Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
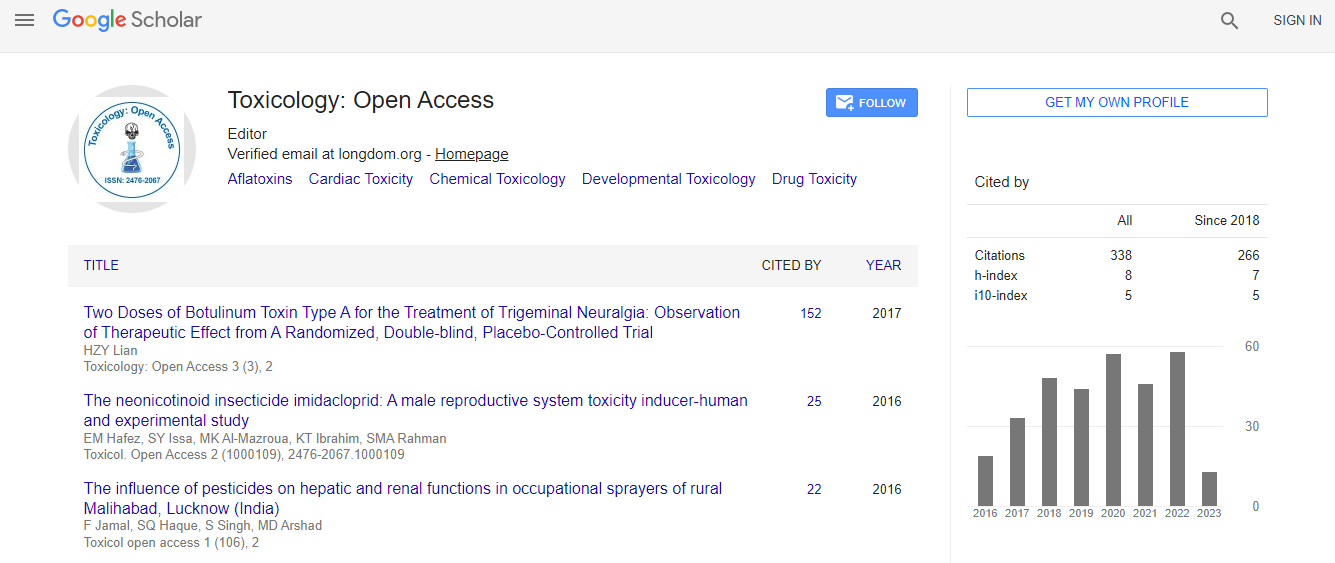
Google Scholar citation report
Citations : 336
Toxicology: Open Access received 336 citations as per Google Scholar report
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Related Subjects
Share This Page
Tissue distribution of Suvorexant in three postmortem cases
14th World Congress on Toxicology and Pharmacology
Brian Waters, Kenji Hara, Natsuki Ikematsu, Mio Takayama, Aya Matsusue, Masayuki Kashiwagi and Shin-ichi Kubo
Fukuoka University, Japan
ScientificTracks Abstracts: Toxicol Open Access
Abstract
Statement of the Problem: Suvorexant (Belsomra�?®) is a relatively new insomnia medication that has been available in the US and Japan since 2014. It is a dual orexin receptor antagonist that promotes sleep by inhibiting the binding of orexin neurons to the OX1R and OX2R receptors. In this report, we describe the detection and quantitation of Suvorexant from the postmortem specimens of three recent autopsy cases handled by our department. Methodology & Theoretical Orientation: Suvorexant was identified by fast GC-MS during routine screening and quantitated by a fully validated LC-MS/ MS method. Quantitation was achieved by positive electrospray ionization in the selected reaction monitoring mode. Monitored transitions were m/z 451>186 for quantitation and m/z 451>104 for qualification. Diazepam-d5 was used as an internal standard. Findings: Suvorexant was detected and quantitated in the body fluids and tissues of three autopsy cases. The specimens included cardiac blood, peripheral blood, urine, liver, kidney, spleen, pancreas, lung, muscle, fat and cerebrospinal fluid. Tissue distribution across the three cases will be presented and discussed. Conclusion & Significance: The use of Suvorexant as an insomnia medication has recently increased around the world. To our knowledge this is the first instance of Suvorexant being quantitated from actual autopsy cases. It is possible the presence of this medication in clinical and forensic samples has been missed due to its high boiling point and thus late elution in gas chromatography. We were able to detect Suvorexant in three cases by using fast GC-MS which significantly reduced its retention time. It is likely that this compound will be encountered more often by the forensic and clinical toxicology communities going forward. Recent Publications 1. Hara K, Waters B, Ikematsu N, et al. (2016) Development of a preparation method to produce a single sample that can be applied to both LCâ�?�?MS/MS and GCâ�?�?MS for the screening of postmortem specimens. Legal Medicine; 21: 85-92. 2. Waters B, Ikematsu N, Hara K, et al. (2016) GC-PCI-MS/MS and LC-ESI-MS/MS databases for the detection of 104 psychotropic compounds. Legal Medicine; 20: 1-7. References 1. Carson M, Kerrigan S (2017) Quantification of Suvorexant in urine using gas chromatography/mass spectrometry. Journal of Chromatography B; 1040: 289-294. 2. Iqbal M, Ezzeldin E, Khalil N, Al-Rashood S, Al-Rashood K (2017) Simple and Highly Sensitive UPLC-ESI-MS/MS Assay for Rapid Determination of Suvorexant in Plasma. Journal of Analytical Toxicology; 41: 114-120.Biography
Brian Waters has received his Master of Science degree in Criminalistics from California State University Los Angeles, USA. After working as a Criminalist for the County of Los Angeles, Department of Coroner/Medical Examiner for almost eight years, he accepted a position as an Assistant Professor in the Department of Forensic Medicine at Fukuoka University in Japan. His specialty is postmortem forensic toxicology and he has published academic papers on fast gas chromatography-mass spectrometry, the analysis of novel psychoactive compounds, preparation methods for postmortem samples and the analysis of volatile hydrocarbons in blood.
Email:bwaters@fukuoka-u.ac.jp

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi