Research Article Open Access
Magnitude of Changes in the Activity of Amylases and Cellulase and its Association with the Biochemical Composition during Maturation and Ripening of Banana (Musa spp.)
| Theji Mohan1, Rajesh PN2*, Fathimathu Zuhra K3 and Vijitha K3 | |
| 1Department of Biochemistry, St Mary’s College Thrissur, Kerala, India | |
| 2Department of Biochemistry, Jimma University, Jimma, Ethiopia | |
| 3Department of Life Sciences, University of Calicut, Kerala, India | |
| *Corresponding Author : | Dr. Rajesh PN Assistant Professor, Department of Biochemistry Jimma University, Jimma 378, Ethiopia Tel: +251 47 111 1458 E-mail: rajnarayananp@yahoo.com |
| Received January 10, 2014; Accepted February 28, 2014; Published March 05, 2014 | |
| Citation: Mohan T, Rajesh PN, Zuhra KF, Vijitha K (2014) Magnitude of Changes in the Activity of Amylases and Cellulase and its Association with the Biochemical Composition during Maturation and Ripening of Banana (Musa spp.). Biochem Physiol 3:127. doi:10.4172/2168-9652.1000127 | |
| Copyright: © 2014 Mohan T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Biochemistry & Physiology: Open Access
Abstract
Introduction: Banana fruit (Musa spp.) is world’s most popular and cheaply available fruit item. Banana fruit maturation and ripening takes place as a result of interplay of several degrading and synthetic enzyme and is responsible for biochemical and morphological variations during fruit maturation and ripening. Objectives: This study was carried out in the “Nendran” variety of Musa spp. to investigate the activity of two major degrading enzymes – amylases and cellulase during fruit maturation and ripening. Amylases (which include both alpha amylase and beta amylase) are the major category of starch degrading enzymes and cellulase is responsible for the cell wall (cellulose) degradation in fruit tissue that results in tissue softening. The changes in the concentration of various biochemical constituents were also investigated. Materials and methods: Fresh samples of fruits (“Nendran”, local name) were collected at particular intervals directly from plants starting from the very young stage (10th day) up to the over ripe stage (102nd day) and were extracted for the crude enzyme preparation. Specific activities of amylases and cellulase were analyzed by standard assay procedures. The concentration of total carbohydrate, starch, cellulose, fructose, total protein and dry recovery percent were also determined. Results and discussion: Significant variations in the specific activity of amylases and cellulase were observed between different periods of maturity. The activity of amylases was found to be very low (4.8 ± 0.26 Units) during the tender stage, remained low up to 90th days (14.00 ± 0.86 Units) of maturation, but when the fruits starts ripening after 90 days it increased to about 10 folds on the 92nd day (103.40 ± 8.20 Units) and then gradually decreased towards the overripe stage ie. on the 102nd day (5.78 ± 0.37 Units). Similarly, the specific activity of cellulase was also significantly changed; the peak activity was observed on the 90th day (769.00 ± 18.54 Units) that was found to be several times high as compared to the tender stages (78.90 ± 3.26 Units on the 10th day and 21.00 ± 1.26 Units on the 20th day), facilitating the process of wall degradation. The activity of amylase and cellulase had significant correlations with the concentrations of related biochemical constituents such as starch and cellulose. Conclusions: Amylases and cellulase had significant roles to play towards the morphological and biochemical transformations associated with the fruit maturation and ripening processes, such as starch accumulation, starch degradation and tissue softening in banana fruit (“Nendran”).
| Keywords |
| Banana; Nendran; Amylase; Cellulase; Specific activity; Starch; Cellulose; Total protein; Dry recovery |
| Introduction |
| Bananas and plantains constitute the fourth largest and popular fruit crop in the world following grapes, citrus fruits and apple. The Banana plant (Musa spp.) often erroneously referred to as a tree is a large herb with succulent very juicy stem (properly, pseudo-stem) which is a cylinder of leaf petiole sheaths reaching a height of 6-15 feet and arising from a fleshy rhizome. A banana plant bears fruits from 10- 12 months after planting. The inflorescence is a spike originating from the rhizome. As the young fruits develop from female flowers, they appear as angled slender green fingers during growth reaching harvest maturity usually 90-120 days after flowering [1]. |
| The fruit technically a berry turns from deep green to yellow during ripening. The fruits range from 6.4 to 30 cm in length and 0.75 to 2 inches in width. The flesh is ivory white to yellow or salmon yellow in color, may be firm astringent even gummy with latex when unripe and turning mellow or rather dry and mealy or starchy when ripe [2]. The flavor may be mild and sweet or sub-acid with a distinct apple tone. Fruit ripening is generally programmed, highly co-ordinate process of transformation from unripe to ripe stage to yield an attractive edible fruit with an optimum blunt of color, taste, aroma, and texture [3]. Fruits are usually harvested when 70% mature. The fruit is mainly composed of water and carbohydrate and has almost negligible content of protein and fat. The ripe banana is essentially sugary and easily digestible food. The unripe fruit contains more starch and less sugar than ripe fruit. Generally a large banana has 2 grams of proteins and 4 grams of fiber. The starch in green banana is replaced by sucrose, glucose and fructose in the ripe fruit. Maltose has been detected in small amounts. Banana is considered as an important food to boost the health of malnourished children. The fruit is rich in vitamin A and vitamin C. A full range of B vitamins such as thiamine, riboflavin, niacin vitamin B6 are also present. Among the minerals, calcium, magnesium, iron, phosphorus, potassium and zinc are the major ones. Banana peel contains beta-sistosterol, stigma-sterol campe-sterol cycloeucalenol, cycloartenol and 2-4 methylene cycloartenol. All these principles have variety of bioactivity and have a lot of applications in traditional medicine [4]. Moreover banana is a cooling fruit that can lower both physical and emotional temperatures [1]. |
| The ripe banana fruit is utilized in a magnitude of ways in human diet from simply being peeled and eaten out of hand to being sliced and served in fruit cups, salad and sandwiches, and also incorporated in to ice creams and bread. Flour can be derived from dry fruits and used for pastries and mixed with other flours. Dry fruits of both banana and plantain are commonly made in to chips by frying slices in oil and salting. In African countries ripe bananas are used in the preparation of wines. Banana peel is used as a cattle feed. The green leaves are used as plates and wrapping food for cooking and storage [5]. |
| The present work was under taken on banana fruits (Musa spp., “Nendran”-local name). This is one of the commonly cultivated variety in Kerala on a commercial basis irrespective of altitude variations. Nendran is very popular as a fruit and is particularly on demand for making chips and also varieties of curries as a part of traditional food habit in Kerala. Fruit maturation and ripening in banana is a result of the coordinated functioning of various enzymes and a number of studies have already been conducted in this regard on so many banana varieties. [6,7]. However, in this regard no studies have been conducted so far on “Nendran”, one of the very popular and commercial verities cultivated in the southern states of India. The major objective of the study was into understand the magnitude of changes in the activities of enzymes such as amylases (alpha amylase and beta amylase together) and cellulase during the maturation and ripening of banana fruits. The simultaneous changes in the concentration of biochemical constituents such as total carbohydrate, starch, cellulose, fructose, total protein, percentage of dry recovery etc. were also investigated. |
| Materials and Methods |
| The fruit samples used for analyses were obtained from an agricultural farm at Thenhipalam (Figure 1), near to our laboratory at the Department of Life Sciences (University of Calicut, Kerala, India), where the investigations have been carried out. Six banana plants were selected for sample collection. The average day temperature in this area was 30 degree during that time. The selection of plants was based on the following criteria. The plants were observed to be healthy and the fruit formations (fruit outbreaks) were on the same day. |
| Collection of fruit samples |
| The first set of fruit samples were collected directly from each plant on the 10th day of initiation of fruit development (fruit outbreak) by using a sharp knife (Figure 2b). Special care was taken not to damage the nearby fruits (fingers) in the same bunch. This was followed by regular sampling at an interval of 10 days (20th, 30th and so on.) until the 90th day when the fruit became fully mature (Figure 2a). Analyses of the samples for enzymes were done on the same day of sample collection. On the 91st day fruits were harvested as a whole and kept in a wooden box covered with jute bags at room temperature. The temperature range was observed to be 25-31 degree during the storage period. From this stored fruits, samples were collected and analyzed on every alternative day (92nd, 94th, 96th, 98th, 100th and 102nd) until the overripe stage (102nd) when the fruit starts decaying (Figure 2b). |
| Enzyme analysis |
| Analysis of the enzymes activity was done on the same day of sample collection using the fresh sample. In this study specific activity of amylases and cellulase were carried out using standard procedures [8]. The procedure was standardized initially for banana fruits before the actual analysis was carried out. |
| Determination of specific activity of amylase |
| The specific activity of amylase was determined by colorimetric measurement of the reducing sugar liberated in the reaction system –by using Dinitro- salisylic acid reagent (DNS) [9,10]. |
| Reagents: Sodium acetate buffer (0.1 M, pH 7), Starch 1% soln. in acetate buffer, DNS reagent, 40% Rochelle salt soln. and Phosphate buffer (0.1 M , pH .7) were the reagents used for the preparation of the extract, reaction medium and the following colorimetric procedure. |
| Enzyme extraction: Extracted 2 gms of fresh sample of the fruit with 5 ml of phosphate buffer using a mortar and pestle. The extract was centrifuged and the supernatant was used as a source of amylase. All operations were carried out at 4 degree. |
| Procedure: The reaction system was prepared by mixing 1% starch solution and 1 ml of properly diluted enzyme in a clean test tube (15 ml capacity). The tube was kept at 27 degree for 15 minutes so that the amylase will act on the starch to liberate maltose or glucose in the medium. The reaction was stopped by adding 2 ml of DNS reagent (DNS is an enzyme denaturing agent as well). 1 ml of starch solution taken in another test tube in which the enzyme solution was added at the end of the incubation time just prior to the addition of the DNS reagent, served as the control. 1 ml of starch solution, 1 ml of phosphate buffer and 2 ml of DNS reagent served as the blank. 1 ml of starch solution, 0.9 ml of phosphate buffer, 0.1 ml of maltose solution (1 mg/ ml) and 2 ml of DNS reagent served as the standard tube. All tubes were kept in a boiling water bath for 5 minutes. When the tubes were warm, we added 1 ml of Potassium-Sodium-Tartrate solution (Rochelle salt). The tubes are then cooled in running tap water and made up to 10 ml by the addition of 5 ml distilled water. Measured the absorbance in a spectrophotometer Shimadzu UV- Visible, Japan) at 560 nm. The specific activity of the enzyme was calculated from the amount of reducing sugar liberated and the protein concentration in the extract (Lowry’s method).The specific activity was expressed as micro grams of maltose liberated for mg protein /minute. |
| Specific activity of cellulase |
| The concentration of reducing sugar (D- Glucose) released by the cellulolytic activity of cellulose was measure by DNS method [9]. |
| Reagents: Sodium citrate buffer (0.1 M, pH 5), Carboxy-methyl cellulose (CMC) - 1% solution in buffer, DNS reagent, 40% Rochelle salt solution and Phosphate buffer (0.1M , pH 7) were the reagents used for the preparation of the reaction medium and the following colorimetric procedure. |
| Enzyme extraction: Extracted 2 grams of fresh sample of the fruit with 5 ml of Citrate buffer (pH.5) using a mortar and pestle. The extract was centrifuged at 4 degree and the supernatant was used as a source of amylase. All operations were carried out at 4 degree. |
| Procedure: The reaction system was prepared by mixing 0.45 ml of 1% CMC solution and 0.05ml of enzyme extract in a clean test tube (15 ml capacity). The tube was kept at 37 degree (optimum temperature for cellulose) for 15 minutes so that the cellulase will act on the CMC to liberate glucose in the medium. The reaction was stopped by adding 2 ml of DNS reagent. 0.45 ml of 1% CMC taken in another test tube in which 0.05 ml of the enzyme extract was added at the end of the incubation time just prior to the addition of 2 ml DNS reagent , served as the control. 0.45 ml of 1% CMC, 0.05 ml of citrate buffer and 2 ml of DNS reagent served as the blank. 0.45 ml of 1% CMC 0.05 ml of Glucose solution (1 mg / ml) and 2 ml of DNS reagent served as the standard tube. All tubes were kept in a boiling water bath for 5 minutes. When the tubes were warm we added 1 ml of Rochelle salt solution. The tubes were then cooled in running tap water and made up to 10 ml by the addition of 5 ml distilled water. We also measured the absorbance in a spectrophotometer at 540 nm. The specific activity of the cellulase enzyme was calculated from the amount of D- Glucose liberated and the protein concentration in the extract (Lowry’s method). The specific activity was expressed as micro grams of Glucose liberated for mg protein /minute. |
| Measurement of dry recovery |
| Fruit samples collected during each stage is peeled and weighed. It was then placed in hot air oven and dried at 55 degree. Drying was continued until the samples showed consistent weight. From the initial weight and dry weight the percentage of dry recovery was calculated. The dry samples were properly packed in polythene bags and kept in a BOD incubator to avoid any fungal growth. These samples were then used for the analyses of various biochemical constituents. |
| Biochemical constituents |
| The dried samples were powdered and used for estimating the concentrations of biochemical constituents such as total carbohydrate, starch, fructose, cellulose and total protein [9]. |
| Total carbohydrate: The total carbohydrate content in the samples was determined calorimetrically by Anthrone method. This method was colorimetric and involved the use of a Spectrophotometer (Model- Zhimadzu). 100 mg of the powdered sample was accurately weighed using a microbalance and was used for the estimation of total carbohydrate. Starch: Starch content was also estimated calorimetrically by Anthrone method [9,11]. 100 mg of the powdered sample was accurately weighed for the estimation of starch. Fructose: The estimation of fructose was done calorimetrically by using resorcinol reagent [9]. |
| Cellulose: Anthrone method was used for the colorimetric estimation of cellulose in the sample [9]. |
| Protein: The total protein content was estimated calorimetrically by Folin- Lowry’s method [12]. |
| Statistical analyses |
| The data was analyzed by student’s “t” test. All the values are expressed as Mean + SD. P values of 0.05 or less were considered significant. All analyses were performed on computer using the statistical package SPSS. |
| Results |
| Analysis of enzymes - amylases and cellulase |
| The changes in the activity of total amylases (alpha amylase + beta amylase) are given in Table 1a. |
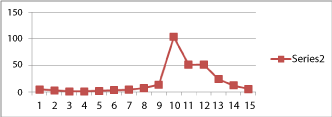
| Amylases are important in the degradation of starch in to soluble sugars which impart the sweet taste to ripe fruit. The specific activity of amylases was very low during the initial stage ( 4.8 ± 0.26 Units, 10th day) fruit development and was not undergone very significant change up to 70th day of fruit maturation (4.40 ± 0.26 units ). A significant elevation in amylase activity was noticed on the 92nd day (103.40 ± 8.20* Units) and was the highest level noticed during the complete process of fruit maturation and ripening. The activity then gradually decreased from 94th (51.50 ± 3.35* Units) to 102nd day (5.78 ± 0.37Units) when the fruit became overripe and started decaying (Chart 1a). |
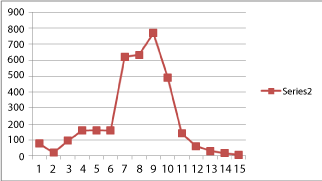
| The specific activity of cellulase at different stages of maturation and ripening of banana fruit is given in Table 1b. Relatively higher levels of cellulose activity was noticed during the early stages (78.90 ± 3.26* Units, 10th day). The activity was found to be slightly decreased by the 20th day (21.00 ± 1.26* Units). It then gradually increased from 30th day (96.00 ± 5.20* Units) up to the 60th day (160.00 ± 7.54* Units). A sudden increase was noticed on 70th day (620.00 ± 22.73* Units), however, the peak activity was noticed on the 90th day (769.00 ± 18.54* Units) and was around 10 times that of the 10th day. A significant fall in the specific activity of cellulase was observed from the 92nd day (490.00 ± 12.66* Units) towards the 102nd day (7.45 ± 0.44* Units) ie. during fruit ripening (Chart 1b and Chart 2). |
| Dry recovery |
| Dry recovery percentages of fruit at different stages of maturation and ripening were calculated and the results are indicated in Table 2. |
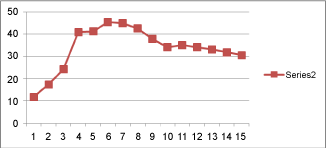
| The dry recovery percent was very low on the 10th day (11.82 ± 0.76*) gradually increased during fruit maturation to reached the peak level on the 60th day (45.43 ± 1.3*). A significant decrease in dry recovery percent was observed towards the later stages of maturation and ripening (37.82 ± 0.85* on the 90th day and 30.53 ± 0.80* on the 102nd day). |
| Analysis of biochemical constituents |
| The percentage of total carbohydrates, starch, fructose, cellulose and total protein are indicated in Table 3. The total carbohydrate content was less up to the 20th day of maturation (18.5 ± 0.22), but shoot up by 30th day (44.38 ± 1.2*), reached the peak level by the 60th day (52.70 ± 1.0*), and gradually decreased towards the ripe (46.90 ± 0.25* on 98th day) and overripe stages (44.85 ± 0.19 * on 102nd day). |
| The concentration of starch was also very negligible during the initial stage (3.25 ± 0.17 mg%) gradually increased during maturation and peak level was achieved by 80- 90 day (38 / 38.88 mg%). A sudden decline in the starch content was observed during ripening and it reached a very low level in the overripe stage (3.25 ± 0.12 mg%, on 102nd day). |
| The concentration of cellulose was found to be relatively high at the tender stage (12.00 ± 0.18 mg% on the 10th day and 10.00 ± 0.23 mg% on the 30th day) and decreased during fruit maturation (7.06 ± 0.17*mg % on the 60th day and 9.75 ± 0.09* mg% on 90th day). The cellulose concentration was found to be decreased significantly in the ripe and overripe stage (4.25 ± 0.08*mg% on the 100th day and 3.75 ± 0.11* mg% on the 102nd day). |
| The concentration of fructose was very low up to 92nd day (1.75 ± 0.10 mg% on the 10th day and 4.04 ± 0.11* mg% on the 92nd day) and a significant increase was noticed from 96th day (30.20 ± 0.09 mg%) onwards and reached the peak level on the 102 day (36.65 ± 0.11* mg%). |
| The total protein content was found to be relatively high during the initial stage (2.75 ± 0.13* mg % on 10th day) and then gradually decreased towards maturation (0.88 ± 0.15*mg% on the 70th day). A gradual increase was then noticed during the start of the ripening process (1.38 ± 0.10mg % on 90th day and 2.75 ± 0.13*mg % on 94th day), followed by a decline towards the over-ripe stage (1.85 ± 0.17*mg % on the 102nd day). |
| Discussion |
| The maturation and ripening of banana fruit is the result of interplay of various enzymes whose activities may vary with time resulting in an overall change in the biochemical composition. The major morphological changes during fruit maturation and ripening were the change in size shape and in color. The small curved fruit was transformed in to a long slender one. A gradual change in color from green (up to 40th day) to light green (40th to 80th day) yellowish green (80th to 90th day) and yellow (90th to 102nd day) was observed. These physical changes are a result of a large number biochemical transformations taking place in the banana fruits during its maturation and ripening. As far as banana fruit is concerned, the immature stage (tender fruits) is characterized by less starch and when it matures accumulation of starch occurs. When the fruit started ripening starch will be degraded in to soluble sugars mainly fructose, the cell wall will be degraded and as a result the fruit become sweet and soft [13-15]. There will be a change in the pigmentation also. The tender and mature fruits are externally green in color, but the ripe ones is usually light yellow. Inside, tender and mature fruits are pale that change in to yellowish or slightly reddish in color [2]. The overall change in morphology and biochemical composition is a result of the activity of various enzymes. |
| The present study conducted in the “Nendran” local variety of banana (Mussa spp.), we have analyzed the specific activity of the two major enzymes which are playing pivotal roles in the maturation and ripening of banana, include amylases (both alpha-minor, and betamajor) and cellulase. Relatively very low activity of amylase until 80th day of fruit maturation (4.8 ± 0.26 Units on 10th day and 7.50 ± 0.28* Units on 80th day) was responsible for the accumulation of starch, by indirectly favoring its and accumulation, resulting in an increase in fruit size. A very high activity of amylase was noticed from 92nd (103.40 ± 8.20* Units) up to 96th (51.78 ± 2.26* Units)) day and was responsible for the degradation of starch during the early stage of fruit ripening process. Amylase activity then gradually decreased towards the overripe stage (12.67 ± 1.30* Units on 100th day and 5.78 ± 0.37 on 102nd day) by that time starch has already been transformed in to soluble sugars. The low activity during the initial stages of fruit development and fruit maturation and the significantly high activity during the stages of fruit ripening clearly established the role of amylase enzyme in the transformation of starch in to soluble sugars in banana fruits. This finding has been supported by several previous studies. A change in the activity of amylases during ripening of banana was previously reported [14]. There are also earlier reports about starch degradation and sugar transformation during the maturation and ripening of banana [16]. The inhibitory effect of IAA on the activity of amylase enzyme and starch degradation in banana was already being studied [17]. Previous studies on the starch granules in fruit tissue of banana revealed that the size of the granule is decreased during ripening in which alpha and beta amylases are involved [18]. Bassinello et al. [19] demonstrated the amylolytic activities in fruits using banana as a model. They found that mature banana can achieve 20% of its starch content starch which is being degraded during the ripening period in a complex process involving alpha and beta amylases. Garcia et al. also observed that starch transformation during banana ripening is a result of increase in the amylolytic activity by two enzymes amylase and glucosidase. There is also report on an increase in the expression of beta amylase gene during ripening process as evidenced by an increase in the concentration of the corresponding mRNA [20]. However, no studies had been conducted in the “Nendran” variety so far. |
| It has already been reported that starch phosphorylase also has a similar pattern of activity as amylases during fruit maturation and ripening of banana [21]. There are also similar reports on changes in the activity of starch phosphorylases during the ripening of banana [13]. Starch synthase is the enzyme responsible for the starch synthesis during starch accumulation phase of maturation. Amylase acts opposite to this enzyme. There are earlier reports that during fruit ripening, ethylene (plant hormone) production triggers a developmental cascade that is accompanied by a massive conversion of starch sugars and an increase in the rate of protein synthesis [22]. An increase in the activity of degrading enzymes such as amylases (alpha and beta types), starch phosphorylase acid phosphatase and oxidative enzymes such as peroxidase and catalase was noticed during banana ripening [23]. The role of enzymes such as amylases [17,24-26], cellulase [27,28] hemicellulases [27], starch phosphorylase [21] sucrose phosphate synthase, sucrose synthase [15,29], Glucosidase [14], etc. during fruit ripening were already being reported. Thus the starch content in banana fruit at any stage is a function of three enzymes- Amylase (alpha and beta amylases) , starch synthase and starch phosphorylases, all having more or less equal roles. In the present study a correlation was found to exist between the amylase activity and starch content in fruits at different stages of maturation- tender, mature and ripe. Differential expression of genes concerned with the synthesis of enzymes, both synthetic (starch synthase) and degrading (amylases and starch phosphorylase) plays a key role that results in maturation and ripening of banana fruits as evident from the present investigation. |
| Another enzyme studied was cellulase which is responsible for the cell wall degradation and tissue softening during ripening (together with pectinases). The cellulase activity was found to be very low in the tender ones (21.00 ± 1.26* Units on the 20th day). From about 70th day, there noticed a significantly very high cellulase activity (620.00 ± 22.73* Units on 70th day and 632.00 ± 20.22* on 80th day) resulting in an increase in the rate of cellulose degradation. This could be the reason why a higher percentage of cellulose is noticed in the tender stage and the cellulose percentage gradually decreases on maturation. The peak activity of cellulose was noticed on 90th day (769.00 ± 18.54* Units) ie. during the onset of ripening process. During ripening process the cellulase activity gradually falls to 7.46 units in the over-ripe sample (102nd day of maturation). This can be due to the usual process of inactivation as well as non-synthesis of the enzymes associated with cell disruption and cell death. These findings clearly proved the role of cellulase enzyme in the degradation of cellulosic cell wall of the fruit tissue resulting in tissue softening. There are previous reports about the role of hydrolases mainly cellulases, and pectinases in fruit during ripening process of bananas [30]. This was also in line with the pattern of cellulase activity observed by Abu-Sarra et al. [31] in Mango fruits. |
| Starch is the major carbohydrate present in mature banana fruits and makes the major percentage of the total carbohydrate content. There observed a change in the concentration of starch at different levels of maturation and ripening. During ripening the starch will be transformed in to soluble sugars and then to fructose. The present study revealed that the starch concentration at any stage of fruit maturation and ripening had an inverse relationship with the activity of amylase enzyme. During stages of maturation and starch accumulation the amylase activity was very low (7.50 ± 0.28* Units on 80th day) and the starch content was observed to be very high (38.00 ± 0.31*mg % on 80th day) But, during the onset of fruit ripening the starch concentration gradually decreased (24.44 ± 0.28* mg %on the 92nd day) associated with a significant elevation in amylase activity (103.40 ± 8.20* Units, on the 92nd day). These findings clearly established that there is a clear-cut relationship between amylase activity and starch degradation during fruit ripening in banana. There are earlier report about an inverse correlation between the activity of amylase and starch content in avocado fruit pulp [32]. |
| A gradual decrease in the percentage of cellulose was observed from the young fruit stage (12.00 ± 0.18 mg % on 10th day) to overripe stage (3.75 ± 0.11 mg% on 102nd day). But the level was observed to be stabilized in between 60 and 90 days of maturity i.e. in the mature stage. This stage showed an increased dry recovery percent and might be due to the increased cellulose (major component of crude fiber) and starch content. Total carbohydrate, starch and cellulose all has its peak level from 60 – 90 days of maturity. These observations are all in line with the previous observations made by many others. It was reported that the concentration of cellulose is decreased during the ripening of banana fruits [33]. There are similar reports that there is a decrease in the concentration of cellulose in the cell wall during the ripening fig fruits [34]. A gradual decrease in the concentration of cellulose towards ripening can be correlated with an increase in cellulase activity discussed before. |
| Fructose is commonly known as the fruit sugar. The concentration of fructose remained low during the entire period of fruit maturation till the beginning of ripening process (1.75 ± 0.10 mg% on the 10th day and 4.04 ± 0.11*mg% on the 92nd day). But, there noticed a sudden increase in fructose content by 96th day (30.20 ± 0.09 mg%) reaching the maximum level on 102nd day (36.65 ± 0.11* mg %), imparting the sweet taste. The sweetness of banana fruit is mainly due to the high concentration of fructose formed during ripening. The sudden increase in the concentration of fructose during ripening was associated with the degradation of sucrose and starch resulting in an overall increase in the concentration of soluble sugars particularly of Fructose. It has been already reported that ripening of banana fruit is associated with an increase in the rate of starch breakdown and a simultaneous increase in concentration of soluble sugars such as glucose, fructose and sucrose [15]. There are similar report that there will be an increase in the concentrations of sucrose and soluble sugars during the ripening of plantains and bananas [35]. The liberation of soluble sugars during ripening can be the result of the activity of several enzymes including starch phosphoylase, amylase etc. discussed before. |
| A gradual reduction in the total protein content towards maturation (0.88 ± 0.15* mg% on the 70th day) compared to the tender stages (2.75 ± 0.13* mg% on 10th day) might be due to the relatively high concentration of starch during these stages. But, towards ripening stage the protein concentration was found to be relatively high (1.38 ± 0.10 mg % on 90th day and 2.75 ± 0.13* mg% on 94th day), and might be due to the activation and expression of many of the genes resulting in an overall increase in the concentration of enzyme proteins associated with the process of fruit ripening. These observations are in agreement with previous reports. There are earlier studies which revealed an increase in the rate of protein synthesis during the ripening of banana fruits [36]. An increase in protein concentration was also reported during the ripening of banana [37]. The decrease in the protein content in the overripe fruits might be because of the usual process of inactivation/ degradation /non-synthesis of the protein by cell death and tissue damage associated with the onset of decaying process. |
| The pattern of changes in the biochemical composition during this study is in line with the report of Spiny cacti [38] who observed that the starch level decreases, monosaccharide increases and protein concentration increases and then decreases during banana ripening. The most significant observation is that a very good inverse correlation was found between the starch content and amylase activity and also between cellulose content and cellulase activity at various stages of maturation. For example, the amylase activity was very less up to 90th day of maturity during that time the deposition of starch has taken place, but after 90th day there is a sudden increase in amylase activity associated with ripening process that resulted in starch degradation and the starch content during this stage was found to be too low. Similarly, the cellulose content was very high during young stages, gradually decreased, during maturation and reached a very low level during ripening. This is associated with a sudden increase in cellulase activity by about 80- 90 days of maturation that is just before the start of the actual ripening process. The cellulose content of the ripe fruit was found to be very negligible (3.75 mg%). |
| The analysis of two of the important degrading enzymes (cellulase and amylases) and the estimation of biomolecules such as starch, cellulose, fructose, total carbohydrate and protein provided a more clear insight in to the maturation and ripening of banana fruit (Nendran). It has been revealed that one reason behind the maturation and ripening is a change in the activity of amylases and cellulase that results in subsequent biochemical and morphological changes. This in turn is a result of differential gene expression during maturation and the early days of the actual ripening process. A change in gene expression pattern during banana ripening was studied by analyzing the mRNA content [39]. Recently, expression pattern of a set of genes have been studied by reverse Northern blotting technique and established that certain genes are specifically expressed at the time of ripening of banana and mango [40]. |
| Conclusion |
| The study conducted in ‘Nendran ‘(local name) variety of banana (Musa spp.) clearly revealed that fruit maturation and ripening in banana is a result of large number of biochemical changes particularly with respect to the carbohydrate metabolism. These biochemical changes transform a tender fruit to a mature one and then the mature fruit in to the ripe one. Cellulase and amylases play an important role in the maturation and ripening of banana. |
| The findings of the present investigation can be summarized as follows: |
| • Low levels of amylase activity during the early stages of fruit development leads to the accumulation of starch and increase in the fruit size. |
| • Increase in the activity of amylases after maturation (during the onset of ripening) results in the conversion of starch in to soluble sugars that lead to ripening of the fruits and its sweet taste. |
| • Increased accumulation of fructose during ripening leads to further sweetening of the ripe fruit. |
| • Increased activity of cellulase during the initial phase of ripening leads to cell wall degradation and tissue softening in the ripe fruit. |
| A significant increase in protein content just before the start ripening revealed that certain genes concerned with the ripening process are activated at that time and is important in the ripening process. |
| All the biochemical changes associated with fruit maturation and ripening can be attributed to differential gene expression particularly of certain enzymes and is to be investigated further in detail. |
| Acknowledgement |
| The authors are thankful to the Head of the Department and all other staff of the Department of Life Sciences, Calicut University, Thenhipalam, Kerala, India, for their co-operation and encouragement. |
References
|
Tables and Figures at a glance
| Table 1a | Table 1b | Table 2 | Table 3 |
Figures at a glance
 |
 |
| Figure 1 | Figure 2 |
 |
 |
 |
| Chart 1a | Chart 1b | Chart 2 |
Relevant Topics
- Analytical Biochemistry
- Applied Biochemistry
- Carbohydrate Biochemistry
- Cellular Biochemistry
- Clinical_Biochemistry
- Comparative Biochemistry
- Environmental Biochemistry
- Forensic Biochemistry
- Lipid Biochemistry
- Medical_Biochemistry
- Metabolomics
- Nutritional Biochemistry
- Pesticide Biochemistry
- Process Biochemistry
- Protein_Biochemistry
- Single-Cell Biochemistry
- Soil_Biochemistry
Recommended Journals
- Biosensor Journals
- Cellular Biology Journal
- Journal of Biochemistry and Microbial Toxicology
- Journal of Biochemistry and Cell Biology
- Journal of Biological and Medical Sciences
- Journal of Cell Biology & Immunology
- Journal of Cellular and Molecular Pharmacology
- Journal of Chemical Biology & Therapeutics
- Journal of Phytochemicistry And Biochemistry
Article Tools
Article Usage
- Total views: 15342
- [From(publication date):
June-2014 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 10680
- PDF downloads : 4662
