Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
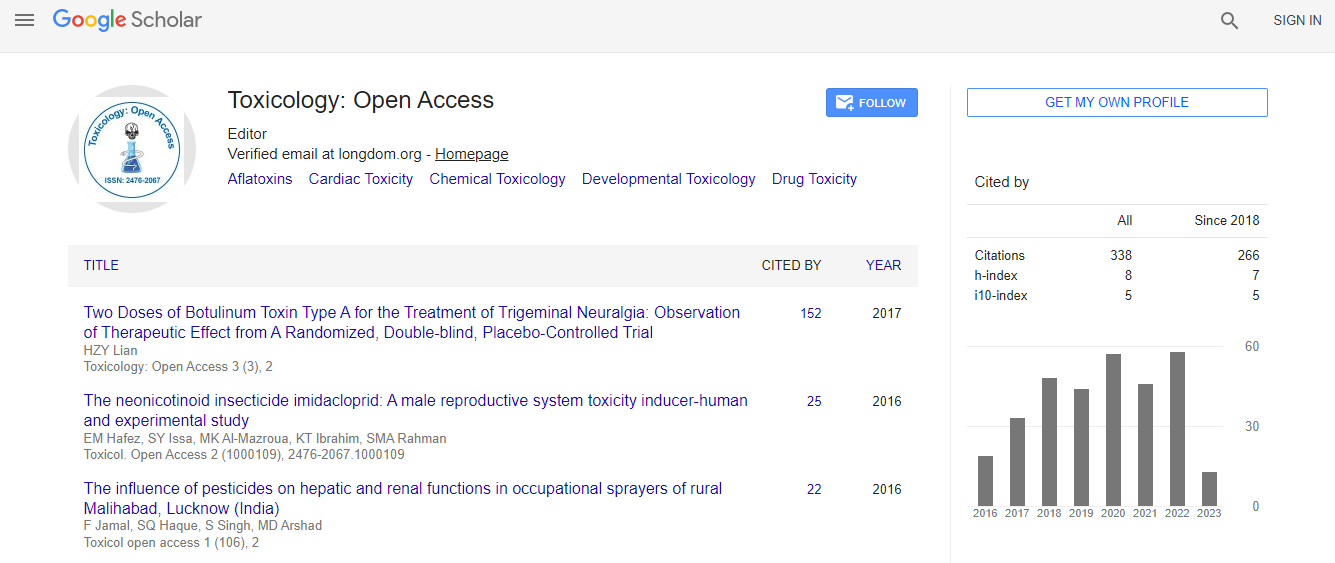
Google Scholar citation report
Citations : 336
Toxicology: Open Access received 336 citations as per Google Scholar report
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Related Subjects
Share This Page
Synthetic bacteriophages as nanoparticles for intravenous administration of targeted gene therapy for cancer
14th World Congress on Toxicology and Pharmacology
Keith Potent, Armand Sinclair
Monash University, Australia Novother Cancer Research, New Zealand
ScientificTracks Abstracts: Toxicol Open Access
Abstract
We present a first in human case of a 50 year-old patient with end-stage metastatic ovarian cancer infused with a novel, intravenously administered, synthetically engineered bacteriophage-based gene therapy (Metavec) for metastatic solid malignancies. Compared to mammalian virus-based delivery vehicles, bacteriophage-based vectors bring many preferable features for treatment in humans. Their genomes have been extensively sequenced and, with modern technologies, they are relatively malleable allowing them to be extensively modified. Unlike mammalian viruses, bacteriophages are not natural pathogens to humans yet their capsid can have equivocal cargo carrying capacity. To the authors��? best knowledge, no other bacteriophage-based applications have succeeded with intravenous administration. This advance in nanotechnology and novel approach could revolutionize medical care. The patient we discuss received a dose-escalating regime up to 1x1011 particles per dose, three times a week for three weeks. The infusions were very well tolerated. Symptoms include nausea, low-grade fever, and also discomfort in areas where larger tumors were present. Post-infusion investigations included serum biochemistry, serum tumor markers, and computed tomography. The paradigm shift, results, and discussion will be presented.Biography
Keith Potent is currently a PhD candidate in Translational Research at Monash University. After completing undergraduate degrees in Mathematics and Chemistry, Dr Potent has completed his medical degree. He is a practicing doctor in Queensland.
Email:keith_potent@hotmail.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi