Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
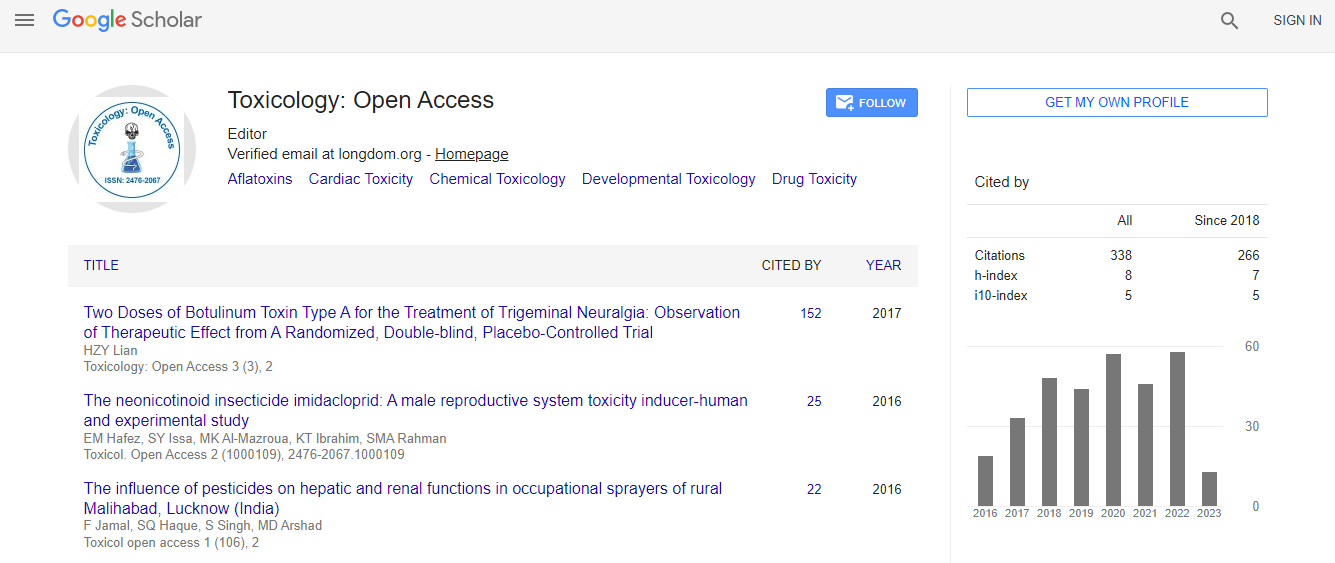
Google Scholar citation report
Citations : 336
Toxicology: Open Access received 336 citations as per Google Scholar report
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Related Subjects
Share This Page
Evaluation of toxic contents and metals in different cosmetics available in Arabian market
8th World Congress on Toxicology and Pharmacology
Sahar Y Issa
Alexandria University Faculty of Medicine, Egypt
Posters & Accepted Abstracts: Toxicol Open Access
Abstract
The definition of a cosmetic identifies the site of application (epidermis, hair system, nails, lips, eyes) and the intended functions (cleaning, perfuming, changing the appearance, correcting body odours, protecting and, keeping in good condition). Cosmetic formulations are that class of products whose atypical features can be well thought-out, from a biochemical and toxicological point of view, as intermediary between both foods and drugs. For undeniably, the frequency of use of cosmetic formulations is generally scheduled on a daily basis, and in some instances several cosmetic products, like, for example, a lipstick or a hand cream, can be applied to the body twice or more times a day. Simultaneously, the techniques that are generally used in the production of a great variety of cosmetic formulations are directly derived from the experience of pharmaceutics. During the past decades the safety of cosmetic products and their ingredients has attracted greater than ever attention; thus their toxicological safety evaluation is a relatively young discipline, which evolved in the second half of the 20th century. Up to the 1960s it was commonly believed that cosmetic products will never go beyond the surface of the human body. Therefore, local effects were the primary safety concern. The first consistent in vivo tests for skin and eye irritation were developed in the 1940s. Oral exposure can occur from wearing of cosmetic products containing heavy metal impurities around the mouth and also from hand to mouth contact.Biography
Sahar Y Issa has completed her Doctorate degree in Clinical Toxicology & Forensic Medicine in 2008, from Faculty of Medicine, Alexandria University, Egypt and is a Lecturer of Clinical Toxicology & Forensic Medicine in the same university. She is currently a Consultant Toxicologist, and the Medical Director, supervising Emergency Toxicology, Molecular Toxicology and Therapeutic Drug Monitoring units in Dammam Poison Control Center, MOH - Saudi Arabia. She has published more than 25 papers in reputed journals and serving as an Editorial Board Member of repute.
Email: sahar_issa71@yahoo.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi