Research Article Open Access
The Effectiveness of a Partial Meal Replacement Program in Extremely Obese Individuals: A Systematic Retrospective Chart Review of Medifast Weight Control Centers
| Jessica R. Kiel1*, Christopher D. Coleman1, Andrea H. Mitola2, Janice S. Langford1, Kevin N. Davis1 and Linda M. Arterburn1 | |
| 1Department of Scientific and Clinical Affairs, Medifast, USA | |
| 2Independent Consultant, Clifton Park, USA | |
| Corresponding Author : | Jessica R. Kiel Department of Scientific and Clinical Affairs Medifast, Inc. 11445 Cronhill Drive Owings Mills, MD, 21117, USA Tel: 410-504-8164 Fax: 443-471-3341 E-mail: JKiel@choosemedifast.com |
| Received: July 13, 2015 Accepted: July 30, 2015 Published: July 31, 2015 | |
| Citation: Kiel JR, Coleman CD, Mitola AH, Langford JS, Davis KN, et al. (2015) The Effectiveness of a Partial Meal Replacement Program in Extremely Obese Individuals: A Systematic Retrospective Chart Review of Medifast Weight Control Centers. J Obes Weight Loss Ther S5:007. doi:10.4172/2165-7904.S5-007 | |
| Copyright: © 2015 Kiel JR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
| Related article at Pubmed, Scholar Google | |
Visit for more related articles at Journal of Obesity & Weight Loss Therapy
Abstract
Background: Extreme obesity is associated with elevated risks of morbidities and mortality, and the prevalence of this condition has been rising. Lifestyle interventions are the cornerstone of all treatment options, yet relatively few studies have assessed the effectiveness of commercial programs for attaining clinically meaningful weight loss (≥ 5%) in this population. The purpose of this study was to evaluate the effectiveness of the Medifast 5 & 2 & 2 Plan™ administered along with counseling in obese adults, a majority of whom were extremely obese. Methods: We conducted a systematic retrospective chart review of 62 obese clients from 17 Medifast Weight Control Centers® (MWCCs). Weight, body composition and cardiometabolic risk factor data were abstracted through 24 weeks. Data were recorded electronically, and key data points were independently verified. The primary endpoint was change from baseline body weight at 12 weeks, assessed using Wilcoxon signed rank tests. Results: The population consisted of 57% men, and 82% had a body mass index of ≥ 40 kg/m2. Mean body weight among completers was reduced by 12.9±7.1 kg (-8.6%, n=37) at the 12-week primary endpoint and by 19.3 ± 11.4 kg (12.5%, n=17) at 24 weeks (p<0.0001). At 12 and 24 weeks, 76% and 88% of those remaining on the plan, respectively, had lost ≥ 5% of their baseline body weight. Fat mass accounted for a majority (68-80%) of the weight lost, resulting in improvements in body composition. Significant improvements in blood pressure and central adiposity were also observed. Program adherence was >80%, and the meal plan was well-tolerated. Conclusions: The 5 & 2 & 2 Plan used at MWCCs was effective for achieving clinically meaningful weight loss and improving cardiometabolic risk factors in a population of extremely obese individuals. This lifestyle program represents a viable first line approach for meeting treatment goals in extremely obese adults. #NCT0215083.
Methods: We conducted a systematic retrospective chart review of 62 obese clients from 17 Medifast Weight Control Centers® (MWCCs). Weight, body composition and cardiometabolic risk factor data were abstracted through 24 weeks. Data were recorded electronically, and key data points were independently verified. The primary endpoint was change from baseline body weight at 12 weeks, assessed using Wilcoxon signed rank tests.
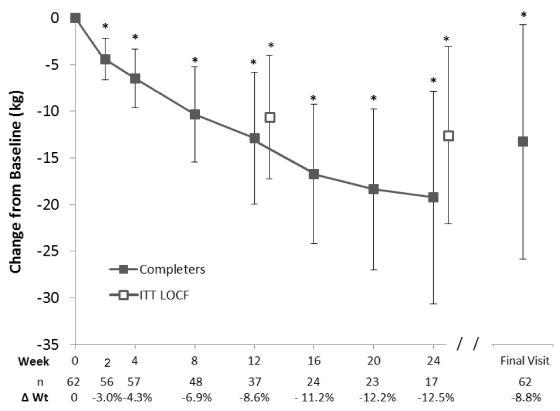
Results: The population consisted of 57% men, and 82% had a body mass index of ≥ 40 kg/m2. Mean body weight among completers was reduced by 12.9 ± 7.1 kg (-8.6%, n=37) at the 12-week primary endpoint and by 19.3 ± 11.4 kg (12.5%, n=17) at 24 weeks (p<0.0001). At 12 and 24 weeks, 76% and 88% of those remaining on the plan, respectively, had lost ≥5% of their baseline body weight. Fat mass accounted for a majority (68-80%) of the weight lost, resulting in improvements in body composition. Significant improvements in blood pressure and central adiposity were also observed. Program adherence was >80%, and the meal plan was well-tolerated.
Conclusions: The 5 & 2 & 2 Plan used at MWCCs was effective for achieving clinically meaningful weight loss and improving cardiometabolic risk factors in a population of extremely obese individuals. This lifestyle program represents a viable first line approach for meeting treatment goals in extremely obese adults. #NCT0215083.
Pharmacotherapy used as an adjunct to lifestyle changes can assist with weight loss in individuals who are obese, but it is considered as a second line treatment when lifestyle intervention alone is not successful [11]. For the higher BMI categories, bariatric surgery is another treatment option that generally results in greater weight loss and improvements in co-morbid conditions compared to less invasive interventions [8]. However, this option is also only indicated for those who have not responded to lifestyle intervention alone. Additionally, not everyone is eligible for surgery, not everyone wants to have bariatric surgery, and the side effects associated with this option are generally higher than those of lifestyle interventions which can still result in a more modest weight loss of 5-10% that is clinically meaningful and has been shown to lower disease risk [12].
The disproportionate growth of the extremely obese population, combined with the increased health risks and the low number treated with surgical interventions, highlight the need for comprehensive lifestyle interventions where the weight loss achieved is, at a minimum, clinically meaningful [9,13]. In fact, current guidelines recommend participation in a comprehensive lifestyle program as the first step in the treatment of obesity, including extreme obesity, and lifestyle interventions serve as the basis for all treatments of obese individuals [8]. These same guidelines support the use of commercial programs that provide a comprehensive lifestyle intervention, provided they are backed by evidence of their safety and efficacy. However, few commercial programs have focused on the extremely obese population, and relatively few clinical studies have investigated the effectiveness of diet and lifestyle interventions in this population.
The Medifast 5 & 2 & 2 Plan is a commercial program often recommended for individuals with over 100 pounds (45.5 kg) of weight to lose or a BMI ≥40 kg/m2. The 5 & 2 & 2 Plan is one of several programs that feature a combination of Medifast meal replacements, conventional food choices, and customizable levels of support for weight loss and weight maintenance. At Medifast Weight Control Centers® (MWCC), the Medifast meal plans are combined with individualized one-on-one weekly counseling to create a comprehensive lifestyle program. Other Medifast plans (the 5 & 1 Plan® and the 4 & 2 & 1 Plan™) have been evaluated and shown to be effective for weight loss in overweight and obese populations [14-17]. In addition, previous studies have shown that portion-controlled meal replacements enhance dietary compliance and are helpful for achieving clinically meaningful, sustainable weight loss in overweight and obese populations [14,15,18-21], including those with extreme obesity [13,22,23]. The purpose of this study was to evaluate the effectiveness of the Medifast 5 & 2 & 2 Plan for weight loss over 24 weeks (12-week primary endpoint) by systematically reviewing charts from MWCC clients who followed this meal plan, the vast majority of whom were extremely obese. Secondary objectives included assessing effects on body composition and cardiometabolic risk factors in this population.
The weight management program offered at the MWCC consists of weekly one-on-one in-person sessions with MWCC counselors who utilize motivational interviewing and a series of personalized behavior change strategies designed to develop behaviors that promote long-term weight management through a healthy lifestyle. MWCC counselors are trained using a combination of on-the-job and corporate-based training to ensure thorough knowledge of the Medifast products and programs and an understanding of the behavior change strategies used at MWCC. A client’s weight loss goals are determined jointly by the counselor and client, which in turn determines the prescribed length of the client’s active weight loss phase and overall weight management program. Programs generally include active Weight Loss, Transition, and Maintenance phases. The meal plan chosen for weight loss is also determined jointly based on a number of factors including the client’s personal preferences, lifestyle, exercise habits and medical history.
The 5 & 2 & 2 Plan is a calorie- and portion-controlled meal plan designed to stimulate gradual, steady weight loss and provides 1,300-1,500 calories daily. It consists of 5 Medifast Meal Replacements (Medifast, Inc., Owings Mills, MD, USA), 2 self-prepared “lean and green meals” (each including 5-7 oz. of lean protein, 3 servings (~1½ cups) of non-starchy vegetables, and up to 2 healthy fat servings), and 2 healthy snacks (fruit, dairy or whole grains). Medifast Meal Replacements, of which there are over 70 to choose from, each contain 90-110 calories, 11-15 g protein (primarily from soy and/or dairy), 8-15 g carbohydrates, and 0-3.5 g fat; they each share a similar nutritional profile and can be used interchangeably during the Weight Loss phase and with any of the Medifast weight loss meal plans. After completion of the Weight Loss phase, some MWCCs may include a Transition phase, during which total calories and conventional food choices are gradually increased. All individuals who have met their weight loss goal during the active Weight Loss phase or who have completed their prescribed weight loss weeks then have the option to enter the Maintenance phase. The Medifast Maintenance Plan is based on a client’s Total Energy Expenditure (TEE) and generally includes 3 Medifast Meal Replacements and 3 self-prepared meals (consisting of conventional food choices with serving sizes based on the Exchange List for Weight Management; the number of servings is individualized based on TEE).
Data were recorded in client charts at MWCCs by counselors. Counselors were trained to use consistent procedures when obtaining weights and anthropometric measurements. Weight was measured to the nearest 0.1 pound using a high-quality digital scale. Body composition was assessed without shoes and in light indoor clothing by direct, segmental, multi-frequency bioelectrical impedance analysis using either an InBody 230® or InBody 370® body composition analyzer (InBody Co., Cerritos, CA, USA); measurements (fat mass, percent body fat and fat free mass) from the InBody analyzer are highly correlated (r ≥ 0.97) with those obtained using dual energy X-ray absorptiometry [25]. Blood pressure and pulse were measured using digital arm blood pressure monitors. Adherence was assessed based on clients’ visit attendance and self-reported meal replacement consumption.
Weight, pulse, blood pressure, and adherence-related information were abstracted at baseline and weekly throughout the client’s weight loss phase through 24 weeks plus at the Final Visit. The Final Visit was defined as the client’s last visit to the MWCC during active weight loss while following the 5 & 2 & 2 Plan; the time of the Final Visit varied by individual client. Anthropometrics and body composition information, which were measured approximately every 4 weeks at the MWCCs, were also collected. When available, body weight data and the corresponding dates were abstracted at the beginning and end of any other MWCC meal plans or program phases that followed a client’s use of the 5 & 2 & 2 Plan.
Notations of adverse signs, symptoms or incidents that occurred while a client was on the 5 & 2 & 2 Plan were abstracted verbatim from the chart notes, regardless of whether or not the incident appeared to be related to the intervention. This information was reviewed and categorized by a registered nurse, and simple frequencies were tabulated.
Chart data were abstracted by trained study personnel directly into electronic case report forms developed using IBM SPSS Data Collection Author and Interviewer Version 7, according to conventions developed for this study. A two-user, independent (double-data) data entry procedure was used for verification of all key data points.
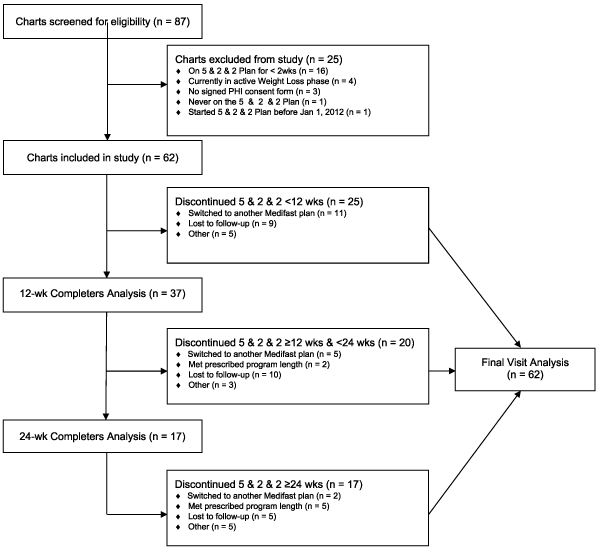
Data were analyzed according to a pre-defined statistical analysis plan. Wilcoxon signed rank tests (i.e., paired t-tests for repeated measures nonparametric data) were used to compare within group changes in weight at 12 weeks compared to baseline for the primary analysis and at other predetermined time points (1, 2, 4, 8, 16, 20 and 24 weeks). Due to the retrospective nature of the study, predefined windows that used data closest to the specified time point were established to optimize the sample without bias: data were included if available within ± 3 days for the 1 and 2 week time points, ± 7 days for the 4 week time point, and within ± 10 days for the remaining time points. For the primary analysis, a completers analysis was used; this analysis included each chart that had data for the given outcome and time point, irrespective of whether the individual completed his/her entire program. Similar analyses were conducted on secondary outcomes. For comparison, an Intention-To-Treat (ITT) Last Observation Carried Forward (LOCF) analysis was pre-specified in the protocol, and performed for the primary outcome. If missing, imputed data were carried through from the last measured observation to each client’s last prescribed week of weight loss. Additionally, in order to maximize the use of all data, including those with missing data, a pre-specified mixed model regression approach was used on the primary outcome, weight, with time as the independent variable and baseline weight as a covariate. The proportions of individuals achieving ≥ 5% and ≥ 10% weight loss from baseline were calculated. Post hoc subgroup analyses for each gender were performed for body weight. Significance was defined as p<0.05 with no adjustments for multiplicity. Analyses were conducted using SPSS Version 14.0 and Stata Version 10.
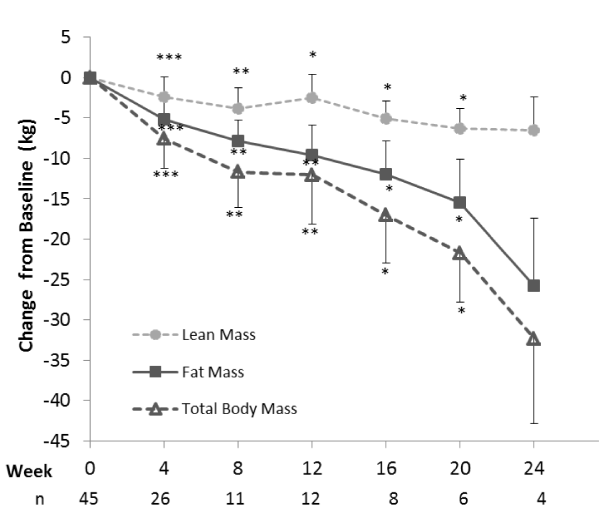
The most rapid reductions in baseline body weight occurred in the earlier time points, with decreases of 4.4 ± 2.3 kg or -3.0% (p<0.0001, n=56) during the first 2 weeks and 6.5 ± 3.1 kg or -4.3% from baseline over the first 4 weeks on the plan (p<0.0001, n=57). Among the 17 individuals who had weight data at 24 weeks, all had lost weight (mean 19.3 ± 11.4 kg, 12.5%), and BMI was reduced by 6.16 ± 3.54 kg/m2 (p<0.0001). In a random effects regression model, controlling for baseline weight, the average rates of weight loss (95% CI) over 2, 4, 12 and 24 weeks were 2.25(2.54,1.95), -1.56(-1.71,-1.42), 1.04( 1.10,0.98), and 0.79(0.85,-0.75) kg per week. Baseline weight and time were the only significant covariates in this model. Mean weight change at the final visit was 13.3 ± 12.6 kg corresponding to an 8.8% reduction in baseline body weight (p<0.0001).
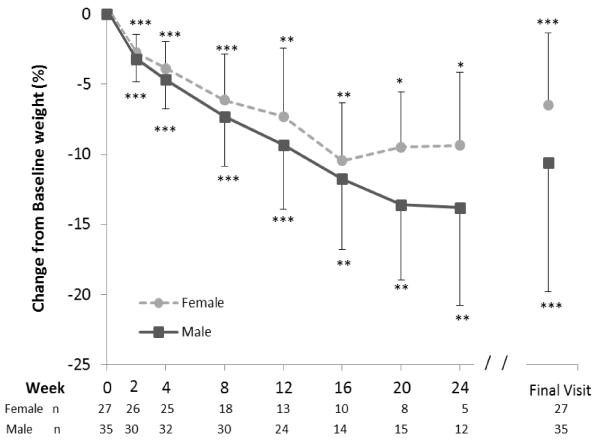
Both males and females had significant reductions from baseline in body weight throughout the study period (p<0.05 for each gender at each time point (Figure 3).
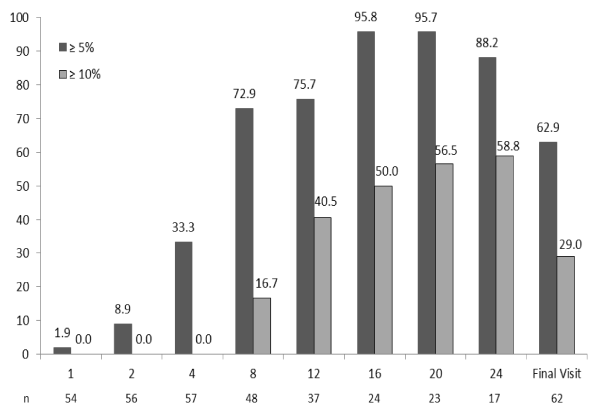
The results of the ITT LOCF analyses also showed significant weight reductions throughout the 24 weeks (p<0.0001 at all time points; 10.6 ± 6.7 and -12.6 ± 9.5 kg at 12 and 24 weeks, respectively, see Figure 2). In the completers population (n=57) at week 4, 33% had lost at least 5% of their baseline body weight (Figure 4). Among individuals who followed the plan for at least 12 weeks (n=37), 76% had lost at least 5% and 41% had lost at least 10% of their baseline body weight. At 24 weeks, 88% of those still on the program (n=17) lost at least 5% and 59% had lost at least 10% of their baseline body weight. At Final Visit (n=62), 62.9% had lost ≥ 5% of their baseline body weight. In the ITT analysis, 63% and 66% had lost ≥ 5% of their baseline weight by 12 (n=62) and 24 weeks (n=56), respectively (data not shown).
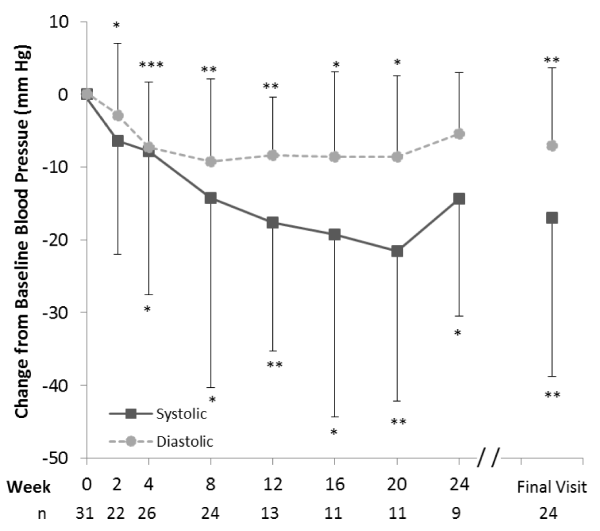
Significant (p<0.05) reductions in systolic and diastolic blood pressure were observed at all time points through 20 weeks (Figure 6). Systolic and diastolic blood pressure were reduced by 17.7 ± 17.6 (p=0.008) and 8.4 ± 8.0 mm Hg (p=0.003), respectively, at 12 weeks (n=13).
At 24 weeks (n=9), one person had an increase in systolic blood pressure, which in the context of the small sample size, lead to a smaller average reduction in the systolic blood pressure (-14.3 ± 16.2 mm Hg, p=0.028) relative to previous weeks. There was a non-significant reduction in diastolic blood pressure (-5.4 ± 8.4 mm Hg, p=0.075) at 24 weeks. Overall, the improvements in blood pressure were reflected in favorable shifts in individual blood pressure categories (i.e. from hypertensive to prehypertensive or from prehypertensive to normal) in 26.9%, 53.8% and 44.4% of individuals at weeks 4, 12 and 24, respectively. Reductions in pulse rates were not significant at any time point (data not shown).
Baseline waist and hip circumferences of 136.2 ± 18.4 cm (n=52) and 150.4 ± 23.3 cm (n=49), respectively, decreased significantly through 20 weeks (p<0.05), with non-significant reductions in both measures at 24 weeks. At 12 weeks, waist and hip circumferences decreased by 9.0 ± 4.7 cm and 10.6 ± 7.5 cm (n=10, p<0.01), respectively, and at 24 weeks, by 10.0 ± 9.9 cm and 13.1 ± 8.5 cm (n=3, p=0.109), respectively. There were no changes in the waist-to-hip ratio.
Individuals on the 5 & 2 & 2 Plan were prescribed an average of 44.2 ± 19.5 weeks (range 14-100 weeks) of weight loss, and stayed on the 5 & 2 & 2 Plan for an average of 18.1 ± 15.8 weeks. Twenty nine percent (18/62) of the individuals who started the Weight Loss phase on the 5 & 2 & 2 Plan subsequently discontinued this plan in order to switch to one or more alternative Medifast weight loss plan(s) and spent an average of 28.6 additional weeks in active weight loss. During this time, they continued to lose an additional 8.8 ± 11.1 kg of body weight (p=0.002; -7.09% of their baseline body weight), for a total loss of approximately 19.6 ± 13.6 kg (-14.4% of their baseline body weight) over their entire Weight Loss phase (using the 5 & 2 & 2 Plan and other Medifast weight loss plans).
Eight percent (5/62) of individuals who originally started the 5 & 2 & 2 Plan entered the Transition phase. The average length of time spent in Transition was 4.2 weeks, during which time individuals regained 0.9 ± 3.1 kg (p=0.893). Eleven percent (7/62) entered the Maintenance phase after losing an average of 39.0 ± 20.7 kg (compared to baseline) during their entire prior Weight Loss and Transition phases, as applicable. While in the Maintenance phase, the group regained an average of 4.6 ± 8.1 kg (p=0.116) or 3.4% of their baseline body weight; the mean length of time spent in the Maintenance Phase was 17 weeks. Despite this regain, those who entered the Maintenance phase experienced a total net weight loss (from baseline to the end of Maintenance) of 34.4 ± 19.2 kg (p=0.018) or 24% of their baseline weight.
The 5 & 2 & 2 Plan generally compared favorably to other published lifestyle interventions in similar populations, although direct study comparisons are challenging given the heterogeneity in intervention types, study duration and study design. The pooled mean weight loss in recent meta-analysis evaluating lifestyle interventions in short term studies (<6 month duration) of individuals with class II and III obesity was 7.2 kg (95% CI: 8.9, 5.5). These lifestyle interventions included a nutrition component (education, recommendations, caloric restriction, etc.) together with a physical activity component. The mean weight loss in this study (-12.9 kg at 12 wks and -19.3 kg at 24 wks in the completers and -10.6 kg at 12 wks and -12.6 kg at 24 wks in the ITT analysis) was greater than the weight loss reported in most, but not all, studies included in that meta-analysis [23]. Weight loss on the 5 & 2 & 2 Plan in both the completers and ITT populations at 6 months also exceeded that reported in two other studies evaluating the effect of physical activity along with behavioral intervention and caloric restriction in extremely obese adults [26,27]. Not surprisingly, given that caloric intake associated with the 5 & 2 & 2 Plan is higher (1,300-1,500 kcal/day), weight loss on the 5 & 2 & 2 Plan was less than that observed in extremely obese individuals following a physician-supervised very-low or low energy diet using meal replacements (usually<1,000 kcal/day) in conjunction with behavioral therapy [22].
In addition to measuring changes in total body weight, changes in body composition were also assessed. While there were significant reductions in both lean and fat mass, the majority (68-80%) of weight loss came from fat mass. Considering that lean mass accounted for an average of 20 to 33% of the weight reduction in this study, this program performed well compared to typical losses of 36% to 40% for men and 31% to 33% for women [28-31]. The retention of lean mass may be partially attributed to the extremely obese population studied, since large baseline fat mass appears to help spare lean mass loss during caloric deficit [29]. However, it may also be linked to the macronutrient composition of the meal plan which provides approximately 135-175 g of high quality protein per day. With the group’s mean baseline weight of 149 kg, this translates to an estimated 0.9 to 1.2 g protein per kg body weight, thus exceeding the average daily requirement of 0.8 g/kg and providing levels similar to those considered ideal for muscle retention during weight loss [32]. Likewise, preservation of lean mass has been observed in previous studies with mixed populations of overweight and obese individuals using other Medifast weight loss meal plans (the 5 & 1 Plan® and the 4 & 2 & 1 Plan™ which also provide >100 g of protein/day), also suggesting that the combination of meal replacements and conventional foods recommended in these programs is beneficial for preserving lean mass during weight loss [14-17,33]. While increases in activity and exercise are encouraged, none of the previously mentioned Medifast programs included a structured exercise regimen, further underscoring the likelihood that the macronutrient composition contributes to the observed preservation of lean muscle mass.
The achievement of clinically meaningful weight loss was reflected in the concomintant improvements in blood pressure. A substantial proportion of individuals (between 42% and 61% depending on whether assessed by self-report or measured) had high blood pressure at baseline, and the mean baseline systolic and diastolic blood pressure of the group (139.8 and 89.9 mm Hg, respectively) was nearly in the hypertensive range. However, at the 12-week primary endpoint, the mean systolic and diastolic blood pressures (116.9 and 79.5 mm Hg, respectively) fell into the normal range. The magnitude of the reduction in blood pressure (-17.7 mm Hg systolic and -8.4 mm Hg diastolic at 12 weeks) is clinically important and is large enough to have a significant impact on cardiovascular disease risk [34]. In fact, over 40% of individuals experienced an improvement in their blood pressure category at 12 and 24 weeks while on the 5 & 2 & 2 Plan. The apparent increase in the mean systolic blood pressure at 24 weeks appeared to have a basis in the small sample size rather than being representive of a true trend. Also of note with regard to cardiometabolic risk is the significant reduction (9.0 cm) in waist circumference at 12 weeks. This measure is an indicator of visceral fat which is linked to diabetes and cardiovascular risk [35]. The reduction in waist circumference was directionally larger (-10.0 cm), but not significant at 24 weeks (p=0.109), a consequence of the small sample size.
Weight maintenance data in this study were limited by the small number of individuals (11%, n=7) who entered this phase. The available data indicate a non-significant trend toward partial weight regain (3.4% of baseline body weight), yet the overall mean weight change during the entire program (mean of 61 weeks over the Weight loss, Transition (as applicable) and Maintenance Phases) was significant (-34 kg, or 24% below the group’s baseline weight), suggesting the program was effective for weight loss and weight maintenance for the limited subset that entered the Maintenance phase.
All signs, symptoms and health-related incidents were collected and analyzed in a systematic fashion. When evaluated, these incidents were consistent with previously reported side effects [15], primarily constipation, and general complaints of hunger, cravings, and stress which are also often associated with intentional weight loss. Both obesity and weight loss are known to be significant risk factors for the development of gallstones [36,37], and one person (1.6%) experienced gallbladder pain/stones, ultimately requiring surgery. Aside from this cholecystectomy, only one other serious (unrelated) incident was reported. The observed rate of serious events (3.2%) is similar to placebo rates seen in pharmacotherapy trials with obese populations [38-40]. Overall, the data suggest the 5 & 2 & 2 Plan was generally well-tolerated in this population of obese individuals.
One limitation of this study was the relatively small starting sample size (n=62) combined with attrition from the meal plan which, together, resulted in limited datasets, particularly at the later time points and for many of the secondary outcomes. Data at the primary endpoint, change in bodyweight at 12 weeks, was not available for approximately 40% of the starting study population. To address these concerns, tripartite analyses (completers, ITT and regression analyses) for body weight changes were conducted, the latter two of which accounted for missing data. All three analyses showed similar effectiveness, albeit with the greatest weight loss in the completer’s analysis. As previously noted, many individuals switched to other Medifast weight loss plans over time, and a portion of the difference between the completers and ITT results can be attributed to the large proportion (29%) of individuals, who as their BMI dropped to below 40 kg/m2, switched, per center procedure, to other Medifast weight loss plan(s), and continued to lose weight. These were among the most successful individuals on the 5 & 2 & 2 Plan, thus reducing the overall observed weight loss at the later time points in the ITT analysis. Retention on the 5 & 2 & 2 Plan at 12 weeks (60%) was in the range observed with some other commercial weight loss programs (approximately 45-70%) , but was better if one considers the total proportion that continued on any of the Medifast weight loss plans at 12 weeks (n=48, 77%) [22,41]. Another limitation was the retrospective nature of the study and absence of a control group. A larger, prospective, randomized, controlled trial could strengthen these results and address efficacy in a broader population. Nonetheless, the results are of interest since they are a true representation of the program effectiveness in real clients in the weight loss centers.
In conclusion, the 5 & 2 & 2 Plan administered at MWCCs appeared to be well-tolerated and resulted in clinically meaningful weight loss in a majority of the study population, which consisted primarily of extremely obese individuals. Concomitant improvements in cardiometabolic risk factors were also observed. Based on these results, the 5 & 2 & 2 Plan represents a viable first line treatment option for individuals with extreme obesity and may have utility prior to advocating more intensive treatments, such as pharmacotherapy or bariatric surgery. Given the need for an accompanying lifestyle intervention, this program could also be an effective pairing for use with other treatment modalities.
References
- Ogden CL, Carroll MD, Kit BK, Flegal KM (2014) Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA 311: 806-814.
- An R (2014) Prevalence and Trends of Adult Obesity in the US, 1999-2012. ISRN Obes 2014: 185132.
- Sturm R, Hattori A (2013) Morbid obesity rates continue to rise rapidly in the United States. Int J Obes (Lond) 37: 889-891.
- Twells LK, Gregory DM, Reddigan J, Midodzi WK3 (2014) Current and predicted prevalence of obesity in Canada: a trend analysis. CMAJ Open 2: E18-26.
- Thomas DM, Weedermann M, Fuemmeler BF, Martin CK, Dhurandhar NV, et al. (2014) Dynamic model predicting overweight, obesity, and extreme obesity prevalence trends. Obesity (Silver Spring) 22: 590-597.
- Fryar CD, Carroll MD, Ogden CL (2014) Prevalence of overweight, obesity, and extreme obesity among adults: United States, 1960–1962 through 2011–2012.
- National Heart Lung and Blood Institute, National Institute of Diabetes and Digestive and Kidney Diseases (U.S.) (1998) Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults : the evidence report. National Institutes of Health, National Heart, Lung, and Blood Institute, Bethesda, MD.
- Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, et al. (2013) 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Obesity Society. Circulation 128: 1-69.
- Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, et al. (2014) 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 129: S102-138.
- Kitahara CM, Flint AJ, Berrington de Gonzalez A, Bernstein L, Brotzman M, et al. (2014) Association between class III obesity (BMI of 40-59 kg/m2) and mortality: a pooled analysis of 20 prospective studies. PLoS Med 11: e1001673.
- Jagielski AC, Brown A, Hosseini-Araghi M, Thomas GN, Taheri S (2014) The association between adiposity, mental well-being, and quality of life in extreme obesity. PLoS One 9: e92859.
- Apovian CM, Aronne LJ, Bessesen DH, McDonnell ME, Murad MH, et al. (2015) Pharmacological management of obesity: an endocrine Society clinical practice guideline. J ClinEndocrinolMetab 100: 342-362.
- Blackburn GL, Wollner S, Heymsfield SB (2010) Lifestyle interventions for the treatment of class III obesity: a primary target for nutrition medicine in the obesity epidemic. Am J ClinNutr 91: 289S-292S.
- Unick JL, Beavers D, Bond DS, Clark JM, Jakicic JM, et al. (2013) The long-term effectiveness of a lifestyle intervention in severely obese individuals. Am J Med 126: 236-2, 242.
- Davis LM, Coleman C, Kiel J, Rampolla J, Hutchisen T, et al. (2010) Efficacy of a meal replacement diet plan compared to a food-based diet plan after a period of weight loss and weight maintenance: a randomized controlled trial. Nutr J 9: 11.
- Coleman C, Kiel J, Hanlon-Mitola A, Sonzone C, Fuller N, et al. (2012) Use of the Medifast meal replacement program for weight loss in overweight and obese clients: a retrospective chart review of three Medifast Weight Control Centers (MWCC). Food and Nutrition Sciences 03: 1433-1444.
- Shikany JM, Thomas AS, Beasley TM, Lewis CE, Allison DB (2013) Randomized controlled trial of the Medifast 5 & 1 Plan for weight loss. Int J Obes (Lond) 37: 1571-1578.
- Coleman CC, Kiel JR, Mitola AH, Langford JS, Davis KN, et al. (2015) Effectiveness of a Medifast meal replacments program on weight, body composition and cardiometabolic risk factors in overweight and obese adults: a multicenter systematic retrospective chart review study. Nutrition Journal.
- Heymsfield SB, van Mierlo CA, van der Knaap HC, Heo M, Frier HI (2003) Weight management using a meal replacement strategy: meta and pooling analysis from six studies. Int J ObesRelatMetabDisord 27: 537-549.
- Heymsfield SB (2010) Meal replacements and energy balance. PhysiolBehav 100: 90-94.
- Ashley JM, St Jeor ST, Perumean-Chaney S, Schrage J, Bovee V (2001) Meal replacements in weight intervention. Obes Res 9 Suppl 4: 312S-320S.
- Hannum SM, Carson L, Evans EM, Canene KA, Petr EL, et al. (2004) Use of portion-controlled entrees enhances weight loss in women. Obes Res 12: 538-546.
- Anderson JW, Grant L, Gotthelf L, Stifler LT (2007) Weight loss and long-term follow-up of severely obese individuals treated with an intense behavioral program. Int J Obes (Lond) 31: 488-493.
- Baillot A, Romain AJ, Boisvert-Vigneault K, Audet M, Baillargeon JP, et al. (2015) Effects of lifestyle interventions that include a physical activity component in class II and III obese individuals: a systematic review and meta-analysis. PLoS One 10: e0119017.
- Vassar M, Holzmann M (2013) The retrospective chart review: important methodological considerations. J EducEval Health Prof 10: 12.
- Karelis AD, Chamberland G, Aubertin-Leheudre M, Duval C; Ecological mobility in Aging and Parkinson (EMAP) group (2013) Validation of a portable bioelectrical impedance analyzer for the assessment of body composition. ApplPhysiolNutrMetab 38: 27-32.
- Cooper JN, Columbus ML, Shields KJ, Asubonteng J, Meyer ML, et al. (2012) Effects of an intensive behavioral weight loss intervention consisting of caloric restriction with or without physical activity on common carotid artery remodeling in severely obese adults. Metabolism 61: 1589-1597.
- Goodpaster BH, Delany JP, Otto AD, Kuller L, Vockley J, et al. (2010) Effects of diet and physical activity interventions on weight loss and cardiometabolic risk factors in severely obese adults: a randomized trial. JAMA 304: 1795-1802.
- Dixon JB, Lambert EA, Grima M, Rice T, Lambert GW, et al. (2015) Fat-free mass loss generated with weight loss in overweight and obese adults: What may we expect? Diabetes ObesMetab 17: 91-93.
- Heymsfield SB, Gonzalez MC, Shen W, Redman L, Thomas D (2014) Weight loss composition is one-fourth fat-free mass: a critical review and critique of this widely cited rule. Obes Rev 15: 310-321.
- Chaston TB, Dixon JB, O'Brien PE (2007) Changes in fat-free mass during significant weight loss: a systematic review. Int J Obes (Lond) 31: 743-750.
- Tang M, Leidy HJ, Campbell WW (2013) Regional, but not total, body composition changes in overweight and obese adults consuming a higher protein, energy-restricted diet are sex specific. Nutr Res 33: 629-635.
- Krieger JW, Sitren HS, Daniels MJ, Langkamp-Henken B (2006) Effects of variation in protein and carbohydrate intake on body mass and composition during energy restriction: a meta-regression 1. Am J ClinNutr 83: 260-274.
- Beavers KM, Gordon MM, Easter L, Beavers DP, Hairston KG, et al. (2015) Effect of protein source during weight loss on body composition, cardiometabolic risk and physical performance in abdominally obese, older adults: a pilot feeding study. J Nutr Health Aging 19: 87-95.
- McInnes GT (2005) Lowering blood pressure for cardiovascular risk reduction. J HypertensSuppl 23: S3-8.
- Gaggini M, Saponaro C, Gastaldelli A (2015) Not all fats are created equal: adipose vs. ectopic fat, implication in cardiometabolic diseases. HormMolBiolClinInvestig 22: 7-18.
- Heshka S, Heymsfield S (2001) Obesity and gallstones. International Textbook of Obesity, Wiley, Chichester 399-409.
- Njeze GE (2013) Gallstones. Niger J Surg 19: 49-55.
- Smith SR, Weissman NJ, Anderson CM, Sanchez M, Chuang E, et al. (2010) Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med 363: 245-256.
- Gadde KM, Allison DB, Ryan DH, Peterson CA, Troupin B, et al. (2011) Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet 377: 1341-1352.
- Smith SR, Prosser WA, Donahue DJ, Morgan ME, Anderson CM, et al. (2009) Lorcaserin (APD356), a selective 5-HT(2C) agonist, reduces body weight in obese men and women. Obesity (Silver Spring) 17: 494-503.
- Finley CE, Barlow CE, Greenway FL, Rock CL, Rolls BJ, et al. (2007) Retention rates and weight loss in a commercial weight loss program. Int J Obes (Lond) 31: 292-298.
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Android Obesity
- Anti Obesity Medication
- Bariatric Surgery
- Best Ways to Lose Weight
- Body Mass Index (BMI)
- Child Obesity Statistics
- Comorbidities of Obesity
- Diabetes and Obesity
- Diabetic Diet
- Diet
- Etiology of Obesity
- Exogenous Obesity
- Fat Burning Foods
- Gastric By-pass Surgery
- Genetics of Obesity
- Global Obesity Statistics
- Gynoid Obesity
- Junk Food and Childhood Obesity
- Obesity
- Obesity and Cancer
- Obesity and Nutrition
- Obesity and Sleep Apnea
- Obesity Complications
- Obesity in Pregnancy
- Obesity in United States
- Visceral Obesity
- Weight Loss
- Weight Loss Clinics
- Weight Loss Supplements
- Weight Management Programs
Recommended Journals
Article Tools
Article Usage
- Total views: 16571
- [From(publication date):
specialissue-2015 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 11961
- PDF downloads : 4610
