Review Article Open Access
New Vaccines Introduction in Nigeria: Catalyst for Improving Immunization Coverage and Child Survival
| Muhammad and Nuhu A* | |
| National Primary Health Care Development Agency (NPHCDA), Abuja, Nigeria | |
| Corresponding Author : | Nuhu A National Primary Health Care Development Agency Abuja FCT, Nigeria Tel: 234 802 303 1206 E-mail: adamunuhu2001@yahoo.co.uk |
| Received February 09, 2015; Accepted May 28, 2015; Published June 01, 2015 | |
| Citation: Muhammad, Nuhu A (2015) New Vaccines Introduction in Nigeria: Catalyst for Improving Immunization Coverage and Child Survival. J Pain Relief 4: 185. doi: 10.4172/2167-0846.1000185 | |
| Copyright: © 2015 Muhammad et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Pain & Relief
Abstract
Most recently the Nigeria immunization programme has reported improvements in its routine immunization coverage, further propelled by the introduction of new vaccines (Pentavalent, PCV, IPV, Rotavirus vaccines etc.). This will further strengthen the country’s efforts to significantly reduce childhood mortality and accelerate the achievement of the MDG 4. Also, there were only 6 cases of wild poliovirus cases reported in 2014; a drastic reduction in cases compared to the previous years. The new vaccines when completely introduced will help avert an additional 486,957 deaths over a 6-year period.
| Keywords |
| New vaccines; Vaccine-preventable diseases; Vaccination coverage |
| Introduction |
| The Federal Government of Nigeria (FGoN) through the National Primary Health Care Development Agency (NPHCDA) supports and provides leadership for the development of primary health care (PHC) system in Nigeria. The Agency has 7 goals as follows: control preventable diseases; improve access to basic health services; improve quality of care; strengthen institutions; develop high performing health workforce; strengthen partnerships; and strengthen community engagement. It achieves these goals by supporting States and local government areas (LGAs) in developing sustainable system of PHC services that are accessible, affordable and of good quality through the participation of individuals, families and communities in partnership with government and non-governmental organizations (NGOs) [1]. |
| In the recent past, some major accomplishments have been recorded in the increasing coverage for routine immunization (RI), Midwives Service Scheme (MSS), Measles Control, and the Polio Eradication Initiative (PEI) [1]. The improvement in the RI coverage has immensely contributed to the reduction in the morbidity and mortality from vaccine-preventable diseases (VPDs). For instance; the sustained OPV3 coverage >80% is most probably responsible for the significant reduction in the WPV transmission in the country; sustained improvement in the measles RI coverage >80% in most States is also responsible for the reduced morbidity and mortality from measles cases nationwide [2]. Some of the key factors responsible for improved RI performance in the country are sustained vaccine availability; the overhauling of the immunization system in general, coordinated planning in collaboration with our development partners including other key stakeholders, and the new vaccine introduction in the country. The NPHCDA has ensured that this is on-going on a sustainable basis. |
| New vaccines introduction (NVI) |
| Infectious diseases are still responsible for nearly 30% of all deaths worldwide; more than 15 million people die every year, mostly in low-income and middle-income countries. The Global Immunization Vision and Strategy (GIVS) 2006-2015, for fighting VPDs has 3 priority objectives: (1) immunize more people against more diseases (2) introduce a range of newly available vaccines and technologies, and (3) provide a number of critical health interventions through immunization [3]. |
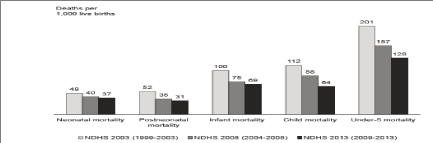
| The Millennium Development Goals (MDGs) prescribes attainment by 2015, of an under 5-mortality rate of not more than 75/1000 live births (MDG4). The 2008 National Demographic & Health Survey (NDHS) estimated Nigeria’s under 5-mortality rate to be 157, declining at a rate of 1.2% annually [4]. This rate of progress was deemed insufficient to keep the country on track to achieve MDG4. Nigeria must therefore commit to and implement high impact interventions aimed at reducing the child deaths (Figure 1). The country’s effort of the new vaccines introduction is supported by the Global Alliance for Vaccines and Immunization (GAVI). |
| To achieve the MDG 4 targets (70 U-5 deaths per 1,000 live births by 2015), Nigeria needs prompt action to drive down U-5 mortality by ~ 30% (from the current 128/1000 live births). As Nigeria’s population is approximately 16% of Africa’s, our attainment of the health MDG targets will significantly improve the health outcomes in Africa as a whole [5]. |
| Pentavalent vaccine introduction: The Pentavalent vaccine is a combination vaccine that protects infants against the following 5 diseases: diphtheria, pertussis, tetanus, hepatitis B and Haemophilus influenzae type b (Hib) pneumonia and meningitis. Prior to 2012, the Nigeria RI schedule consisted of the traditional vaccines: BCG, OPV, DPT, Measles and Yellow Fever Vaccines. Commencing in May 2012, the country introduced the Pentavalent Vaccine into its National Immunization Schedule in a phased manner over a 3-year period; in order to reduce substantially child mortality from pneumonia and meningitis and thus save up to 30,000 lives annually following full introduction. Currently, it is available in all PHC facilities nationwide. |
| However, pneumonia is a disease that can be caused by a number of other infectious agents that include bacteria, viruses and fungi; the most common in children being Streptococcus pneumoniae (pneumococcus). The Haemophilus influenzae type b (Hib) – is the second most common cause of bacterial pneumonia. Other causes are the Respiratory Syncytial Virus (RSV) and in infants infected with HIV, Pneumocystis jiroveci is one of the commonest causes of pneumonia. |
| Pneumococcal conjugate vaccine-10 (PCV-10) introduction: Most recently, the country also introduced the PCV-10 to protect infants against the diseases caused by the pneumococcal bacteria (Streptococcus pneumoniae) such as pneumonia, meningitis and bacteraemia. The national launch event was conducted on the 22nd December 2014 at Lokoja, Kogi State. The 12 States of the federation that commenced this phased introduction were selected on the basis of zonal spread, their immunization coverage and capacities to receive and store the new vaccine, viz: Adamawa, Yobe, Kaduna, Katsina, Kogi, Plateau, Ondo, Osun, Edo, Rivers, Anambra and Ebonyi. This introduction of the PCV- 10 is continuing in a phased manner so that by 2017, it will be available to infants in all the States in the country. |
| Supplemental immunization vaccines: Other supplemental vaccines such as Measles, Yellow Fever and the newer MenAfriVac™ vaccine that protects against the commonest cause of cerebro-spinal meningitis (CSM serotype A) are also given as needed. In the period 2011-2014, the MenAfriVac™ vaccine was administered to an extended age-group of 1-29 years old through the 23 States that fall within the meningitis belt in Nigeria, in our effort to eliminate CSM serotype A as a major public health issue. |
| The NDHS 2003, 2008 and 2013 have indicated a gradual, sustained improvement in child survival indices (Figure 2) [6]. The introduction of these new vaccines will further reduce childhood mortality rates in Nigeria. |
| IPV introduction as end game strategy for the PEI: The country has made an unprecedented progress in its polio eradication strategy (PEI) in the past 1 year. There have been only 6 confirmed cases of wild poliovirus type 1 (WPV1) in 2 States in 2014 compared to 50 cases in 9 States for the same period in 2013; with the date of onset of latest WPV case being 24th July 2014. There have been no confirmed WPV3 since November 2012 [7]. |
| The import of the above is that Nigeria is moving towards the interruption of the transmission of the wild poliovirus (WPV); achievable in the later part of 2015. Through the combined efforts of governments and other key stakeholders, it is hoped that this positive outcome may continue into the next couple of years for which the country may be certified as polio-free. |
| Hitherto, only variants of the oral polio vaccine (OPV) have been used in our RI and the PEI efforts. However, as the country enters into the polio endgame, it has now introduced the injectable polio vaccine (IPV) to complement the supplemental effort. Initially, the IPV was introduced in the polio-endemic States of Adamawa, Borno, Yobe, Bauchi and Kano. But, commencing from the first quarter of 2015, the IPV will also be available to be delivered in the RI schedule to all eligible children at age of 14 weeks, in addition to the OPV that is given in the traditional four-dose RI schedule (Table 1). The IPV given at that age is intercalated with the OPV and is expected to boost the child’s immunity further to confer higher protection against the poliovirus. |
| The rotavirus vaccine |
| The rotavirus vaccine is an oral vaccine against rotavirus infection, a common cause of diarrhea and sickness, commonly affecting young children. Diarrhea is a leading killer of children across Africa, causing approximately 12% of deaths in children under five years of age in the World Health Organization’s African Region (WHO AFR) [8]. Rotavirus, the most common cause of severe diarrhea in young children world-wide, causes more than 450,000 deaths each year in children under five and is responsible for millions of hospitalizations and clinic visits [9]. The incidence and severity of the rotavirus infection have declined significantly as seen in those countries that have added Rotavirus vaccination to their routine childhood immunization. When introduced into our RI schedule in the 3rd quarter of 2015, it will be administered to infants on 2 visits at ages of 6 and 10 weeks (Table 1). |
| Other health strengthening efforts |
| Apart from the Rota Virus vaccine that is administered orally, all the other newly introduced vaccines are in form of injection. This necessitates for the health service provider to have good working knowledge of injection safety and apply the appropriate injection techniques during vaccine administration. The programme ensures the use of auto-destruct (AD) syringes, universal safety boxes and proper waste disposal, including incineration of the injection wastes. |
| Through other channels and related programmes, families and communities are regularly educated on the benefits of other nonvaccine but highly effective measures such as exclusive breastfeeding for the newborn up till 6 months of age, adequate nutrition, reducing indoor air pollution and hand washing. Zinc supplements are given to reduce morbidity from diarrhoeal diseases while health-worker training in PHCs nationwide include capacity building for early detection and to manage and/or treat minor ailments. |
| The midwives service scheme (MSS) and SURE-P maternal and child health (MCH) |
| The MSS and SURE-P MCH both operate in 1,000 health facilities with a compliment of 4 midwives and 2 CHEWs per facility; this is a human resource pool of about 8,000 midwives and 2,000 CHEWs at the frontlines. We have put in place mechanisms for supply of drugs and commodities and in some places provided boreholes to complement the health services. These schemes serve a population of over 25 million Nigerians and have not only provided a platform for immunization services in the country but also a base for improving the health system of the country with special emphasis on PHC [10]. |
| The MSS and the SURE-P MCH projects which are under the NPHCDA feed into the Save One Million Lives (SOML) Initiative, which is coordinated by the Federal Ministry of Health. A combination of expanded access to maternal health care with the introduction of the new vaccines will surpass the anticipated reduction in disease burden, improve quality of motherhood and childhood, and in general improve the quality of life of the average Nigerian. |
| Conclusion |
| As Nigeria gradually introduces the new vaccines into its immunization schedule, the vaccine coverage improves, resulting in better protection of the children thereby aiding survival. This will further accelerate the country towards the achievement of the MDG4. |
References
- NPHCDA Annual Programme Implementation Report, 2013
- Nigeria Monthly RI Administrative Data Performance Feedback Report for October 2014
- Serdobova I, Kieny MP (2006) Assembling a Global Vaccine Development Pipeline for Infectious Diseases in the Developing World. Am J Public Health 96: 1554-1559
- National Demographic & Health Survey, Nigeria, 2008
- NPHCDA Facilitator’s Manual for Bottleneck Analysis Methodology (1stedn) Nov. 2012.
- National Demographic & Health Survey, Nigeria, 2013
- Nigeria Weekly Polio Statistics Update, 26th December 2014
- Liu L, Johnson HL, Cousens S (2012) Global, regional and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. The Lancet 379: 2151-2161.
- Tate JE, Burton AH, Boschi-Pinto C (2012) 2008 estimate of worldwi de rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: a systematic review and meta-analysis. The Lancet Infectious Diseases 12: 136-141
- NPHCDA Midwives Service Scheme; Concept and Progress, August 2012.
Tables and Figures at a glance
| Table 1 |
Figures at a glance
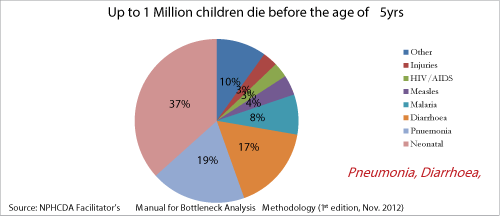 |
 |
| Figure 1 | Figure 2 |
Relevant Topics
- Acupuncture
- Acute Pain
- Analgesics
- Anesthesia
- Arthroscopy
- Chronic Back Pain
- Chronic Pain
- Hypnosis
- Low Back Pain
- Meditation
- Musculoskeletal pain
- Natural Pain Relievers
- Nociceptive Pain
- Opioid
- Orthopedics
- Pain and Mental Health
- Pain killer drugs
- Pain Mechanisms and Pathophysiology
- Pain Medication
- Pain Medicine
- Pain Relief and Traditional Medicine
- Pain Sensation
- Pain Tolerance
- Post-Operative Pain
- Reaction to Pain
Recommended Journals
Article Tools
Article Usage
- Total views: 16277
- [From(publication date):
May-2015 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 11603
- PDF downloads : 4674
