Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
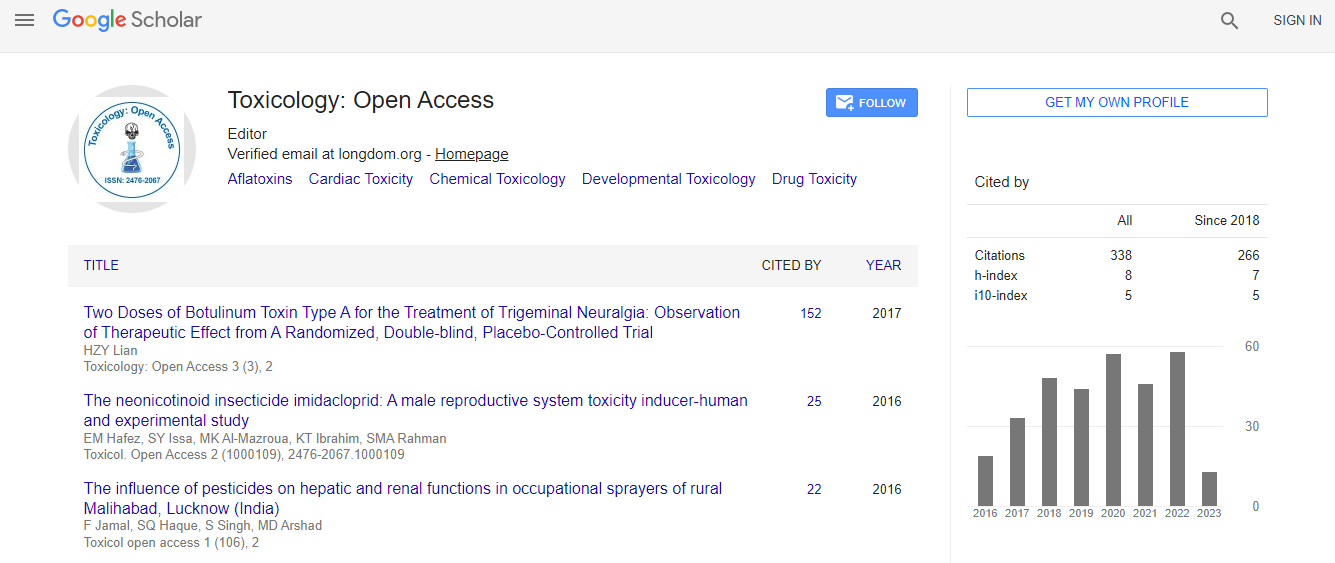
Google Scholar citation report
Citations : 336
Toxicology: Open Access received 336 citations as per Google Scholar report
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Related Subjects
Share This Page
Baclofen systemic toxicity: Experimental histopathological and biochemical study
8th World Congress on Toxicology and Pharmacology
Sahar Y Issa, Essam M Hafez, Asmaa S El-Banna, Safaa M Abdel Rahman, Maha K AlMazroua and Mostafa Abo El-Hamd
Alexandria University Faculty of Medicine, Egypt Minia University, Egypt Central Laboratory, Egypt Dammam Poison Control Center, KSA
ScientificTracks Abstracts: Toxicol Open Access
Abstract
The study was performed on 30 healthy adult male Albino rats divided into four groups with five rats in each control group, and ten rats in either experimental groups (two experimental and two control groups). Five rats (negative control) were kept in a quite non-stressful environment, provided with food ad libitum and free access to water. Normal saline (1 ml) was given orally as placebo in the positive control group (n=5). Experimental group III, Baclofen acute toxicity group (10 rats): Each animal received a single dose of LD50 of Baclofen orally by gavage. It equals 145 mg/kg b wt. The rats were observed for acute toxicity manifestations as well as for LD50 deaths. Group IV, (Baclofen dependent group 10 rats): Each animal received Baclofen (1/10th LD50) in gradually increasing doses for one month. The levels of blood urea nitrogen (BUN), creatinine kinase (CK), alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GGT), cardiac troponin I (CTnl), prothrombin time (PT), in both Baclofen treated groups showed significant elevation when compared to controls. There were brain, lung, gastric, hepatic, and renal histopathological changes in Baclofen treated rats whose severity varied between the two experimental groups.Biography
Sahar Y Issa has completed her Doctorate degree in Clinical Toxicology & Forensic Medicine in 2008, from Faculty of Medicine, Alexandria University, Egypt and is a Lecturer of Clinical Toxicology & Forensic Medicine in the same University. She is currently a Consultant Toxicologist, and the Medical Director, supervising Emergency Toxicology, Molecular Toxicology, & Therapeutic Drug Monitoring units in Dammam Poison Control Center, MOH - Saudi Arabia. She has published more than 25 papers in reputed journals and serving as an Editorial Board Member of repute.
Email: sahar_issa71@yahoo.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi