Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
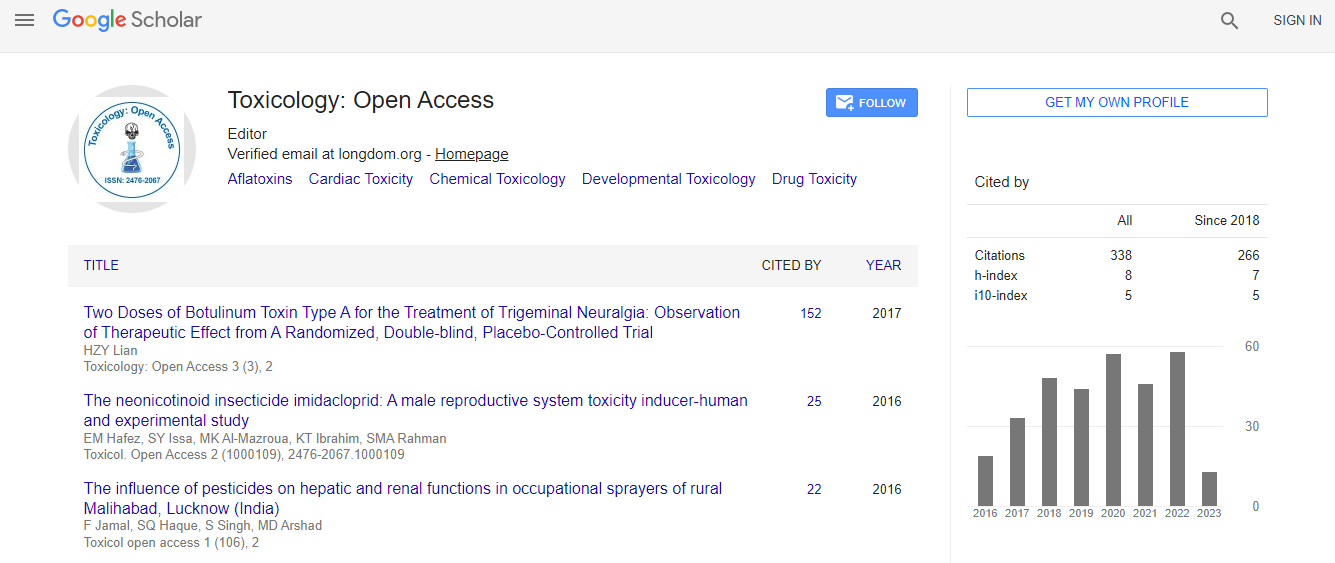
Google Scholar citation report
Citations : 336
Toxicology: Open Access received 336 citations as per Google Scholar report
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Related Subjects
Share This Page
Clinical case study: Toxicity of dietary supplement used for weight reduction contaminated with adulterants
8th World Congress on Toxicology and Pharmacology
Sahar Y Issa, Maha K Al-Mazroua, Essam M Hafez, Mohammed G Saad, Asmaa S El-Banna and Heba A Hussain
Dammam Poison Control Center, KSA Alexandria University Faculty of Medicine, Egypt Minia University, Egypt King Fahad Specialist Hospital-Dammam, Saudi Arabia
ScientificTracks Abstracts: Toxicol Open Access
Abstract
Nowadays, dietary supplements��? consumption, especially those of plant origin, has been gaining more popularity among consumers owing to misbelieve that they are natural products posing no risks to human health. In many regions of the world including the European Union and the United States, dietary supplements are legally considered as special categories of food, thus are not popularly being submitted to any safety assessment prior to their commercialization. Among the safety issues, comes adulteration by the illegal addition of pharmaceutical substances or their analogues, since unscrupulous producers can falsify these products to provide for quick effects and to increase their profits and sales. This case study is about one product used locally as a dietary supplement, and was marketed for weight loss, muscle building, lead to several health complications in one user, who presented with renal impairment and also describes about several conventional and advanced analytical techniques used to detect and identify amphetamine-like adulterants in the dietary supplement.Biography
Sahar Y Issa has completed her Doctorate degree in Clinical Toxicology & Forensic Medicine in 2008, from Faculty of Medicine, Alexandria University, Egypt and is a Lecturer of Clinical Toxicology & Forensic Medicine in the same university. She is currently a Consultant Toxicologist, and the Medical Director, supervising Emergency Toxicology, Molecular Toxicology and Therapeutic Drug Monitoring units in Dammam Poison Control Center, MOH - Saudi Arabia. She has published more than 25 papers in reputed journals and serving as an Editorial Board Member of repute.
Email: sahar_issa71@yahoo.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi