Commentary Open Access
A Rare Case of Erythromelalgia in a Young Male Patient
Raj N1*, Malik AS2 and Waters B11Department of Rheumatology, Larkin Hospital, Nova Southeastern University, Miami, Florida, USA
2Department of Physical Medicine & Rehabilitation, Larkin Hospital, Nova Southeastern University, Miami, Florida, USA
- *Corresponding Author:
- Raj N
Department of Rheumatology, Larkin Hospital
Nova Southeastern University, Miami, Florida, USA
Tel: +954 433 8112
E-mail: ntr203@gmail.com, nraj81@yahoo.com
Received date:June 12, 2014; Accepted date: August 28, 2014; Published date: Septeber 02, 2014
Citation: Raj N, Malik AS, Waters B (2014) A Rare Case of Erythromelalgia in a Young Male Patient. Microinflammation 1:107. doi: 10.4172/2381-8727.1000107
Copyright:2014 Raj N et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at International Journal of Inflammation, Cancer and Integrative Therapy
Abstract
Erythromelalgia is a rare disorder characterized by redness, warmth, and intense burning pain in the distal extremities, most commonly in the legs and feet. This neuropathic pain results from nociceptive hyperexcitability in peripheral nerve fibers. The condition can be primary or secondary to a variety of disease conditions. A successful treatment has so far been elusive, and patients often have a high degree of morbidity. This case report presents a young male patient, who has been diagnosed with erythromelalgia which is believed to be secondary to his Type 1 Diabetes Mellitus. This case has diagnostic value for clinicians who have not seen this disorder.
Case Presentation
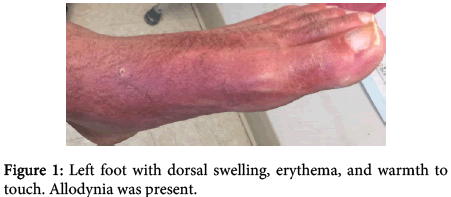
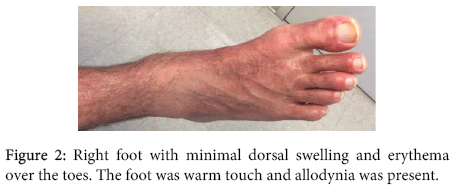
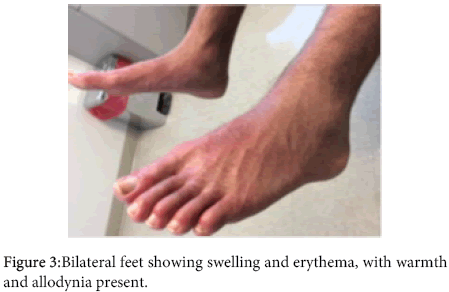
A 30 year old Caucasian male presented to our clinic with redness, warmth, swelling, and burning pain in his bilateral lower legs and feet. The pain was so intense he needed to ambulate with a walker, and at times even light touch elicited debilitating pain. This began gradually two years ago, initially with a slight redness over his shins. Six months ago, he was diagnosed with Type 1 Diabetes Mellitus. One month after his diagnosis, the symptoms in his lower extremities worsened. The patient had already been to a number of specialists and had not been given a definite diagnosis. The mother had brought the patient to our outpatient rheumatology clinic as a last resort. On physical exam, vital signs were normal. He had bilateral lower extremity redness starting from mid-calf and extending to the dorsal feet, but sparing the plantar side (Figures 1-3). Allodynia was noted to the dorsum of both feet. Trace pitting edema was noted to the dorsum of both feet, and there was edema to all toes. Temperature strip to the dorsum of the left food was 34°C, and the right foot was 33°C. Motor strength to the lower extremities was 5/5 bilaterally. Sensory examination of the lower extremities was normal to pinprick bilaterally. Knee jerk reflexes were 2+ bilaterally. Ankle jerk reflex was absent bilaterally. The rest of the physical exam was normal. His only medical history was Type 1 diabetes. He had no surgical history. Social history was negative for tobacco and alcohol use. His medications included subcutaneous insulin, gabapentin 3600 mg daily (600 mg, 6 pills per day), duloxetine 60 mg daily, tramadol 50 mg every 6 hours, and triamcinolone acetonide 0.1% topical cream. He had a number of studies performed prior to coming to our clinic. A previously obtained non contrast magnetic resonance imaging (MRI) study of the lumbar spine revealed a small central disk protrusion with mild thecal sac compression at L4-L5, without significant spinal canal stenosis or neuroforaminal narrowing. There was no spondylolisthesis noted. The patient also had a lower extremity angiogram, which revealed no vessel occlusion. Electromyographic (EMG) and nerve conduction velocity (NCV) testing was performed. Evaluation of the left sural sensory and right sural sensory nerve showed no response (7 cm and 14 cm, respectively). All remaining nerves were within normal limits. All F wave latencies were within normal limits, and all H reflex latencies were within normal limits. Needle evaluation of the right anterior tibialis, the right peroneus longus, the right gastrocnemius, the left anterior tibialis, the left peroneus longus, and the left gastrocnemius showed slightly increased polyphasic potentials. All remaining muscles showed no evidence of electrical instability. The results were thus consistent with mild sensorimotor polyneuropathy, primarily axonal in nature. A diagnosis of diabetic neuropathy had initially been rendered by prior clinicians. He saw a dermatologist, and underwent contact allergen patch testing, where 52 specific allergens were applied to the patient’s back 48 hours prior, and removed and interpreted. The results were grossly negative. In addition, he had a skin shave biopsy of the right and left medial foot. The dermatopathology report showed mild epidermal spongiosis with necrotic keratinocytes, and extravasated erythrocytes, with few scattered hemosiderophages. There were no features of vasculitis. PAS staining was negative for fungi.
On our evaluation, we conducted an extensive rheumatologic workup. This included getting bilateral feet, ankle, and tibia/fibula x rays, which were normal. Pertinent rheumatology labs tested included a negative ANA by immunofluorescence as well as by cascading reflex. A negative Anti-topoisomerase antibody (Anti Scl-70), negative double stranded DNA antibody (Anti-Ds DNA), negative PM-ScL antibody, normal complement levels, normal rheumatoid factor, and negative Sjogren’s antibodies (SSA, SSB) were also noted. A celiac disease panel was negative, and serum immunofixation revealed a normal pattern. An infectious disease workup was done, which included tests for HIV, Syphilis, Lyme disease, and EBV; all these tests were negative. Upon review of all the accumulated data, a diagnosis of Erythromelalgia was made. Since a small percentage of Erythromelalgia cases can be due to underlying myeloproliferative disorders, a hematology evaluation was obtained. A JAK2 mutation analysis for thrombocytosis was negative, and blood counts and erythropoietin level were normal. The pathophysiology for Erythomelalgia was determined to be secondary to his diabetes. His current regimen of oral pain and neuro-modulating agents has provided little relief. He tried aspirin, which actually made the pain worse. A sympathetic stellate ganglion block was also performed, which only mildly reduced his pain. He was finally prescribed the class 1B antiarrhythmic mexiletine 150 mg daily, subsequently increased to 150mg twice daily, which has also mildly reduced his pain and swelling.
Discussion
The term erythromelalgia was first used in 1878 to describe a syndrome involving redness, warmth, and burning pain in the extremities [1]. It is a rare condition, and many cases are misdiagnosed. As such, reports vary regarding the exact incidence and prevalence. Based on a retrospective review of 32 cases over 37 years, the incidence of erythromelalgia was proposed to be 1.3 per 100,000 persons per year [2]. A larger study, which involved 168 patients over 14 years, proposed an incidence of 2.5 to 3.3 cases per million persons per year [3,4]. Secondary erythromelalgia was reported to be more common than primary erythromelalgia. Average age of presentation was reported to be 55.8 years, with an age range from 5-91 years. Symptoms were intermittent in 97% of patients, and constant in 3% of patients. Symptoms predominantly involved the feet in 88.1% of patients, and predominantly involved the hands in 25.6% of patients. Erythromelalgia has a proposed prevalence of 18 to 20 cases per million [5].
There are currently no confirmatory tests for erythromelalgia, so diagnosis is based primarily on clinical presentation. The hallmark of this condition is the triad of paroxysmal erythema, warmth, and burning pain. Character of the pain is usually described as “hot”, “burning”, or “numb.” Symptoms most commonly manifest in the feet, however they may also manifest in the hands [3,6,7]. Symptoms are usually intermittent, bilateral, and are often precipitated by exertion or exposure to warmth. Various other precipitating factors have also been reported, including insomnia, stress, caffeine, and alcohol. Episodes may last from several minutes to several months, and cooling of the affected area may alleviate the symptoms. Our patient unfortunately experienced near constant pain over many months. Primary erythromelalgia must be differentiated from secondary erythromelalgia, and an underlying cause should be sought in all new cases. The patient described in this case study presented with the classic clinical triad associated with erythromelalgia. This patient’s pain was debilitating, and was poorly controlled despite being treated with high-doses of neuropathic pain medication. MRI studies of the patient’s lumbar spine failed to reveal abnormalities that correlated with the presenting signs and symptoms. Electrodiagnostic studies revealed abnormalities consistent with mild sensorimotor polyneuropathy. Given the patient’s past medical history of type 1 diabetes mellitus, the presumptive diagnosis of diabetic neuropathy was made. This diagnosis did not, however, explain the patient’s debilitating symptoms. Dermatologic work-up also failed to reveal the etiology of the patient’s presenting complaints. After extensive rheumatological work-up, the patient was ultimately diagnosed with erythromelalgia. While primary erythromelalgia could not be completely ruled out, the patient was most likely afflicted with secondary erythromelalgia associated with type 1 diabetes mellitus.
Primary erythromelalgia, also known as Mitchell’s disease, has a sporadic or an autosomal dominant inheritance pattern, and is associated with the SCN9A gene mutation [8-10]. Work by Dib-Hajj et al. reveals the mutation of the SCN9A gene leads to defective production of a voltage gated sodium channel termed Nav1.7 [11]. This sodium channel plays a critical role in the generation and conduction of action potentials and is located within the dorsal root ganglia (DRG) and sympathetic ganglia neurons. It produces a tetrodotoxin-sensitive, fast-inactivating current and is also characterized by a slow recovery from inactivation and by a slow closed-state inactivation. This results in large responses to small, sub-threshold depolarizations. Missense mutations in Nav1.7 have been shown to produce a hyperpolarizing shift in activation and slow deactivation. These mutations cause an increase in amplitude of the current produced by Nav1.7 in response to slow, small depolarizations [11]. Defective sodium channels in the dorsal root ganglia result in hyperexcitable C-fibers on peripheral sensory neurons and decreased nociceptor threshold to painful stimulus. In a study by Harty et al., a missense mutation of an alanine 863 substitution by proline (A863P) in Nav1.7 was shown to produce hyperexcitability in DRG [12]. It is believed this hyperexcitability produces the allodynia in erythromelalgia. Paradoxically, the symptoms of redness and warmth are thought to be due to sympathetic ganglion neuron hypoexcitability, linked to the Nav1.7 mutation. Rush et al. reports on a Nav1.7 single amino acid substitution mutation, termed L858H, that produces hyperexcitablity in DRG neurons (decreased threshold and repetitive firing), and hypoexcitability (increased threshold and attenuated firing) within sympathetic ganglion neurons [13]. Since the sympathetic system controls cutaneous vascular tone, alterations in vessel constriction and dilation lead to the signs of redness, warmth and swelling characteristic of hyperemia.
Primary erythromelalgia is considered an ion channelopathy, and is cited as the first channelopathy described that has an association with chronic neuropathic pain [6-8,10]. Most cases of primary inherited erythromelalgia present during early childhood, though not always. A case of a patient presenting with hereditary erythromelalgia in the second decade, was determined to have a novel Nav1.7 mutation, termed Q10R. Q10R induces hyperexcitability in DRG neurons, but produces increases in excitability that are smaller than the change produced by I848T, an early-onset erythromelalgia mutation [14-16]. Though our patient has not undergone genetic testing or mutation analysis, we do not believe he has late-onset inherited erythromelalgia, since he has no family history of the condition. Aside from primary inherited erythromelalgia, there are cases of sporadic erythromelalgia in persons with no previous family history of the condition. Han et al. reports a case where a single amino acid substitution in the DIIS4-S5 linker of Nav1.7 was present in two children with erythromelalgia whose parents were asymptomatic. It was determined that the asymptomatic father was genetically mosaic for the mutation [14]. Secondary erythromelalgia has been associated with myeloproliferative disorders, autoimmune disorders, neuropathic disorders, infectious diseases, neoplasms, drug reactions, and other causes [4,6,9,15]. There is a paucity of studies addressing the mechanism of secondary erythromelalgia, and the pathophysiology is not completely understood.
Treatment of erythromelalgia can be challenging. The patient described in this case study was being treated with several medications meant to control neuropathic pain. However, they did not provide him with adequate relief. Invasive procedures for management of erythromelalgia have been described, and include epidural block, sympathetic ganglion block, brachial plexus block, and spinal cord stimulator implantation [9,16,17]. Our patient underwent a sympathetic stellate ganglion block. However, this procedure had only minimal effects on the patient’s pain. The development of effective oral treatment has proved to be a challenge. Several classes of medications, including local anesthetics (lidocaine), antiarrhythmics (mexiletine) and anti-epileptics (carbamazepine, phenytoin) target sodium channels and act as sodium blockers. However, they do not show sodium channel subtype specificity, and thus do not specifically target Nav1.7 [18]. These medications have varying degrees of efficacy in patients with pain due to Nav1.7 mutations [18]. Our patient was prescribed the non-selective sodium channel blocker mexiletine, which has been reported to improve the symptoms of erythromelalgia in certain patients [19,20]. Though incompletely understood, mexiletine is shown to have a normalizing effect on the pathological gating properties of the NaV 1.7 voltage gated sodium channel [21]. Interestingly, some Nav1.7 mutations enhance sensitivity to medications like carbamazepine and mexilitine, and patients with these particular mutations respond well to this treatment. Fischer et al. reports on a nav1.7 mutation termed V400M, seen in a three generation Canadian family with hereditary erythromelalgia that had pain relief with carbamazepine (CBZ). CBZ, a state dependent sodium channel blocker has historically been used to reduce neuronal excitability in epilepsy and trigeminal neuralgia. In the study, CBZ at clinically relevant concentrations normalized the effect of V400m mutation on channel activation [22]. Intravenous administration of methylprednisolone sodium succinate up to a 1,000 mg/day has also been employed in refractory cases with some success [23-26]. This treatment modality may be employed in the future for our patient. While the patient’s symptoms are mildly improved, complete resolution of symptoms has not yet been achieved.
Conclusion
Erythromelalgia is a rare disorder characterized by burning pain, warmth, and redness over the distal extremities. Erythromelalgia can either be primary or secondary to an underlying disease. Primary Erythromelalgia is further classified as familial or sporadic, with the familial form inherited in an autosomal dominant manner. The etiology of primary Erythromelalgia involves mutations in the sodium ion channel Nav1.7 that in humans is encoded by the SCN9A gene. Nav1.7 plays a critical role in the generation and conduction of action potentials in nociceptors, the pain sensing neurons. There is no established treatment and a variety of pain and neuro-modulating agents are used. Patients often see many specialists before a diagnosis is made, and the quality of life is severely impaired. This case highlights the importance of recognizing this rare entity.
References
- Mitchell SW (1878) On a rare vasomotor neurosis of the extremities and on the maladies with which it may be confounded. Am J Med Sci 76:17–36.
- Cook-Norris RH, Tollefson MM, Cruz-Inigo AE, Sandroni P, Davis MD, et al. (2011) Pediatric erythromelalgia: A retrospective review of 32 cases evaluated at Mayo Clinic over a 37-year period. J Am AcadDermatol66: 416–423.
- Davis MD, O'Fallon WM, Rogers RS, Rooke TW (2000) Natural history of erythromelalgia: presentation and outcome in 168 patients. Arch Dermatol 136:330-336.
- Al-Minshawy SM, El-Mazary AA (2014) An Egyptian child with erythromelalgia responding to a new line of treatment: a case report and review of the literature. J Med Case Rep 8:69.
- Kalgaard OM, Seem E, Kvernebo K (1997) Erythromelalgia: a clinical study of 87 cases. J Intern Med 242:191-197.
- Bennett DL, Woods CG (2014) Painful and painlesschannelopathies. Lancet Neurol 13:587-599.
- Kang BC, Nam J, Ahn EK, Yoon DM, Cho JG (2013) Secondary erythromelalgia- a case report. Korean J Pain 26:299-302.
- Waxman SG, Dib-Hajj SD (2005) Erythromelalgia: a hereditary pain syndrome enters the molecular era. Ann Neurol. 57:785-788.
- Bang YJ, Yeo JS, Kim SO, Park YH (2010) Sympathetic block for treating primary erythromelalgia. Korean J Pain 23:55-59.
- Waxman SG, Dib-Hajj S (2005) Erythromelalgia: molecular basis for an inherited pain syndrome. Trends Mol Med 11:555-562.
- Dib-Hajj SD, Rush AM, Cummins TR, Hisama FM, Novella S, (2005) Gain-of-function mutation in Nav1.7 in familial erythromelalgia induces bursting of sensory neurons. Brain. 128: 1847-1854. PMID: 15958509
- Cummins TR, Dib-Hajj SD, Waxman SG (2004) Electrophysiological properties of mutant Nav1.7 sodium channels in a painful neuropathy. J Neurosci. 24:8232-8236. PMID: 15385606
- Harty TP, Dib-Hajj SD, Tyrrell L, Blackman R, Hisama FM, et al. (2006) Na(V)1.7 mutant A863P in erythromelalgia: effects of altered activation and steady-state inactivation on excitability of nociceptive dorsal root ganglion neurons. J Neurosci. 26:12566-12575. PMID: 17135418
- Rush AM, Dib-Hajj SD, Liu S, Cummins TR, Black JA, et al. (2006) A single sodium channel mutation produces hyper- or hypoexcitability in different types of neurons. ProcNatlAcadSci U S A 103:8245-8250.
- Han C, Rush AM, Dib-Hajj SD, Li S, Xu Z, Wang Y, et al. (2006) Sporadic onset of erythermalgia: a gain-of-function mutation in Nav1.7. Ann Neurol. 59:553-558. PMID: 16392115
- Han C, Dib-Hajj SD, Lin Z, Li Y, Eastman EM, Tyrrell L, et al. (2009) Early- and late-onset inherited erythromelalgia: genotype-phenotype correlation. Brain. 132: 1711-1722.
- Kondo T, Uehara T, Ikegami A, Ikusaka M (2014) Paroxysmal burning pain caused by erythromelalgia. Lancet. 383:1692.
- Cohen JS (2000) Erythromelalgia: new theories and new therapies. J Am AcadDermatol 43:841-847.
- Cerci FB, Kapural L, Yosipovitch G (2013) Intractable erythromelalgia of the lower extremities successfully treated with lumbar sympathetic block. J Am AcadDermatol 69:270-272.
- Joost PH,Drenth, Stephen G. Waxman (2007) Mutations in sodium channel gene SCN9A cause a spectrum of human genetic pain disorders. J Clin Invest117: 3603-3609.
- Elgueta F, de la Cuadra-Fontaine JC, Clede L, Fierro C, Valderrama A (2013) Erythromelagia: a rare and hard-to-treat condition: a 9-year-old boy responsive to intravenous lidocaine and oral mexilitene. Pain Med 14:311-312.
- Park SK, Han KR, Lee YS, Hyun HS, Kim C (2001) Epidural block of Bupivacaine and mexiletine for erythromelalgia. Korean J Anesthesiol. 41:792-795.
- Cregg R, Cox JJ, Bennett DL, Wood JN, Werdehausen R (2014) Mexiletine as a treatment for primary erythromelalgia: normalization of biophysical properties of mutant L858F NaV 1.7 sodium channels. Br J Pharmacol
- Fischer TZ, Gilmore ES, Estacion M, Eastman E, Taylor S, (2006) A novel Nav1.7 mutation producing carbamazepine-responsive erythromelalgia. Ann Neurol65:733-741.
- Kakizaki A, Fujimura T, Kambayashi Y, Watabe A, Aiba S (2012) Successful treatment of adult-onset erythromelalgia with steroid pulse and pregabalin. Case Rep Dermatol4:242-246.
- Pfund Z, Stankovics J, Decsi T, Illes Z (2009) Childhood steroid-responsive acute erythromelalgia with axonal neuropathy of large myelinated fibers: a dysimmune neuropathy? NeuromusculDisord
Relevant Topics
Recommended Journals
- Journal of Lung Cancer Diagnosis & Treatment
- Advances in Cancer Prevention
- Breast Cancer: Current Research
- Cancer Surgery
- Immunology: Current Research
- Current Trend in Gynecologic Oncology
- Journal of Cancer Diagnosis
- Journal of Gastrointestinal Cancer and Stromal Tumors
- Cervical Cancer: Open Access
- Journal of Mucosal Immunology Research
- Journal of Oncology Research and Treatment
- Journal of Orthopedic Oncology
- Journal of Prostate Cancer
- Research and Reviews on Pathogens
Article Tools
Article Usage
- Total views: 19583
- [From(publication date):
September-2014 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 14918
- PDF downloads : 4665