of bioburden is not seen any clumps in exact status. The penetration depth of gas plasma is around 10 nm, so if BI has clumps (the thickness of BI is around 1 μm) death of inner layer may be hard to attain or delayed, resulting in tailing phenomenon of survival curve or failure of sterility assurance. Physical researchers so often prepare BI by themselves and the prepared BI has significant so-called stacking. As microbiologist calls physical researcher’s attention the importance to conduct experiment using BI free from clumps to attain correct and reproducible sterility result. Furthermore, it is necessary to attain sterility assurance together with material and functional compatibility, otherwise GMP requirement is not achieved.
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 | Figure 5 |
Relevant Topics
- Analytical Biochemistry
- Applied Biochemistry
- Carbohydrate Biochemistry
- Cellular Biochemistry
- Clinical_Biochemistry
- Comparative Biochemistry
- Environmental Biochemistry
- Forensic Biochemistry
- Lipid Biochemistry
- Medical_Biochemistry
- Metabolomics
- Nutritional Biochemistry
- Pesticide Biochemistry
- Process Biochemistry
- Protein_Biochemistry
- Single-Cell Biochemistry
- Soil_Biochemistry
Recommended Journals
- Biosensor Journals
- Cellular Biology Journal
- Journal of Biochemistry and Microbial Toxicology
- Journal of Biochemistry and Cell Biology
- Journal of Biological and Medical Sciences
- Journal of Cell Biology & Immunology
- Journal of Cellular and Molecular Pharmacology
- Journal of Chemical Biology & Therapeutics
- Journal of Phytochemicistry And Biochemistry
Article Tools
Article Usage
- Total views: 14408
- [From(publication date):
August-2014 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 9792
- PDF downloads : 4616
