Research Article Open Access
Purple Rice Extract Exhibits Preventive Activities on Experimental Diabetes Models and Human Subjects
| Hiroshi Shimoda*, Michio Aitani, Junji Tanaka and Shoketsu Hitoe | ||
| Oryza Oil & Fat Chemical Co. Ltd., 1 Numata, Kitagata-cho, Ichinomiya, Aichi 493-8001, Japan | ||
| Corresponding Author : |
Hiroshi Shimoda E-mail: kaihatsu@mri.biglobe.ne.jp |
|
| Received January 06, 2015; Accepted January 27, 2015; Published February 01, 2015 | ||
| Citation: Shimoda H, Aitani M, Tanaka J, Hitoe S (2015) Purple Rice Extract Exhibits Preventive Activities on Experimental Diabetes Models and Human Subjects. J Rice Res 3:137. doi: 10.4172/2375-4338.1000137 | ||
| Copyright: © 2015 Shimoda H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | ||
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
||
Visit for more related articles at Rice Research: Open Access
Abstract
Purple rice is colored rice containing anthocyanins and has mainly been consumed in Southeast Asian countries. Consumption of a large amount of rice would cause undesirable effects in patients with diabetes or high blood glucose. However, if the colored ingredients have antidiabetic effects, purple rice consumption may be an effective alternative dietary approach to prevent diabetes, instead of white rice. Thus, we evaluated the effect of Purple Rice Extract (PRE) on various diabetes models. PRE inhibited α-glucosidase, α-amylase and aldose reductase activities with IC50 values of 409, 135 and 26 μg/mL, respectively. In a diabetic retinopathy model using retinal microvascular endothelial cells, PRE (30 μg/mL) significantly suppressed cell damage induced by a high concentration of glucose. On the other hand, PRE significantly delayed starch and sucrose absorption at 3 and 1 g/kg, respectively, in rats. Moreover, single oral dosing of PRE (25 mg) to human subjects significantly suppressed postprandial blood sugar elevation after the ingestion of rice (200 g). These results suggest that PRE is effective for the control of blood sugar to prevent the progression of diabetic symptoms.
| Keywords | |
| Purple rice; α-Glucosidase; Aldose reductase; Retinal microvascular endothelial cell; Diabetes; Anti-diabetic activity of purple rice extract | |
| Introduction | |
| Purple rice is a colored variety of rice (Oryza sativa L.) that is cultivated widely in Southeast Asia, including China, Indonesia and Thailand. It contains purple black pigments (anthocyanins) and is used as a food colorant in bread, ice cream and liquor. We have investigated the composition of purple rice anthocyanins and found that they consist of cyanidin 3-O-glucoside, peonidin 3-O-glucoside and cyanidin 3-O-gentiobioside [1]. On the other hand, as biological activities of purple rice extract (PRE), we found that it and its constituents prevented various eye diseases in model mice and cultured cells. For example, PRE suppressed strong visible light-induced retinal damage in mice and retinal cells [2]. This inhibitory effect was dependent on the strong anti-oxidative activities of the anthocyanins. On the other hand, PRE suppressed angiogenesis leading to diabetic retinopathy. PRE and the anthocyanidins strongly suppressed the proliferation, migration and tube formation of human retinal microvascular endothelial cells induced by vascular endothelial growth factor [3]. Moreover, PRE suppressed endoplasmic retinal damage in vitro and in vivo [4]. Therefore, PRE has been shown to protect the eyes from strong environmental changes or eye diseases. | |
| Diabetes mellitus involves chronic hyperglycemia due to insulin insufficiency. It is considered to be caused by the combination of some genetic factors and lifestyle, especially excessive carbohydrate consumption. With the increasing development of the economy in certain countries, the lifestyle of the population is changing. For example, the consumption of dietary sugar and fat has been increasing in Southeast Asia. Thus, diabetes has emerged as a serious disease in this region. Specifically, the number of patients with diabetes has been increasing in Indonesia [5], Thailand [6] and China [7]. Against this background, we have already reported that PRE suppressed the progression of retinopathy mostly caused by severe diabetes in mouse and cell models [3]. However, the action of PRE against carbohydrate absorption and metabolism has not been clarified yet from the perspective of preventive care. If PRE suppresses carbohydrate absorption, this could facilitate the control of blood sugar levels through daily lifestyle choices. Therefore, we conducted basic and clinical studies of PRE to investigate its efficacy for preventing diabetes | |
| Materials and Methods | |
| Preparation of PRE | |
| PRE was prepared by a previously described method [3]. Briefly, purple rice bran obtained from China (100 g) was defatted with n-hexane (500 mL) to remove oily constituents. Then, 30% aqueous ethanol (500 mL) was added and extracted for 2 h at 30°C. The solvent was evaporated to obtain PRE (15.2 g). Total anthocyanin and polyphenol contents were 10 and 30%, respectively. | |
| Animals and cells | |
| Male rats (Wistar, 5 weeks old) were purchased from Clea Japan (Tokyo, Japan) and preliminarily maintained for 7 days by feeding on a conventional diet (MF, Oriental Yeast Co. Ltd., Tokyo, Japan) and tap water. The experiments were performed in accordance with the Guidelines for the Proper Conduct of Animal Experiments (Special Council of Japan, June 1, 2006). Human Retinal Microvascular Endothelial Cells (HRMECs) were purchased from DS Pharma Biomedical (Tokyo, Japan) and maintained in CS-C medium containing 10% fetal bovine serum, 50 μg/mL gentamicin, 50 ng/mL amphotericin B and culture boost (growth factors) at 37°C under a 5% CO2 atmosphere. | |
| Reagents | |
| Intestinal acetone powder from rats and porcine pancreatic amylase (Sigma-Aldrich, St. Louis,MO)WST-8[2-(2-methoxy-4-nitrophenyl)- 3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H tetrazolium monosodium salt] and 4-methylumbelliferyl-α-D-glucopyranoside (MUG) were obtained from Dojindo (Kumamoto, Japan). Glycine, Amylase Test Wako, soluble starch, nicotinamide adenine dinucleotide phosphate reduced form (NADPH), DL-glyceraldehyde, aldose reductase and Glucose B-Test Wako were purchased from Wako Pure Chemical Co. Ltd. (Tokyo, Japan). | |
| Evaluation of carbohydrate-digestive and -metabolizing enzyme activities | |
| For the evaluation of α-glucosidase activity, rat intestinal acetone powder was dissolved in 0.1 M citrate/phosphate buffer (pH: 6.5) to 10 mg/mL. The solution was used as a crude enzyme solution containing α-glucosidase. α-Glucosidase activity was determined by fluorescent assay using MUG as a substrate. Various concentrations of PRE (100 μL) were mixed with 0.33 mM MUG (300 μL) and the buffer (500 μL) and incubated at 37°C. Then, crude α-glucosidase solution (100 μL) was added to the mixture and incubated for 30 min. The reaction was stopped by the addition of 0.5 M glycine (3 mL). The absorbance was measured at 366 nm (reference wavelength: 450 nm). To evaluate α-amylase inhibitory activity, Amylase Test Wako was used. The substrate solution in the kit (1 mL) was mixed with 100 μL of PRE solution dissolved in 0.25 M phosphate buffer (pH: 7.0, 50 mM) and preliminarily incubated (37°C) for 5 min. Then, 100 μL of pancreatic amylase solution (50 units/mL) was added. The mixture was incubated for 30 min and the reaction was stopped by heating the test tubes in boiling water for 5 min. Then, reacted solution was mixed with 1 mL of the color developing solution attached to the kit and the absorbance at 620 nm was measured. For the evaluation of aldose reductase inhibitory activity, 0.18 M phosphate buffer (pH 7.0, 500 μL) was mixed with 1.5 mM NADPH (100 μL), 100 mM DL-glyceraldehyde (100 μL), water (295 μL) and PRE dissolved in dimethyl sulfoxide (10 μL). After preliminary incubation (30°C) of the mixture for 5 min, 1 unit/mL aldose reductase solution (5 μL) was added and further incubated at 30 min. The reaction was stopped by ice-chilling. The absorbance of the mixture was measured at 340 nm. The IC50 values were calculated by the probit method. | |
| High-glucose-induced retinal cell damage | |
| HRMECs (5 × 103 cells/well) in a 96-well culture plate were preliminarily cultured for 24 h. PRE and 50 mM glucose were added to the wells and cultured for 24 h. Then, WST-8 was added and cultured for 3 h to evaluate the cytotoxicity. The absorbance at 492 nm (reference wavelength: 660 nm) was measured. | |
| Oral carbohydrate loading test in rats | |
| After 24 h fasting of rats, soluble starch (2 g/kg) or sucrose (2 g/kg) and PRE (1 or 3 g/kg) were given orally to the rats. Every 30 minutes, blood samples were collected from the tail vein using heparinized capillary tubes. These tubes were centrifuged to obtain the serum. Serum glucose level was measured using Glucose B-Test Wako. | |
| Oral carbohydrate loading test in humans | |
| This experiment was performed in accordance with the 6th revision of the Declaration of Helsinki of 2008. Five healthy males and 3 healthy females aged 23 to 48 years old were used as subjects. After fasting for 11 hours, these subjects ate a rice ball (200 g) with a placebo capsule containing 50 mg of dextrin. Blood was collected from their fingertips at 0, 30, 60, 90 and 120 min thereafter and blood glucose was measured using a portable analyzer equipped with a glucose sensor (Terumo Co., Tokyo, Japan). A week later, the same subjects underwent the same procedure by taking a capsule containing 50 mg of purple rice extract-P (Oryza Oil & Fat Chemical Co. Ltd.) consisting of PRE (25 mg) and dextrin (25 mg). | |
| Statistical analysis | |
| Data are presented as mean ± SE. For statistical comparisons in the cell experiment, one-way Analysis Of Variance (ANOVA) followed by Dunnett’s test was performed. Student’s t-test was used for animal and human studies. A value of p<0.05 was considered to indicate statistical significance. | |
| Results | |
| Effect of PRE on carbohydrate-digestive and -metabolizing enzyme activities | |
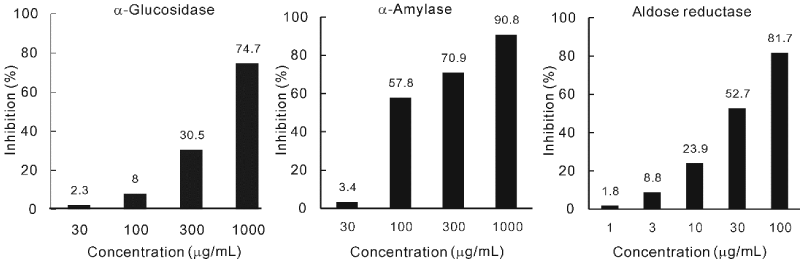
| Figure 1 shows the inhibitory effects of PRE on α-glucosidase, α-amylase and aldose reductase activities. PRE suppressed α-glucosidase activity at 300 and 1,000 μg/mL. The IC50 was 409 μg/mL. PRE also suppressed α-amylase activity at more than 100 μg/mL with an IC50 of 135 μg/mL. PRE strongly suppressed aldose reductase activity with an IC50 of 26 μg/mL. | |
| Effect of PRE on high-glucose-induced retinal cell damage | |
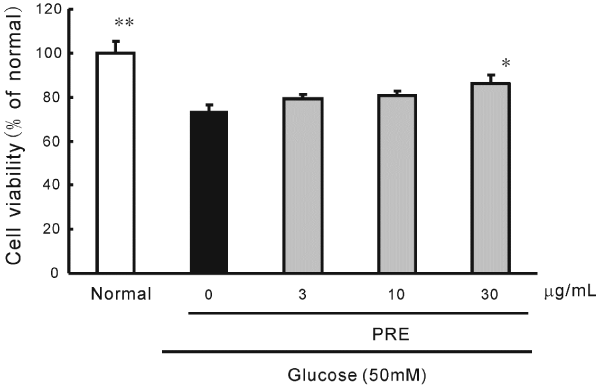
| A high concentration of glucose (50 mM) caused cell damage to HRMECs. The survival rate dropped approximately 20%. However, PRE (30 μg/mL) significantly ameliorated this cell damage (Figure 2). | |
| Effect of PRE on blood glucose changes in carbohydrateloaded rats | |
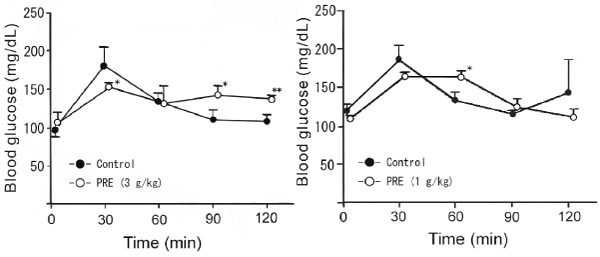
| We investigated the effects of PRE on blood glucose changes after oral loading of starch and sucrose. PRE (3 g/kg) significantly suppressed the maximum increase in blood glucose and delayed starch absorption at 90 and 120 min after its loading. On the other hand, PRE (1 g/kg) significantly delayed glucose absorption at 60 min after sucrose loading (Figure 3). | |
| Effect of PRE on blood glucose changes in human subjects after ingestion of carbohydrate | |
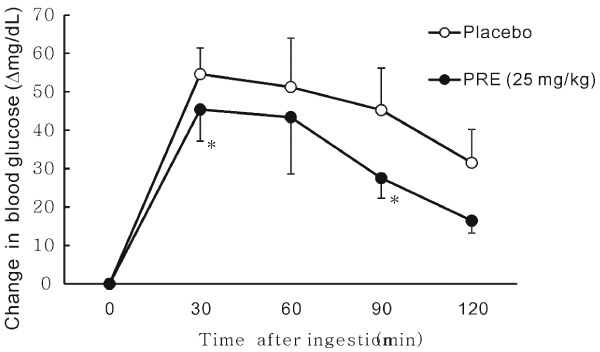
| In the rice loading tests in 5 healthy males and 3 healthy females, significant suppression of blood glucose was observed at 30 and 90 min after the ingestion of rice in the PRE (25 mg) group compared with that in the control group. PRE suppressed the total glucose absorption until 120 minutes after rice ingestion. Thus, PRE was found to suppress the acute increase in blood glucose after meals (Figure 4). | |
| Discussion | |
| In this study, we mainly investigated the potential effects of PRE on carbohydrate absorption and diabetic complications in ocular cells. In terms of the obtained results, PRE delayed dextrin and sucrose absorption in rats based on the inhibition of carbohydrate digestive enzymes including α-glucosidase and α-amylase. In terms of the efficacy of purple rice on diabetes, only Apichai et al. [8] have reported that a diet containing 5% purple rice bran improved the diabetic conditions in streptozotocin-induced diabetic rats by 8-week ingestion. In this previous study, the diet decreased fasting blood glucose and triglyceride, and enhanced glucose transporter 4 (Glut4) level in the soleus muscle. In our experiments, although PRE required a high dosage (1 to 3 g/kg) to suppress carbohydrate absorption, a low dose of PRE may improve chronic high blood glucose levels by relatively long-term ingestion. Regarding the anti-diabetic ingredients in PRE, especially anthocyanins, there are a considerable number of reports about purple corn. This corn is cultivated in South America and contains anthocyanins. Purple corn extract has been reported to suppress diabetic glomerular angiogenesis [9], glomerular monocyte activation and macrophage infiltration [10]. On the other hand, cyanidin 3-O-glucoside, a major anthocyanin of purple rice [1] and purple corn [11], was reported to exhibit an antidiabetic effect in type 2 diabetes mice, including upregulation of Glut4 [12]. Moreover, cyanidin 3-O-glucoside has been reported to inhibit sucrose and α-amylase activities with IC50 values of 0.97 and 0.3 mM. Therefore, PRE was found to suppress carbohydrate absorption based on the inhibition of carbohydrate digestive enzymes by anthocyanins such as cyanidin 3-O-glucoside [13]. | |
| PRE strongly suppressed the activity of aldose reductase, which converts glucose to sorbitol in the first step of the polyol pathway. The polyol metabolites accumulate in peripheral tissues and cause severe diabetic retinopathy [14]. In the high-glucose-induced cell toxicity study in HRMECs, PRE significantly suppressed the damage. Hence, the inhibitory activities of PRE against aldose reductase and high-glucose-induced damage in retinal cells may prevent diabetic complications in severe diabetes. | |
| In a small-scale human study, a relatively low dose of PRE (25 mg) effectively suppressed postprandial increase in blood glucose. Regarding the clinical study of anthocyanin for anti-diabetic activity, only a strawberry beverage consisting of pelargonidin 3-O-glucoside has been reported to improve postprandial insulin and inflammation responses [15]. However, this strawberry beverage did not change postprandial blood glucose. Therefore, our finding that PRE suppressed postprandial glucose is the first observation of its kind. | |
| Conclusion | |
| In conclusion, purple rice extract rich in anthocyanins was found to suppress carbohydrate absorption by inhibiting carbohydrate digestive enzymes. Moreover, it suppressed aldose reductase activity and high-glucose-related retinal damage for the prevention of diabetic ocular diseases. | |
References |
|
|
|
Figures at a glance
 |
 |
 |
 |
|||
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
Relevant Topics
- Basmati Rice
- Drought Tolerence
- Golden Rice
- Leaf Diseases
- Long Grain Rice
- Par Boiled Rice
- Raw Rice
- Rice
- Rice and Aquaculture
- Rice and Nutrition
- Rice Blast
- Rice Bran
- Rice Diseases
- Rice Economics
- Rice Genome
- Rice husk
- Rice production
- Rice research
- Rice Yield
- Sticky Rice
- Stress Resistant Rice
- Unpolished Rice
- White Rice
Recommended Journals
Article Tools
Article Usage
- Total views: 17257
- [From(publication date):
April-2015 - Aug 20, 2025] - Breakdown by view type
- HTML page views : 12396
- PDF downloads : 4861
