Research Article Open Access
Pregabalin Versus Acetaminophen for a Treatment of Chronic Neuropathic Pain on Extremities after Cervical Surgery: A Prospective Randomized, Open-Label Preliminary Study
Takashi Hirai*, Toshitaka Yoshii, Mitsuhiro Enomoto, Tsuyoshi Yamada, Takashi Taniyama, Hiroyuki Inose, Tsuyoshi Kato, and Atsushi Okawa
Department of Orthopedic Surgery, Tokyo Medical and Dental University, Japan
- *Corresponding Author:
- Takashi Hirai
Department of Orthopedic Surgery, Tokyo Medical and Dental University
1-5-45 Yushima, Bunkyo-Ku, Tokyo, 113-8519, Japan
Tel: +81358035279
Fax: +81358035281
E-mail: hirai.orth@tmd.ac.jp
Received date: October 6, 2016; Accepted date: April 19, 2016; Published date: October 21, 2016
Citation: Hirai T, Yoshii T, Enomoto M, Yamada T, Taniyama T, et al. (2016) Pregabalin Versus Acetaminophen for a Treatment of Chronic Neuropathic Pain on Extremities after Cervical Surgery: A Prospective Randomized, Open-Label Preliminary Study. J Pain Relief 5:273. doi:10.4172/2167-0846.1000273
Copyright: © 2016 Hirai T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Pain & Relief
Abstract
Therapeutic efficacy of pregabalin (PGL) for reducing neuropathic pain in patients who received a cervical operation is unclear. The purpose of this prospective pilot study was to compare the efficacy and adverse events between PGL and acetaminophen (ACM) for residual neuropathic pain. Thirty-four patients who received cervical spine surgery at our hospital from September 2011 to April 2013 and were diagnosed with residual neuropathic pain in the extremities were enrolled and were randomized to receive PGL (50 mg/day initially as an induction dose for 2 weeks) or ACM (1200 mg/day) for 8 weeks. PGL dosage was adjusted to 50-150 mg/day according to the severity of pain using a visual analog scale (VAS) after a 2-week time point. The Neck Disability Index (NDI) and VAS were used to evaluate subjective pain of the extremities and sleep condition. Short Form-36 (SF-36) and Japanese Orthopedic Association Cervical Myelopathy Evaluation Questionnaire (JOACMEQ) were used to evaluate neurologic function. Thirty-three patients (PGL 19 cases, ACM 14 cases) completed 8 weeks of follow-up. The average VAS for pain and numbness significantly improved in both groups at the final visit. The scale for sleep improved in the PGL group but was unchanged in the ACM group. The proportion of patients with satisfactory improvement in sleep was 31.6% and 0% in the PGL and ACM groups, respectively. There was a significant difference in VAS for sleep only. There were no significant improvements in the NDI and each domain of JOACMEQ in both groups. The number of patients who complained of somnolence tended to be greater in the PGL group. Thus, both PGL and ACM administration improved residual neuropathic pain in patients treated with cervical spine surgery for myelopathy. PGL was more effective in reducing sleep interference related to refractory neuropathic pain compared with ACM.
Keywords
Pregabalin; Acetaminophen; Neuropathic pain; Cervical spondylosis
Introduction
Neuropathic pain, caused by disease affecting the somatosensory nervous system, has a substantial impact on quality of life, and often leads to a high economic burden for individuals and society [1-3]. Notably, central neuropathic pain caused by spinal cord injury, multiple sclerosis, or stroke is often severe and chronic, and it affects many patients all over the world. The difficulty of treatment of such pain states has been recognized and a few recommendations have been proposed for the pharmacotherapy of chronic pain over the past 10 years [4,5].
Pregabalin (PGL), a 3-substituted analogue of gamma-amino butyric acid, has been widely used for pharmacological therapy in patients with peripheral neuropathic pain, and has been recognized as a primary therapeutic intervention for painful neuropathy for the last decade [6-8]. To date, various studies have demonstrated that PGL is also effective for central neuropathic pain such as post-stroke pain, and chronic pain following spinal cord injury and multiple sclerosis. In patients with cervical compressive myelopathy, the prevalence of neuropathic pain has been reported to be as high as 77% [9]. Thus, it is speculated that not only motor disturbance but also chronic pain caused by cervical myelopathy might impair quality of life. Although many patients who underwent cervical decompression surgery for myelopathy experience residual neuropathic pain [10], no study has investigated which drugs improve this pain state thus far. The objective of this study was to evaluate the safety and analgesic effect of PGL compared with acetaminophen (ACM) for residual neuropathic pain in patients who received a cervical operation and to investigate which functional domains improve after treatment.
Materials and Methods
The study protocol was approved by the hospital’s Research and Ethics Committee and was carried out in accordance with the Helsinki Declaration of 1975, as revised in 1983. All patients were duly informed about the study before their participation and signed the appropriate consent form.
The inclusion criteria were 1) surgery for cervical degeneration such as spondylosis, herniation, or ossification of the posterior longitudinal ligament; (2) diagnosis of neuropathic pain and/or numbness of the upper and/or lower extremities lasting for at least 3 months after the operation; (3) Neuropathic Pain Screening Questionnaire (pain DETECT) score ≥ 6 and visual analog scale (VAS) of neuropathic pain of the upper and/or lower extremities >40 mm; and (4) patient aged 20–75 years who can answer these questionnaires independently. The exclusion criteria were (1) diagnosis of cancer pain; (2) history of neuronal blockage using a neuro-destructive agent within 6 months; (3) pain on the upper and/or lower extremities that are caused by a reason other than a spinal disorder; (4) significant motor deficits and/or bowel or bladder dysfunction; and (5) history of another spinal operation.
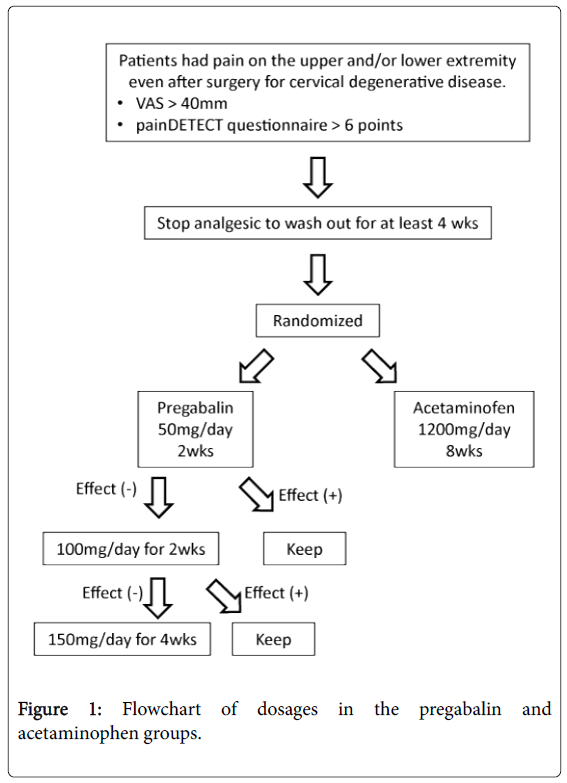
Among 168 patients who received cervical spine surgery at our hospital from September 2011 to April 2013, 34 consecutive patients (26 men, 8 women) who were newly diagnosed with chronic neuropathic pain following cervical spine surgery were enrolled (Figure 1). These patients were randomized into two groups using a random number table. In the pregabalin (PGL) group, patients started pregabalin (Lyrica®) at a dose of 50 mg/day as an induction dose for 2 weeks. Patients with visual analog scale (VAS) of less than 40 mm at the 2-week time point maintained the initial dose for 2 more weeks. If the dose did not produce sufficient pain relief, with a VAS of greater than or equal to 40 mm, it was increased to 100 mg/day for 2 weeks. Patients received the agent at a dose up to 150 mg/day during the follow-up period if VAS was greater than or equal to 40 at the 4-week time point. The total treatment period was 8 weeks. In the acetaminophen (ACM) group, patients received acetaminophen (Calonal®) 1200 mg/day for 8 weeks.
The Neck Disability Index (NDI) and VAS were used to evaluate the severity of subjective pain of the upper and/or lower extremities and condition of sleep. Subjective sleep quality was rated from 0 mm (‘Best night of sleep ever’) to 100 mm (‘worse night of sleep ever’). Short Form-36 (SF-36) and Japanese Orthopedic Association Cervical Myelopathy Evaluation Questionnaire (JOACMEQ) were used to evaluate the neurologic function of each patient before administration and at each visit.
Changes in VAS (ΔVAS), each item of JOACMEQ and NDI from baseline were calculated for each patient and the averages were compared between the two groups using the Mann-Whitney U-test. A χ2 test for categorical data was used for statistical analysis. All P values less than 0.05 were considered significant.
Results
Thirty-four patients were enrolled in this study. One patient dropped out of this study because of difficulty in ambulation. A total of 33 patients (PGL, 19 cases with a mean age of 67.1 years; ACM, 14 cases with a mean age of 66.4 years) completed 8 weeks of follow-up. Patient characteristics are shown in Table 1. Mean duration from the operation to treatment was 42.8 months for the PGL group and 21.8 months for the ACM group, indicating that residual pain could be recognized as a chronic state in these patients.
| PGL group (n=19) |
ACM group (n=14) |
||
|---|---|---|---|
| Age(y-o) | - | 67.1 ± 9.8 | 66.4 ± 10.7 |
| Male : Female | - | 14 : 5 | 11 : 3 |
| Diagnosis | CSM | 12 | 9 |
| OPLL | 7 | 4 | |
| CDH | - | 1 | |
| Surgical method | ADF | 4 | 2 |
| LAMP | 14 | 8 | |
| ADF + LAMP | 1 | 1 | |
| Mean duration of pain (Months) | - | 42.9 ± 27.4 | 21.8 ± 14.2 |
| Preoperative JOA score | 13.5 ± 2.3 | 13.7 ± 2.9 | |
| Maximum dose | 300 | 1200 | |
| VAS (mm) before and 8 weeks after the treatment | Extremities pain | 63.6 ± 23.6 / 47.8 ± 28.7 | 61.5 ± 18.6 / 36.3 ± 25.2 |
| Extremities numbness | 62.1 ± 23.5 / 38.9 ± 24.3 | 77.1 ± 18.9 / 51.0 ± 24.0 | |
| Sleep | 77.1 ± 23.7 / 59.8 ± 26.5 | 68.7 ± 32.6 / 75.4 ± 26.7 | |
| JOACMEQ | Cervical spine | 57.1 ± 30.0 / 56.2 ± 30.9 | 64.5 ± 30.9 / 60.6 ± 39.9 |
| Upper extremity motor | 71.7 ± 25.0 / 73.3 ± 22.8 | 82.8 ± 17.1 / 85.9 ± 16.6 | |
| Lower extremity motor | 55.6 ± 33.2 / 55.8 ± 33.1 | 75.2 ± 21.9 / 70.6 ± 10.8 | |
| Bladder | 70.8 ± 26.2 / 74.0 ± 24.7 | 79.6 ± 17.6 / 85 ± 13.6 | |
| Quality of life | 47.7 ± 24.0 / 50.1 ± 22.9 | 55.5 ± 13.1 / 56.8 ± 10.9 |
Values represent mean ± standard deviation. PGL, pregabalin; ACM, acetaminophen; CSM, cervical spondylotic myelopathy; OPLL, ossification of the posterior longitudinal ligament; CDH, cervical disc herniation; ADF, anterior decompression with fusion; LAMP, laminoplasty; JOA, Japanese Orthopedic Association; VAS visual analog scale; JOACMEQ, Japanese Orthopedic Association Cervical Myelopathy Evaluation Questionnaire
Table 1: Characteristics of the PGL and ACM groups
None of the patients underwent additional spinal surgery during the follow-up period. The mean final dose was 126 mg/day (range, 50-300 mg/day) in the PGL group. All patients in the ACM group took 1200 mg/day. Before the treatment, the average VAS for pain was 63.6 mm in the PGL group and 61.5 mm in the ACM group. The VAS for numbness and sleep were 62.1 mm and 77.1 mm for the PGL group, and 77.1 mm and 68.7 mm for the ACM group, respectively. In terms of each function in the JOACMEQ, there were no significant differences between the two groups (Table 1).
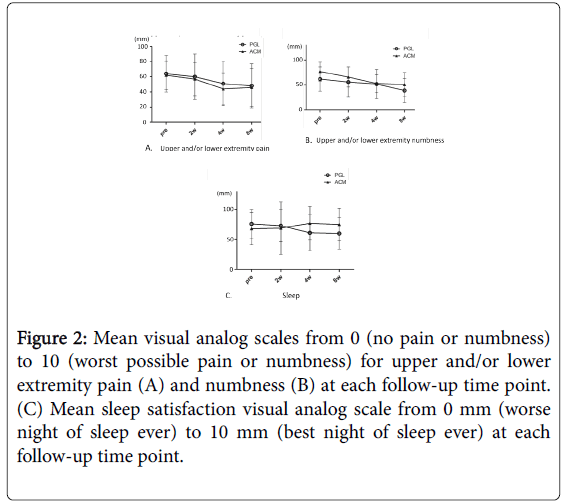
The scales for pain and numbness in the extremities gradually decreased and significantly improved in both groups 8 weeks after treatment compared with baseline (Figure 2A and 2B). Although the scale for sleep improved in the PGL group, it was unchanged even after treatment in the ACM group (Figure 2C). There were no significant differences in changes of the three items between the two groups. In addition, ΔVAS of ≥ 30 mm was defined as satisfactory improvement. The proportion of patients with satisfactory improvement in the PGL and ACM groups were 31.6% and 27.2% for pain, 31.6% and 18.2% for numbness, and 31.6% and 0% for sleep, respectively. There was a significant difference only in the VAS for sleep (Table 2).
Figure 2: Mean visual analog scales from 0 (no pain or numbness) to 10 (worst possible pain or numbness) for upper and/or lower extremity pain (A) and numbness (B) at each follow-up time point. (C) Mean sleep satisfaction visual analog scale from 0 mm (worse night of sleep ever) to 10 mm (best night of sleep ever) at each follow-up time point.
| PGL group | ACM group | P | |
|---|---|---|---|
| Proportion of patients with ΔVAS of ? 40mm | - | - | - |
| Extremities pain | 31.60% | 21.40% | 0.42 |
| Extremities numbness | 31.60% | 14.30% | 0.2 |
| Sleep | 31.6%* | 0% | 0.027 |
PGL, pregabalin; ACM, acetaminophen; VAS, visual analog scale
Table 2: Proportion of patients who had satisfactory improvement in the PGL and ACM groups.
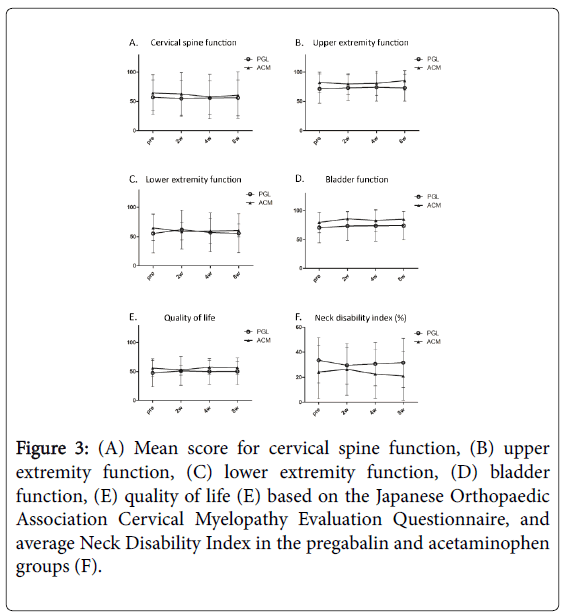
JOACMEQ domains scores before and after treatment are presented in Figure 3A-3E. Each of the five domains was analyzed separately. No significant improvement in each domain was observed in the two groups. Similarly, the NDI gradually improved after the treatments in both groups, but the improvement was not significant compared with baseline (Figure 3F).
Figure 3: (A) Mean score for cervical spine function, (B) upper extremity function, (C) lower extremity function, (D) bladder function, (E) quality of life (E) based on the Japanese Orthopaedic Association Cervical Myelopathy Evaluation Questionnaire, and average Neck Disability Index in the pregabalin and acetaminophen groups (F).
Pregabalin was generally well tolerated with few reported side effects. The most frequently reported adverse events were central nervous system-related somnolence and nausea. Although the incidence of these adverse events showed no significant differences between the PGL and ACM groups (Table 3), the number of patients who complained of somnolence tended to be greater in the PGL group.
| PGL group | ACM group | P | |
|---|---|---|---|
| (n=19) | (n=14) | ||
| Somnolence | 3 | 0 | 0.11 |
| Dizziness | 2 | 1 | 0.74 |
| Nausea | 1 | 0 | 0.38 |
| Peripheral edema | 1 | 0 | 0.38 |
| Weight gain | 1 | 0 | 0.38 |
PGL, pregabalin; ACM, acetaminophen
Table 3: Summary of common treatment-emergent adverse events
Discussion
The results of the present study can be summarized as follows: (1) both pregabalin and ACM administration improved residual neuropathic pain in patients treated with cervical spine surgery for myelopathy; (2) pregabalin was effective for reducing sleep interference related to refractory neuropathic pain compared with ACM; (3) pregabalin sometimes brought about adverse effects such as somnolence, dizziness, and peripheral edema. These results are consistent with the evidence that pregabalin is an effective treatment for chronic neuropathic pain caused by a disease of the central nervous system including post-stroke, spinal cord injury, and multiple sclerosis. However, residual neuropathic pain in patients treated with cervical decompression surgery, on which the present study focused, could be considered as a pain state that is more complicated than diseases investigated by other studies. The pathology consists of two distinct mechanisms: one is neuronal damage in the central nervous system caused by compression, and the other is residual abnormality in the peripheral nervous system. In patients who had suffered from neuropathic pain caused by myelopathy for a long time, these two pain states might be irreversible even after cervical decompression operation and often distress patients.
The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) was introduced in 2000 to assess the quality of evidence and gained widespread international acceptance. To date, various randomized trials for treatment of refractory neuropathic pain have been performed and several drugs have been identified as effective medication based mainly on the GRADE recommendations. Tricyclic antidepressants, serotonin-noradrenaline reuptake inhibitor antidepressants, pregabalin, gabapentin, and gabapentin extended release or enacarbil have strong GRADE recommendations for use in neuropathic pain and are proposed as first-line treatments. Notably, earlier studies of gabapentinoid [11] as a treatment for chronic neuropathic pain had demonstrated that pregabalin and gabapentin decreased mean daily pain intensity with an acceptable safety profile, and had calculated the number needed to treat (NNT) for 50% pain intensity reduction. A meta-analysis concluded that the overall NNT was 7.7 (95% CI, 6.5–9.4) for pregabalin and 6.3 (95% CI, 5.0–8.3) for gabapentin, and these drugs provide good outcomes not only for peripheral neuropathic pain but also central neuropathic pain.
This study revealed that not only PGL but also ACM improved residual refractory pain in patients treated with cervical decompression surgery. Although it has been reported that ACM does not affect neuropathic pain mechanisms [12], there are reports of positive effects of ACM for neuropathic pain. ACM attenuates hypersensitivities caused by neuropathic pain such as chemotherapyinduced pain [13] and partial sciatic nerve ligation models [14]. Several preclinical studies suggested the mechanisms of analgesic action of ACM. One is an increase of serotonin levels released from the brainstem serotonergic neurons in the central nervous system. Another is action either directly as an opioid or cannabinoid receptor ligand or indirectly by increasing the endogenous opioid or cannabinoid receptor ligand [15,16]. These pathways probably explain the reason why systemic administration of ACM was effective for patients with residual neuropathic pain in the present study.
The beneficial effects of pregabalin on sleep and mood disturbances associated with central neuropathic pain have also been shown [17,18]. Our study revealed that the reduction in sleep interference was observed at 2 weeks after initiation of pregabalin and maintained for the subsequent 8 weeks. It is unlikely that the observed improvement in disturbed sleep with pregabalin could be attributed to the adverse event of somnolence. Somnolence often occurred immediately after initiation of the treatment and appeared to be transient. Pregabalin has been shown to be effective in increasing the duration of non-rapid eye movement sleep in rats [19]. In addition to this evidence, pregabalin has also been demonstrated to increase slow-wave sleep during a polysomnography study of healthy volunteers [20,21]. These findings suggest that pregabalin may have a direct effect on sleep architecture in patients with neuropathic pain.
This open-label design of the present study has some limitations. This study did not have a placebo-treated control group. Additionally, patients and physicians were not blinded to treatment. Because of these limitations, the efficacy of the two treatments may be influenced by patients’ and physicians’ expectations. With regard to the drug dosing of pregabalin, the flexible-dose regimen of pregabalin was relatively lower than usual. However, some patients obtained satisfactory pain relief even on 50–100 mg/day. In clinical practice, flexible dosing of a drug optimizes the balance of efficacy and tolerability. We believe that adequate pain relief was achieved with lower doses of the drug together with a reduced incidence of adverse events.
In summary, not only pregabalin but also ACM produced clinically significant reductions in central neuropathic pain. Pregabalin tended to enable satisfactory sleep compared with ACM in patients with refractory pain after cervical spine surgery.
Conclusion
Both PGL and ACM administration improved residual neuropathic pain in patients treated with cervical spine surgery for myelopathy. PGL was more effective in reducing sleep interference related to refractory neuropathic pain compared with ACM.
References
- Widerstrom-Noga EG, Felipe-Cuervo E, Yezierski RP (2001) Chronic pain after spinal injury: interference with sleep and daily activities. Arch Phys Med Rehabil 82: 1571-1577.
- Saverino A, Solaro C (2012) Pain in individuals with multiple sclerosis, knee prosthesis, and post-herpetic neuralgia: learning from focus group patients' experience. Clin J Pain 28: 300-308.
- Attal N, Minet ML, Laurent B, Fermanian J, Bouhassira D (2011) The specific disease burden of neuropathic pain: results of a French nationwide survey. Pain 152: 2836-2843.
- Attal N, Cruccu G, Baron R, Haanpaa M, Hansson P, et al. (2010) Societies EFoN. EFNS guidelines on the pharmacological treatment of neuropathic pain: 2010 revision. Eur J Neurol 17: 1113-e1188
- Moulin DE, Clark AJ, Gilron I, Ware MA, Watson CP, et al. (2007) Pharmacological management of chronic neuropathic pain - consensus statement and guidelines from the Canadian Pain Society. Pain Res Manag 12: 13-21.
- Seventer RV, Feister HA, Young JP, Stoker M, Versavel M, et al. (2006) Efficacy and tolerability of twice-daily pregabalin for treating pain and related sleep interference in postherpetic neuralgia: a 13-week, randomized trial. Curr Med Res Opin 22: 375-384.
- Seventer RV, Bach FW, Toth CC, Serpell M, Temple J, et al. (2010) Pregabalin in the treatment of post-traumatic peripheral neuropathic pain: a randomized double-blind trial. Eur J Neurol 17: 1082-1089.
- Lesser H, Sharma U, LaMoreaux L, Poole RM (2004) Pregabalin relieves symptoms of painful diabetic neuropathy: a randomized controlled trial. Neurology 63: 2104-2110.
- Yamashita T, Takahashi K, Yonenobu K, Kikuchi S (2014) Prevalence of neuropathic pain in cases with chronic pain related to spinal disorders. J Orthop Sci 19: 15-21.
- Higuchi D (2015) Prognostic Value of Preoperative Coping Strategies for Pain in Patients with Residual Neuropathic Pain after Laminoplasty for Compressive Cervical Myelopathy. Asian Spine J 9: 675-682.
- Finnerup NB, Attal N, Haroutounian S, McNicol E, Baron R, et al. (2015) Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol 14: 162-173.
- Morlion B (2011) Pharmacotherapy of low back pain: targeting nociceptive and neuropathic pain components. Curr Med Res Opin 27: 11-33.
- Lynch JJ 3rd, Wade CL, Zhong CM, Mikusa JP, Honore P (2004) Attenuation of mechanical allodynia by clinically utilized drugs in a rat chemotherapy-induced neuropathic pain model. Pain 110: 56-63.
- Dani M, Guindon J, Lambert C, Beaulieu P (2007) The local antinociceptive effects of paracetamol in neuropathic pain are mediated by cannabinoid receptors. Eur J Pharmacol 573: 214-215.
- Bertolini A, Ferrari A, Ottani A, Guerzoni S, Tacchi R, et al. (2006) Paracetamol: new vistas of an old drug. CNS Drug Rev 12: 250-275.
- Mallet C, Daulhac L, Bonnefont J, Ledent C, Etienne M, et al. (2008) Endocannabinoid and serotonergic systems are needed for acetaminophen-induced analgesia. Pain 139: 190-200.
- Sabatowski R, Galvez R, Cherry DA, Jacquot F, Vincent E, et al. (2004) Pregabalin reduces pain and improves sleep and mood disturbances in patients with post-herpetic neuralgia: results of a randomised, placebo-controlled clinical trial. Pain 109: 26-35.
- Siddall PJ, Cousins MJ, Otte A, Griesing T, Chambers R, et al. (2006) Pregabalin in central neuropathic pain associated with spinal cord injury: a placebo-controlled trial. Neurology 67: 1792-1800.
- Wang TX, Yin D, Guo W, Liu YY, Li YD, et al. (2015) Antinociceptive and hypnotic activities of pregabalin in a neuropathic pain-like model in mice. Pharmacol Biochem Behav 135: 31-39.
- Roth T, Lankford DA, Bhadra P, Whalen E, Resnick EM (2012) Effect of pregabalin on sleep in patients with fibromyalgia and sleep maintenance disturbance: a randomized, placebo-controlled, 2-way crossover polysomnography study. Arthritis Care Res (Hoboken) 64: 597-606.
- de Haas S, Otte A, de Weerd A, van Erp G, Cohen A, et al. (2007) Exploratory polysomnographic evaluation of pregabalin on sleep disturbance in patients with epilepsy. J Clin Sleep Med 3: 473-478.
Relevant Topics
- Acupuncture
- Acute Pain
- Analgesics
- Anesthesia
- Arthroscopy
- Chronic Back Pain
- Chronic Pain
- Hypnosis
- Low Back Pain
- Meditation
- Musculoskeletal pain
- Natural Pain Relievers
- Nociceptive Pain
- Opioid
- Orthopedics
- Pain and Mental Health
- Pain killer drugs
- Pain Mechanisms and Pathophysiology
- Pain Medication
- Pain Medicine
- Pain Relief and Traditional Medicine
- Pain Sensation
- Pain Tolerance
- Post-Operative Pain
- Reaction to Pain
Recommended Journals
Article Tools
Article Usage
- Total views: 13378
- [From(publication date):
November-2016 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 12426
- PDF downloads : 952