Research Article Open Access
Sleeve Gastrectomy Ameliorates Mrna Expression of Matrix Metalloprotease I, III, V, IL-1a and IL-6 Genes in Substantia Nigra Tissues of Obese Rats
| Inan Gezgin*, Cem Ozic, Can Hakan Y�?±ld�?±r�?±m, Kemal Kilic, Yusuf Ehi and Miktat Kaya | |
| Department of Neurosurgery, Private Park Hospital, Ad�?±yaman, Turkey | |
| Corresponding Author : | Inan Gezgin Department of Neurosurgery Private Park Hospital, Ad�?±yaman, Turkey Tel: 416 7255066 Fax: 7256502 E-mail: drigezgin1978@ hotmail.com |
| Received December 05, 2015; Accepted January 12, 2016; Published January 15, 2016 | |
| Citation:Gezgin I, Ozic C, Y�?±ld�?±r�?±m CH, Kilic K, Ehi Y, et al. (2016) Sleeve Gastrectomy Ameliorates Mrna Expression of Matrix Metalloprotease I, III, V, IL-1a and IL-6 Genes in Substantia Nigra Tissues of Obese Rats. J Obes Weight Loss Ther 6:294. doi:10.4172/2165-7904.1000294 | |
| Copyright: © 2016 Gezgin I, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
| Related article at Pubmed, Scholar Google | |
Visit for more related articles at Journal of Obesity & Weight Loss Therapy
Abstract
Aim: To reveal metalloproteinase (MMP)-1, MMP-3, MMP-5, interleukin (IL)-1a and IL-6 gene expressions in substantia nigra region of brain in rats which undergone sleeve gastrectomy (SG) surgery. Methods: Rats were allocated into three groups in random, which were normal rats (Group I) (n=14), obese rats (Group II) (n=14), and obese rats subjected to SG (Group III) (n=14). MMP-1, MMP-3, MMP-5, IL-1a and IL-6 gene expressions were determined by polymerase chain reaction (PCR) and Real-time polymerase chain reaction (RTqPCR). Results: When normal (Group I), and obese (Group II) rats were compared, a decrease in expressions of MMP- 3 and IL-6 genes was observed in Group II. When obese rats (Group II) and obese rats subjected to SG (Group III) were compared, increases in the expressions of MMP-3 and IL-6 genes were observed in Group III. This phenomenon demonstrates that SG decreases obesity and consequently increases expressions of MMP-3 and IL-6 genes. Conclusion: These data show alterations of MMP-3 and IL-6 genes in the substantia nigra tissue of obese rats, consistent with the possibility that these changes may contribute to disease molecular background.
| Keywords |
| Obesity; Sleeve gastrectomy; Substantia nigra; Gene expressions |
| Introduction |
| Obesity and its comorbidities have become an important community health problem worldwide [1]. Recent literature revealed that adipocytes not only store fat, but are also an endocrine organ which releases a variety of mediators [2]. These mediators may manipulate, circulating lipid levels, blood pressure, and insulin resistance [2]. Common co-morbidities related with obesity, such as hypertension, hyperlipidemia, and insulin resistance are associated with increased cardiovascular and renal disease, resulting in increased morbidity and mortality [1,2]. A growing body of evidence about the function of adipocytes reveals that they play a key role in inflammatory processes [2]. Sleeve gastrectomy (SG) is a restrictive procedure that not only results in lasting weight loss but also improves insulin sensitivity and glucose tolerance before any significant weight loss has arised [3]. However, it is unclear whether SG has affects in the central nervous system. |
| Metalloproteinases (MMPs) are a group of Zn2+-endopeptidases which are classified by their ability to absorb elements of the extracellular matrix [4]. MMPs are produced mainly by neurons, microglia and astrocytes [5]. Therefore, excessive expression of MMPs may result in neuronal damage. Accumulating evidence indicates that MMPs are participated in a variety of diseases such as bacterial meningitis, stroke, multiple sclerosis, and neurodegenerative diseases [6]. |
| Regardless of the increasing data that MMPs take part in the pathology of obesity, available data on the tissular and cellular expression of the proteins is less detailed [7-9]. Here we aimed to reveal the changes in MMP-1, MMP-3, MMP-5, interleukin (IL)-1a and IL-6 gene expressions, as determined by polymerase chain reaction (PCR) and Real-time polymerase chain reaction (RT-qPCR) in substantia nigra region of brain in rats which undergone SG surgery. |
| Materials and Methods |
| Experimental design |
| All animal studies were carried out with the approval of the Institutional Animal Care and Use Committee. Animals were housed at constant temperature (20-22oC) and humidity (50-60%) with a 12-h light and 12-h dark cycle. They were allowed free access to water and standard rat chow. Rats were allocated into three groups in random, which were normal rats (Group I) (n=14), obese rats (Group II) (n=14), and obese rats subjected to SG (Group III) (n=14). Obese rats were fed with a high-fat diet containing 40% additional fat to the diet. One of the limitations of this study is the short follow up of 8 weeks. |
| Surgical (sleeve gastrectomy) procedure |
| After overnight fasting, rats in the sleeve group were anesthetized with an intraperitoneal injection of 300 mg/kg chloral hydrate and placed in a supine position on a surgical board with the extremities immobilized. A sinister epigastric incision was used for the operation. The length was about 1.5-2 cm in total. The incision was kept open with a blade retractor and dissociates gastric omentum to disclose gastric cardium. The gastric cavity was closed with vascular clamps, and then gastric cavity and hemostating were cut off with a cauterizer. A gastric tube was created with 8-0 unabsorbable suture from the distal antrum (1.5-2 mm from the pylorus) to the Hiss angle. The fundus was completely removed (70-80% of total stomach). After the gastric tube was rebuilt, the peritoneal cavity was cleaned with saline, and then closed with 6-0 silk suture. |
| PCR and RT-qPCR analysis |
| Total RNA was isolated from cultured cells by using TRIzol reagent (Sigma Chemical Co., St. Louis, MO, USA). Total RNA was isolated from exponentially substantia nigra tissues TRIzol Reagent (Sigma Chemical Co., St. Louis, MO, USA). Total RNA was treated with RQ1 DNAse I (Promega, Madison, WI, USA). RT was performed according to the manufacturer’s directions (Fermentas, Vilnius, Lithuania) using 1 unit of MMLV reverse transcriptase with 5 μg of total RNA and oligo dT22 primer. Similar amplification efficiencies of all gene targets and GAPDH were validated, permitting quantitative measurement of gene expression. |
| PCR gene expression of MMP-1, MMP-3, MMP-5, IL-1a and IL-6 were quantified using MyGenie96 Thermo BÄ°ONER PCR. The 25 μl PCR mixture contained were performed using 1 μL of cDNA template, 0.25 μL (5u/ μL) of Taq polymerase, 2.5 μL (100 ng) of primers, 1.5 μL (25mM) dNTP, 2.5 μL 10× PCR buffer and 1.5 μL MgCl2. PCR reactions were performed: 94°C for 3 minutes, 30 cycles at 94°C for 45 seconds, 50-60°C for 45 seconds, and 72°C for 1 minutes, followed by a final extension at 72°C for 10 minutes. |
| Real-time PCR gene expression of MMP-1, MMP-3, MMP-5, IL- 1a and IL-6 were quantified using the Roche Light Cycler real-time PCR analyzer according to the manufacturer’s instructions (Roche Molecular Diagnostics, Mannheim, Germany). The 25 μl PCR mixture contained 12.5 μl of SYBR Green Universal PCR Master Mix (Perkin- Elmer Applied Biosystems, California, USA) 500 nmol/l of primers (forward and reverse), 100 nmol/l of labeled probe, and 1 μl of cDNA template. PCR was performed at 50°C for 2 min, 95°C for 10 min, and then run for 50 cycles at 95°C for 15 s and 60°C for 1 min. |
| The RT-PCR and RT-qPCR assay was used to detect the differential expression of matrix metalloprotease I, III, V, IL-1a and IL-6 between substantia nigra tissues. GAPDH was used as an internal control to normalize samples. Gene-specific primers were designed as follows: metalloprotease I, III, V, IL-1a and IL-6 and GAPDH, 5’-TGATGACATCAAGAAGGTGGTGAAG-3’ and 5’-TCCTTGGAGGCCATGTGGGCCAT-3’. |
| Results |
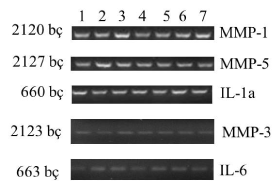
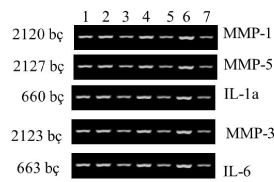
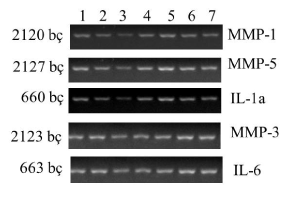
| Within the context of this study, when normal (Group I), and obese (Group II) rats were compared, a decrease in expressions of MMP-3 and IL-6 genes was observed in obese rats (Figures 1 and 2). When obese rats (Group II) and obese rats subjected to SG (Group III) were compared, an increase in the expressions of MMP-3 and IL-6 genes was observed in obese rats subjected to SG (Figure 3). This phenomenon demonstrates that SG decreases obesity and consequently increases expressions of MMP-3, and IL-6 genes. |
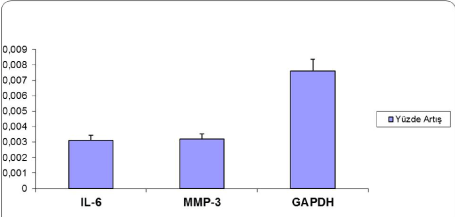
| When groups were compared regarding genes, decreases in the expressions of MMP-3 and IL-6 were observed secondary to obesity. When real-time PCR analysis was performed with the intention to quantitatively control the expressions of genes, decrease in MMP-3 and IL-6 gene expressions was determined (Figure 4). |
| Discussion |
| Here we aimed to reveal the changes in MMP-1, MMP-3, MMP- 5, interleukin (IL)-1a and IL-6 gene expressions in substantia nigra region of brain in rats which undergone SG surgery. Our results showed alterations of MMP-3 and IL-6 genes in the substantia nigra tissue of obese rats, which may contribute to disease molecular background. In the present study, based on the results of PCR and Real-Time PCR, decrease in the expressions of MMP-3 and IL-6 genes in obese rats were resumed after SG procedure, which revealed increase in the expressions of these genes. |
| Obesity is defined by a substandard inflammatory status, generating its consequences on numerous organs. Earlier literatures have found that weight loss is related with a decrease in inflammation markers [e.g. C-reactive protein (CRP)], pro-inflammatory cytokines [e.g.interleukin (IL)-6, tumor necrosis factor (TNF)], Metallothioneins (MT1A and MT2A), adipokines leptin (LEP), adiponectin (ADIPOQ), the nuclear receptor, Peroxisome Proliferator Activated Receptor γ (PPARγ) and Leptin [10-15]. |
| Weight reduction by conservative methods is difficult to achieve, and bariatric surgery is currently the most noteworthy management of significant and sustained body weight loss in morbidly obese patients [16]. Hasani et al. showed that observed a significant difference for REE between obese patients after the surgery and controls as well as between prior-and post-surgery [17]. Recent evidence suggests that SG leads to a decreased food intake not only by mechanical restriction of the stomach but also by reducing a subject’s appetite [18]. SG is supposed to modulate meal-related hormones of the gut-brain axis such as Ghrelin (signal=hunger) or peptide YY (slows gastric emptying) or GLP-1 (decreases food intake) [19]. |
| Traurig et al., using PCR analysis, compared obese and nonobese individuals, and revealed that MMP3 mRNA levels were decreased in preadipocytes isolated from obese individuals, and also showed a significant negative correlation between MMP3 mRNA expressions and percent body fat [20]. Adipose tissue remodeling is mainly controlled by MMPs [21,22]. The expressions of different MMPs and tissue inhibitors of MMPs (TIMPs) were examined in adipose tissue of obese mouse models by Chavey et al. and Maquoi et al. [21,22]. Both studies revealed statistically significant alterations in expression levels and emphesized that adipose tissue remodeling in obese individuals is modulated by these proteins. Also in various studies, MMPs also seem to be important for adipogenesis [21-24]. |
| There are various studies both confirming and opposing our results of reduced MMP3 expression in obese subjects’ preadipocytes/stromal vascular cells. Maquoi et al. showed that MMP3-/- mice had improved hypertrophy of adipocytes as fed with a high-fat diet; while Alexander et al. showed that MMP3-/- mice had increased differentiation of adipocytes [25,26]. On the contrary, both Maquoi et al. and Chavey et al. revealed that MMP3 expression levels were enhanced in obese mice adipose tissue [21,22]. The explanation for the mentioned divergence is uncertain. This disparity might probably be generated by variations in model systems (human vs. mouse models, or non–diet-induced vs. diet-induced obesity) or alternatively by differences in adipose tissue depots (subcutaneous vs. gonadal). Geffken et al. revealed that physically more active individuals have lower IL-6 and CRP levels and other inflammation markers [27]. Furthermore, continuous exercise may attenuate the inflammatory process, thus decreasing levels of proinflammatory cytokines. Similarly, obese individuals are identified by having elevated levels of inflammatory markers than do skinny individuals [28]. Ziccardi et al. and Abad et al. showed that weight loss due to diet reduces CRP, IL-6, and TNF levels [29,30]. Dileone, Elmquist et al. Figlewicz et al. and Hommel et al. showed that leptin receptors are also expressed in the substantia nigra, which would likely influence the dorsal striatum [31-34]. Li et al. showed that suggests that the upor down-regulation of tyrosine hydroxylase mRNA expression in the VTA, VMH, and SN is mainly due to the intake of macronutrient type rather than body weight [35]. In the present study, when normal and obese rats at substantia nigra were compared, a decrease in expressions of MMP-3 and IL-6 genes was observed in obese rats at substantia nigra. When obese rats and obese rats subjected to SG were compared, an increase in the expressions of MMP-3 and IL-6 genes was observed in obese rats at substantia nigra in subjected to SG. This phenomenon demonstrates that SG decreases molecular background obesity and consequently increases expressions of MMP-3, and IL-6 genes. |
| One of the limitations of this study is the short follow up of 8 weeks. However, this time period is the equivalent of 6 human years [36]. While we cannot exclude the potential for weight regain following sleeve gastrectomy, other investigators have demonstrated that weight loss is maintained at a 15-week timepoint following sleeve gastrectomy in rodents [37,38]. Li et al. showed that examined tyrosine hydroxylase mRNA expression in obese mice fed a high-fat diet. After 8 week feeding of high-fat diet mice were classified as diet-induced obese and obese-resistant according to body weight gain [35]. Dunn-Meynell et al. showed that ibotenic acid injected into the VMN, substantia nigra, pars reticulata, and pars compacta destroyed intrinsic neurons and/or their local processes and decreased low-affinity 3H-mazindol binding by 13%-22% [39]. Teske et al. showed that OXA-stimulated SPA is not secondary to enhanced arousal, propensity for SPA parallels inclination to run and that orexin action on dopaminergic neurons in SN may participate in mediation of SPA and running wheel activity [40]. Another limitation of the study is a relatively high complication rate in these rats. These results were obtained after several pilot studies, refinements of technique, and development of a rigorous perioperative-care protocol as delineated in the methods. Although we still consider our results as preliminary, they warrant a larger comprehensive study, one which would include more rats at substantia nigra in order to evaluate more precisely the role of MMPs in obesity molecular background. |
| Conclusion |
| In conclusion, the authors showed alterations of MMP-3 and IL-6 genes in the substantia nigra tissue of obese rats, which may contribute to disease molecular background for the first time in the English literature. We revealed that SG decreases molecular obesity and consequently increases expressions of MMP-3, and IL-6 genes. |
References
- Huffman MD, Capewell S, Ning H, Shay CM, Ford ES, et al. (2012) Cardiovascular health behavior and health factor changes (1988-2008) and projections to 2020: results from the National Health and Nutrition Examination Surveys.Circulation 125: 2595-2602.
- Wu G, Li H, Zhou M, Fang Q, Bao Y, et al. (2014) Mechanism and clinical evidence of lipocalin-2 and adipocyte fatty acid-binding protein linking obesity and atherosclerosis.Diabetes Metab Res Rev 30: 447-456.
- Peterli R, Wölnerhanssen B, Peters T, Devaux N, Kern B, et al. (2009) Improvement in glucose metabolism after bariatric surgery: comparison of laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy: a prospective randomized trial.Ann Surg 250: 234-241.
- Stetler-Stevenson WG (1996) Dynamics of matrix turnover during pathologic remodeling of the extracellular matrix.Am J Pathol 148: 1345-1350.
- Asahina M, Yoshiyama Y, Hattori T (2001) Expression of matrix metalloproteinase-9 and urinary-type plasminogen activator in Alzheimer's disease brain.Clin Neuropathol 20: 60-63.
- Costa S, Planchenault T, Charriere-Bertrand C, Mouchel Y, Fages C, et al. (2002) Astroglialpermissivity for neuritic outgrowth in neuron-astrocyte cocultures depends on regulation of laminin bioavailability. Glia 37:105-113
- Huang HL, Wu S, Hsu LA, Teng MS, Lin JF, et al. (2013) Genetic variants associated with circulating MMP1 levels near matrix metalloproteinase genes on chromosome 11q21-22 in Taiwanese: interaction with obesity. BMC Med Genet 14:30
- Belo VA, Souza-Costa DC, Lana CM, Caputo FL, Marcaccini AM, et al. (2009) Assessment of matrix metalloproteinase (MMP)-2, MMP-8, MMP-9, and their inhibitors, the tissue inhibitors of metalloproteinase (TIMP)-1 and TIMP-2 in obese children and adolescents.Clin Biochem 42: 984-990.
- Belo VA, Luizon MR, Carneiro PC, Gomes VA, Lacchini R, et al. (2013) Effect of metabolic syndrome risk factors and MMP-2 genetic variations on circulating MMP-2 levels in childhood obesity. MolBiol Rep 40:2697-2704.
- Guex N, Peitsch MC (1997) SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling.Electrophoresis 18: 2714-2723.
- Fenske WK, Dubb S, Bueter M, Seyfried F, Patel K, et al. (2013) Effect of bariatric surgery-induced weight loss on renal and systemic inflammation and blood pressure: a 12-month prospective study.Surg ObesRelat Dis 9: 559-568.
- Nicklas BJ, Ambrosius W, Messier SP, Miller GD, Penninx BW, et al. (2004) Diet-induced weight loss, exercise, and chronic inflammation in older, obese adults: a randomized controlled clinical trial. Am J Clin Nutr 79:544-551.
- Cancello R, Zulian A, Gentilini D, Mencarelli M, Della Barba A, et al. (2013) Permanence of molecular features of obesity in subcutaneous adipose tissue of ex-obese subjects. International Journal of Obesity 37: 867-873.
- Pereira-Fernandes A, Dirinck E, Dirtu AC, Malarvannan G, Covaci A, et al. (2014) Expression of Obesity Markers and Persistent Organic Pollutants Levels in Adipose Tissue of Obese Patients: Reinforcing the Obesogen Hypothesis? PLoS ONE 9: e84816.
- Wasim M (2015) Obesity and Leanness Caused by Mutations in the Leptin Gene: Already 6 Pathogenic Mutations Reported in this Gene. J Obes Weight Loss Ther 5:276
- Stefater MA, Pérez-Tilve D, Chambers AP, Wilson-Pérez HE, Sandoval DA, et al. (2010) Sleeve gastrectomy induces loss of weight and fat mass in obese rats, but does not affect leptin sensitivity. Gastroenterology 138:2426-2436
- Hasani M, Mirahmadian M, Taheri E, Qorbani M, Talebpour M,et al.(2015) The Effect of Laparoscopic Gastric Plication Surgery on Body Composition, Resting Energy Expenditure, Thyroid Hormones and Physical Activity in Morbidly Obese Patients. J Obes Weight Loss 5: 275
- DimitriadisE, Daskalakis M, Kampa M, Peppe A, Papadakis JA, et al. (2013) Alterations in gut hormones after laparoscopic sleeve gastrectomy: a prospective clinical and laboratory investigational study. Ann Surg 257:647-654.
- Basso N, Capoccia D, Rizzello M, Abbatini F, Mariani P, et al. (2011) First-phase insulin secretion, insulin sensitivity, ghrelin, GLP-1, and PYY changes 72 h after sleeve gastrectomy in obese diabetic patients: the gastric hypothesis. Surg Endosc 25:3540-3550.
- Traurig MT, Permana PA, Nair S, Kobes S, Bogardus C, et al. (2006) Differential expression of matrix metalloproteinase 3 (MMP3) in preadipocytes/stromal vascular cells from nonobesenondiabetic versus obese nondiabetic Pima Indians. Diabetes 55:3160-3165
- Chavey C, Mari B, Monthouel MN, Bonnafous S, Anglard P, et al. (2003) Matrix metalloproteinases are differentially expressed in adipose tissue during obesity and modulate adipocyte differentiation. J BiolChem 278:11888-11896
- Maquoi E, Munaut C, Colige A, Collen D, Lijnen HR (2002) Modulation of adipose tissue expression of murine matrix metalloproteinases and their tissue inhibitors with obesity. Diabetes 51:1093-1101
- Croissandeau G, Chrétien M, Mbikay M (2002) Involvement of matrix metalloproteinases in the adipose conversion of 3T3-L1 preadipocytes.Biochem J 364: 739-746.
- Bouloumie A, Sengenes C, Portolan G, Galitzky J, Lafontan M (2001) Adipocyte produces matrix metalloproteinases 2 and 9: involvement in adipose differentiation. Diabetes 50:2080-2086.
- Alexander CM, Selvarajan S, Mudgett J, Werb Z (2001) Stromelysin-1 regulates adipogenesis during mammary gland involution.J Cell Biol 152: 693-703.
- Maquoi E, Demeulemeester D, Vörös G, Collen D, Lijnen HR (2003) Enhanced nutritionally induced adipose tissue development in mice with stromelysin-1 gene inactivation.ThrombHaemost 89: 696-704.
- Geffken D, Cushman M, Burke G, Polak J, Sakkinen P, Tracy R (2001) Association between physical activity and markers of inflammation in a healthy elderly population. Am J Epidemiol 153:242-250
- Smith JK, Dykes R, Douglas JE, Krishnaswamy G, Berk S (1999) Long-term exercise and atherogenic activity of blood mononuclear cells in persons at risk of developing ischemic heart disease.JAMA 281: 1722-1727.
- Ziccardi P, Nappo F, Giugliano G, Esposito K, Marfella R, et al. (2002) Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 105:804-809.
- Abad LW, Schmitz HR, Parker R, Roubenoff R (2002) Cytokine responses differ by compartments and wasting status in patients with HIV infection and healthy controls. Cytokine 18:286-293.
- DiLeone RJ (2009) The influence of leptin on the dopamine system and implications for ingestivebehavior.Int J Obes (Lond) 33 Suppl 2: S25-29.
- Elmquist JK, Bjørbaek C, Ahima RS, Flier JS, Saper CB (1998) Distributions of leptin receptor mRNA isoforms in the rat brain.J Comp Neurol 395: 535-547.
- Figlewicz DP, Evans SB, Murphy J, Hoen M, Baskin DG (2003) Expression of receptors for insulin and leptin in the ventral tegmental area/substantianigra (VTA/SN) of the rat.Brain Res 964: 107-115.
- Hommel JD, Trinko R, Sears RM, Georgescu D, Liu ZW, et al. (2006) Leptin receptor signaling in midbrain dopamine neurons regulates feeding. Neuron 51: 801-810.
- Li Y, South T, Han M, Chen J, Wang R, et al. (2009) High-fat diet decreases tyrosine hydroxylase mRNA expression irrespective of obesity susceptibility in mice.Brain Res 1268: 181-189.
- Christou NV, Look D, Maclean LD (2006) Weight gain after short- and long-limb gastric bypass in patients followed for longer than 10 years. Ann Surg 244:734-740.
- Kotidis EV, Koliakos GG, Baltzopoulos VG, Ioannidis KN, Yovos JG, et al. (2006) Serum ghrelin, leptin and adiponectin levels before and after weight loss: comparison of three methods of treatment--a prospective study.Obes Surg 16: 1425-1432.
- Patrikakos P, Toutouzas KG, Perrea D, Menenakos E, Pantopoulou A, et al. (2009) A surgical rat model of sleeve gastrectomy with staple technique: long-term weight loss results.Obes Surg 19: 1586-1590.
- Dunn-Meynell AA, Levin BE (1997) Location and effect of obesity on putative anorectic binding sites in the rat brain.Obes Res 5: 201-207.
- Teske JA, Billington CJ, Kotz CM (2014) Mechanisms underlying obesity resistance associated with high spontaneous physical activity.Neuroscience 256: 91-100.
Figures at a glance
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
Relevant Topics
- Android Obesity
- Anti Obesity Medication
- Bariatric Surgery
- Best Ways to Lose Weight
- Body Mass Index (BMI)
- Child Obesity Statistics
- Comorbidities of Obesity
- Diabetes and Obesity
- Diabetic Diet
- Diet
- Etiology of Obesity
- Exogenous Obesity
- Fat Burning Foods
- Gastric By-pass Surgery
- Genetics of Obesity
- Global Obesity Statistics
- Gynoid Obesity
- Junk Food and Childhood Obesity
- Obesity
- Obesity and Cancer
- Obesity and Nutrition
- Obesity and Sleep Apnea
- Obesity Complications
- Obesity in Pregnancy
- Obesity in United States
- Visceral Obesity
- Weight Loss
- Weight Loss Clinics
- Weight Loss Supplements
- Weight Management Programs
Recommended Journals
Article Tools
Article Usage
- Total views: 10134
- [From(publication date):
February-2016 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 9233
- PDF downloads : 901
