Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
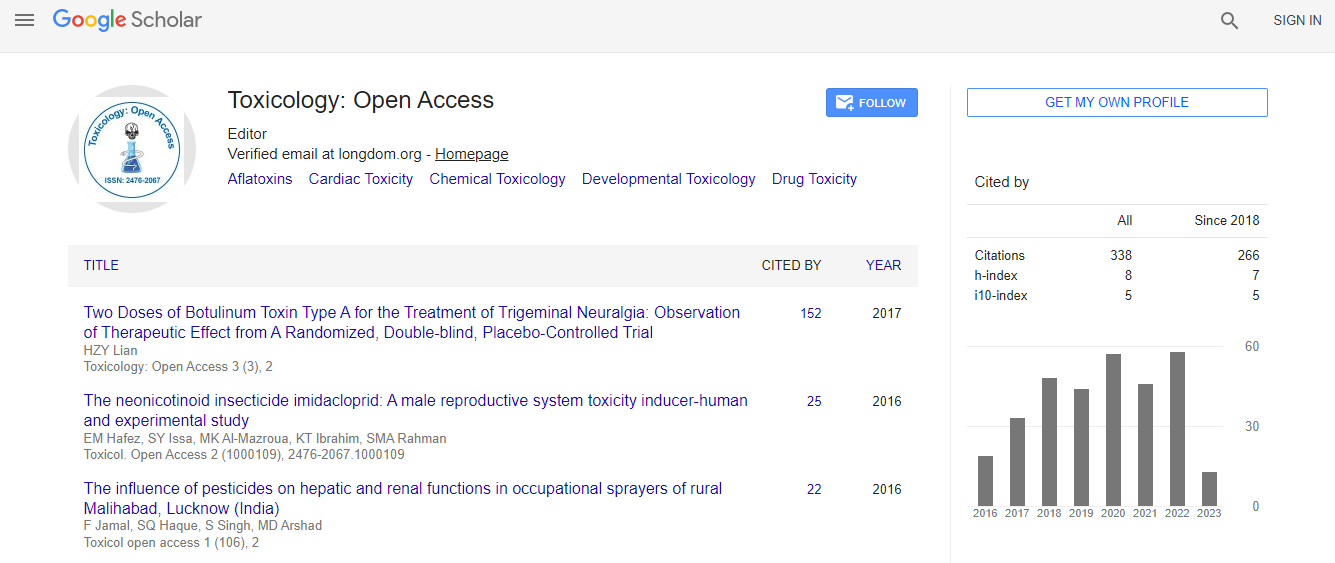
Google Scholar citation report
Citations : 336
Toxicology: Open Access received 336 citations as per Google Scholar report
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Related Subjects
Share This Page
DNase/endonuclease network: A new target for mitigation of acute nephrotoxicity
20th World Congress on Toxicology and Pharmacology
Alexei G Basnakian
University of Arkansas for Medical Sciences, USA
Keynote: Toxicol Open Access
Abstract
Deoxyribonucleases (DNases) universally induce irreversible cell death by fragmenting DNA in response to cell injury. All of the nine known cell death DNases are endonucleases. Despite that most of the DNase activity is used after cell death, a genetic inactivation of DNases provide protection of cells and tissues against DNA breaks induced by cytotoxic stimuli and partially protect against tissue injury. Therefore, DNases act before the point-of-no-return in cell death and can be potentially used as therapeutic targets for tissue protection against injury. Our studies identified two DNases, DNase I and EndoG, as being responsible for tubular epithelial toxicity during acute kidney injury induced by Cisplatin, rhabdomyolysis or ischemia. However, inhibitors of DNases are not available. The aim of this study was to identify DNase inhibitors, which might be used for mitigation of acute kidney injury. To identify DNase inhibitors, we have developed a high-throughput screening assay based on a proprietary fluorescent probe. This assay allowed the identification of several new inhibitors of deoxyribonuclease I (DNase I), which were also active against two other DNases, endonucleases G (EndoG) and Deoxyribonuclease II (DNase II). The DNase inhibitors were able to significantly protect kidney tubular epithelial cells in vitro and mouse kidneys in vivo against acute kidney toxicity induced by Cisplatin, glycerol (rhabdomyolysis), or renal ischemia-reperfusion. The inhibitors showed no toxicity in vivo at 5x therapeutic doses. DNases can be used as a therapeutic target for mitigation of toxic or hypoxic acute kidney injury. The identified DNase inhibitors or similar compounds have a great potential for tissue protection against toxic kidney failure. Considering that DNases are expressed and responsible for cell death in all tested cells, tissues and animal models, it is likely that the same compounds may be used for universal tissue protection against various injuries.Biography
Alexei G Basnakian has completed his PhD and DSc degrees from the Russian Academy of Medical Science, both in the field of DNA-degrading enzymes. He had Post-doctoral trainings in Molecular Biology at the Harvard Medical School and in Toxicology/Cancer Research at the National Center for Toxicological Research/U.S. Food and Drug Administration. He is a tenured Professor at the Department of Pharmacology and Toxicology and also Director of the DNA Damage and Toxicology Core Center at the University of Arkansas for Medical Sciences and Research Career Scientist at the Veterans Hospital in Little Rock, USA. He is the author of 87 peer-reviewed papers and 14 reviews or book chapters. He is an Editorial Board Member of four biomedical journals and also a Member of NIH, AHA and VA grant study sections. His research interests are in DNases/endonucleases and DNA damage associated with toxicity, anti-cancer therapy, tissue injury and cell death.
E-mail: basnakianalexeig@uams.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi