Case Report Open Access
Scedosporium Prolificans Sclerokeratitis Following Pterygium Excision with Mitomycin C: A Case Requiring Enucleation
Nickisa M Hodgson1, Helen King2, Bradford W Lee1, Deirdre Amaro3, Don O Kikkawa1, Jonathan H Lin3, Bobby S Korn1 and Randy A Taplitz2*1Division of Ophthalmic Plastic and Reconstructive Surgery, Shiley Eye Institute, University of California, San Diego, California
2Division of Infectious Diseases, Department of Medicine, University of California, San Diego, California
3Department of Pathology, University of California, San Diego, California
- *Corresponding Author:
- Randy A Taplitz
Division of Infectious Diseases, Department of Medicine
University of California, San Diego, California
Tel: (858) 822-0711
Fax: (858) 822-5322
E-mail: rtaplitz@ucsd.edu
Received date: March 07, 2017; Accepted date: March 27, 2017; Published date: April 05, 2017
Citation: Hodgson NM, King H, Lee BW, Amaro D, Kikkawa DO, et al. (2017) Scedosporium prolificans Sclerokeratitis Following Pterygium Excision with Mitomycin C: A Case Requiring Enucleation. J Emerg Infect Dis 2:125. doi: 10.4172/2472-4998.1000125
Copyright: © 2017 Hodgson NM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Infectious Disease and Pathology
Abstract
Scedosporium prolificans is a rare cause of fungal sclerokeratitis following pterygium excision with beta-irradiation or Mitomycin C (MMC). We present a case of a male horticulturist with history of bilateral pterygium excision with MMC who presented for evaluation of refractory sclerokeratitis and was found to have S. prolificans sclerokeratitis. Despite aggressive topical and combination intravenous antifungal therapy, enucleation of the affected eye was required due to evidence of posterior scleral extension on imaging. S. prolificans should be considered in cases of refractory sclerokeratitis. Infections due to S. prolificans often do not respond to antifungal therapies alone and often require swift surgical management.
Keywords
Scedosporium prolificans ; Sclerokeratitis; Fungal; Pterygium; Mitomycin C; Enucleation
Introduction
Scedosporium prolificans (S. prolificans ) is a ubiquitous filamentous fungus found in soil that causes a broad range of clinical infections, often in immunocompromised hosts. Disseminated infection with this emerging opportunistic pathogen is associated with a high mortality rate, partly due to unpredictable antifungal susceptibility patterns and often high-level of resistance to all antifungal classes. There are few prior reports of S. prolificans sclerokeratitis associated with pterygium excision and adjunctive β irradiation or mitomycin C (MMC) and thus no consensus on appropriate clinical management. We present a case of necrotizing sclerokeratitis caused by S. prolificans following pterygium excision with adjunctive MMC managed successfully by enucleation and combination antifungal therapy.
Case Report
A 67-year-old male horticulturist with history of bilateral pterygium excision with MMC presented for evaluation of refractory sclerokeratitis. His medical history was significant for hypertension, coronary artery disease, coronary artery bypass grafting, and hypothyroidism. On initial evaluation at an outside hospital, he presented with left eye pain and redness following recent ocular exposure to gardening mulch. His vision was 20/20 in the right eye and 20/50 in the left eye, and examination revealed periorbital edema and proptosis.
He was treated with oral corticosteroids for presumed pseudotumor and his condition deteriorated with development of a superior corneal ulcer. Two sets of corneal cultures were obtained with no bacterial or fungal growth. A scleral biopsy was performed and unremarkable. Due to scleral thinning, a scleral patch graft was placed and the eye was irrigated with topical voriconazole. Over the course of his treatment, he received intravenous antibiotics, topical voriconazole, and oral fluconazole without clinical improvement. Systemic and topical corticosteroids were resumed due to positive anti-nuclear (ANA) antibody and double stranded-DNA (dsDNA) titers, concerning for an autoimmune process. Despite continued treatment with corticosteroids, antibacterials and antifungal agents the patient’s condition deteriorated, and he was referred to our tertiary care hospital after one month of treatment.
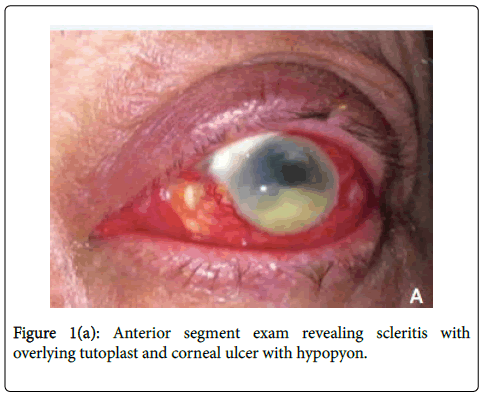
On initial presentation to our facility, his vision was 20/20 in the right eye and hand motion in the left eye. Slit lamp examination was normal in the right eye and notable for marked scleral injection of the left eye with a superonasal scleral patch graft and two small islands of avascular sclera nasally. He had a 4 x 4 mm central corneal infiltrate with satellite lesions and a 4 mm hypopyon (Figure 1a).
B-scan ultrasonography was performed showing scleral thickening and a positive T-sign without evidence of vitritis. Examination was also notable for mild periorbital edema and approximately 5 mm of left proptosis with mild extraocular motility restriction.
The patient was subsequently admitted to the hospital for diagnostic evaluation and inpatient treatment. Corneal scrapings were cultured on blood agar, chocolate agar, Saboraud’s agar and thioglycolate broth. Computed tomography (CT) imaging revealed marked scleral thickening and proptosis. Topical natamycin and oral voriconazole were initiated, due to concern for fungal infection with history of recent ocular exposure to organic debris. Oral prednisone was continued due to concern for an autoimmune process with prior positive ANA and dsDNA results. Rheumatology and infectious disease services were consulted. Repeat rheumatologic workup was notable for ANA 1:160 homogenous titer and negative anti-dsDNA. HIV antibody, syphilis serology and tuberculosis interferon gamma release assay testing were negative.
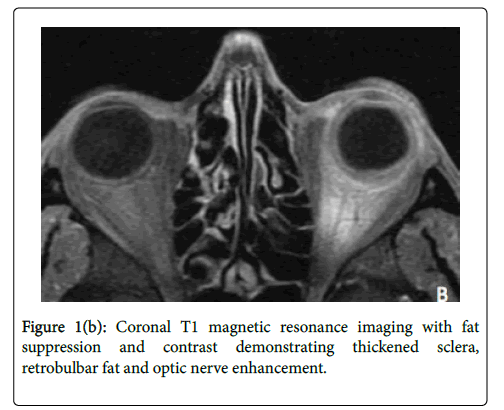
On the third hospital day, fungal culture from the corneal biopsy grew fungus without species identification. At this juncture, the systemic corticosteroid dose was reduced with plans for an expedited taper. On the fourth day of hospitalization, the fungal organism was identified as Scedosporium prolificans . Since S. prolificans has an unpredictable antifungal susceptibility pattern and is typically highly resistant to most therapeutic classes, oral terbinafine and IV micafungin were added, and oral voriconazole was continued. Magnetic Resonance Imaging (MRI) was obtained to evaluate for posterior spread and revealed enhancement of retrobulbar fat and optic nerve of the left eye (Figure 1b).
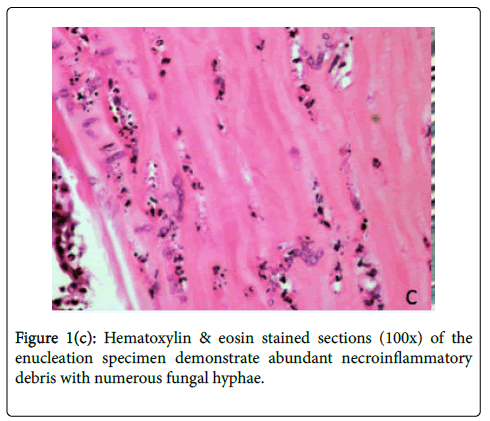
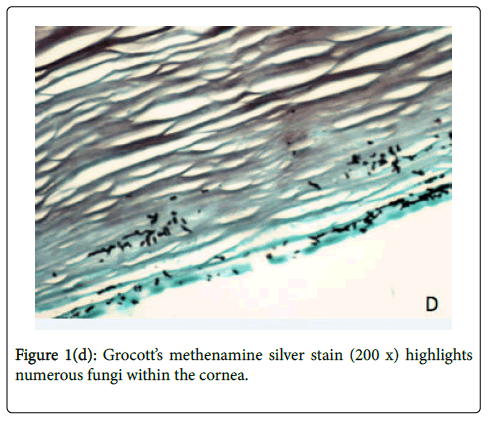
The decision was made to enucleate the eye to prevent further extension of infection, considering evidence of posterior orbital extension of infection on MRI and the highly resistant nature of S. prolificans to antifungal agents. On hospital day 6, the affected eye was enucleated, involving disinsertion and excision of extraocular and oblique muscles and the globe. Implant placement was deferred due to the risk of seeding. Histologic examination of the globe showed numerous fungal hyphae and abundant necroinflammatory debris throughout the cornea and sclera (Figures 1c-1d). Notably, the optic nerve margin and orbital apex, which enhanced on MRI, were free of involvement, confirmed by tissue stains for fungus. Prior to discharge from the hospital, IV micafungin was discontinued after 8 days of treatment. The patient was continued on oral voriconazole 4 mg/kg every 12 hours and terbinafine 250 mg every 12 hours for a total of 10 weeks after enucleation. He has no evidence of disease recurrence and will have an autologous fat graft to the left orbit.
Discussion
Ocular manifestations of S. prolificans occur in both immunocompromised and immunocompetent hosts. Immunocompromised hosts often present with endogenous endophthalmitis representing hematogenous spread and disseminated infection. Cases of ocular S. prolificans infection in immunocompetent hosts have been described subsequent to traumatic inoculation of fungal organisms, surgery, or from contiguous spread. Ocular presentations include keratitis, anterior and posterior scleritis, corneoscleritis and sclerokeratitis [1].
Cases have been reported in Australia, California, and the Southern United States [2]. Susceptibility profiling reveals high mean inhibitory concentrations (MIC) for Scedosporium species and voriconazole has the most favorable profile. Terbinafine in combination with azole therapy has demonstrated in-vitro synergy in 85% of Scedosporium spp. isolates, and other antifungal combinations may also be synergistic [3]. Despite aggressive antifungal therapy, S. prolificans typically requires surgical debridement and is frequently fatal in disseminated disease [4].
One similar case of S. prolificans sclerokeratitis following pterygium excision with MMC has been reported, 5 and three cases described following pterygium excision with β irradiation [5-8]. Two of these reported sclerokeratitis cases required enucleation to control infection [5,6]. A series of 9 cases of Scedosporium spp. ophthalmic infections, including Scedosporium apiospermum , reported two cases of S.prolificans keratitis and endophthalmitis following pterygium excision with β irradiation and both cases required enucleation [9]. Avascular sclera from irradiation, MMC, and excessive cautery can destroy vasculature leaving the eye susceptible to infectious organisms [4]. Diagnosis may be delayed if the presentation mimics noninfectious conditions, such as orbital pseudotumor or autoimmune disease, or if the fungus is not identified on pathology or culture.
Interestingly all cases of S. prolificans sclerokeratitis have been reported in Australia. To the best of our knowledge, this is the first report of S. prolificans sclerokeratitis following pterygium excision with adjunctive MMC in the United States. In our case, the patient had negative corneal cultures and scleral biopsy, delaying definitive diagnosis. Various corneal culture techniques may preclude optimal culture results. Additionally, the patient received corticosteroid therapy likely accelerating the condition. Though corticosteroids were discontinued and maximum medical therapy, enucleation was performed due to posterior scleral involvement on MRI, the highly resistant nature of S. prolificans to antifungal agents, and the risk of dissemination and associated high mortality rate [10].
This report emphasizes the inclusion of this rare infection in the differential diagnosis of sclerokeratitis. In addition, a history of pterygium excision with mitomycin C can result in avascularity and is a predisposing risk factor. S. prolificans has poor response to antifungal therapy and management requires early diagnosis and debridement of infected tissue. Our experience emphasizes that posterior scleral involvement portends a poor prognosis for globe preservation and may require enucleation to control the disease.
References
- Cortez KJ, Roilides E, Quiroz-Telles F, Meletiadis J, Antachopoulos C, et al. (2008) Infections caused by Scedosporium Spp. Clin Microbiol Rev 21: 157-197.
- Idigoras P, Perez-Trallero E, Pineiro L, Larruskain J, Lopez-Lopategui MC, et al. (2001) Disseminated infection and colonization by Scedosporium prolificans: a review of 18 cases, 1990-1999. Clin Infect Dis 32:E158-E165.
- Meletiadi J, Mouton JW, Meis JF, Verweij PE (2003) In vitro drug interaction modeling of combinations of azoles with terbinafine against clinical Scedosporium prolificans isolates. Antimicrob Agents Chemother 47:106-117.
- Thomas PA (2003) Current perspectives on ophthalmic mycoses. ClinMicrobiol Rev 16: 730-797.
- Singh RP, McCluskey P (2005) Scedosporium prolificans sclerokeratitis 10 years after pterygium excision with adjunctive mitomycin C. Clin Experiment Ophthalmol 33: 433-434.
- Sullivan LJ, Snibson G, Joseph C, Taylor HR (1994) Scedosporium prolificans sclerokeratitis. Aust N Z J Ophthalmol 22: 207-209.
- Kumar B, Crawford GJ, Morlet GC (1997) Scedosporium prolificans corneoscleritis: a successful outcome. Aust N Z J Ophthalmol 25: 169-171.
- Moriarty AP, Crawford GJ, McAllister IL, Constable IJ (1993) Severe Corneoscleral Infection: A Complication of Beta Irradiation Scleral Necrosis Following Pterygium Excision. Arch Ophthalmol 111: 947-951.
- Jhanji V, Yohendran J, Constantinou M, Sheorey H, Vajpayee RB (2009) Eye Contact Lens. Scedosporium scleritis or keratitis or both: case series 35: 312-315.
- McKelvie PA, Wong EY, Chow LP, Hall AJ (2001) Scedosporium endophthalmitis: Two fatal disseminated cases of Scedosporium infection presenting with endophthalmitis. Clin Exp Ophthalmol 29: 330-334.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 4546
- [From(publication date):
March-2017 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 3567
- PDF downloads : 979