Research Article Open Access
Acute Disseminated Encephalomyelitis following Dengue Virus Infection
Sanjeev Kumar Bhoi1, Suprava Naik2 and Jayantee Kalita1, Usha Kant Misra1*
1Department of Neurology, Sanjay Gandhi Post Graduate medical Sciences, Lucknow, India
2Department of Radio diagnosis, Sanjay Gandhi Post Graduate medical Sciences, Lucknow, India
- *Corresponding Author:
- Misra UK
Department of Neurology
Sanjay Gandhi Post Graduate Institute of Medical Sciences
Raebareily Road, Lucknow - 226014, India
Tel: +91 522 2494167
E-mail: drukmisra@rediffmail.com
Received date: November 14, 2013; Accepted date: January 15, 2014; Published date: January 21, 2014
Citation: Sanjeev Kumar B, Naik S, Jayantee K, Misra UK (2014) Acute Disseminated Encephalomyelitis following Dengue Virus Infection. J Neuroinfect Dis 5:139. doi: 10.4172/2314-7326.1000139
Copyright: © 2014 Sanjeev Kumar B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Neuroinfectious Diseases
Abstract
A 16 year boy was admitted with fever, headache, muscle pain, maculopapular rash and progressive altered sensorium. His serum dengue IgM was positive and was improving on conservative management. The patient developed right hemi paresis after 19 days and his MRI revealed multiple white mater lesions. He was diagnosed as acute disseminated encephalomyelitis (ADEM) following dengue hemorrhagic fever. ADEM is a rare neurological complication of dengue fever. However, the increasing use of magnetic resonance imaging in endemic areas may help to identify ADEM.
Keywords
Dengue haemorrhagic fever; Acute disseminated encephalomyelitis; Magnetic resonance image; ADEM; Prognosis
Introduction
Dengue is a RNA virus belongs to the family flaviviridae and is widely prevalent in both developed and developing nations. Neurological complications of dengue virus infection can be categorized into dengue encephalitis, encephalopathy, immune-mediated syndromes, dengue muscle dysfunction and neuro-ophthalmological disorders [1-3]. Acute Disseminated Encephalomyelitis (ADEM) is a monophasic, immune mediated acute inflammation and demyelination of central nervous system following recent infection or vaccination. There is paucity of report of ADEM following dengue virus infection. We report a patient with dengue hemorrhagic fever who developed ADEM.
Case Report
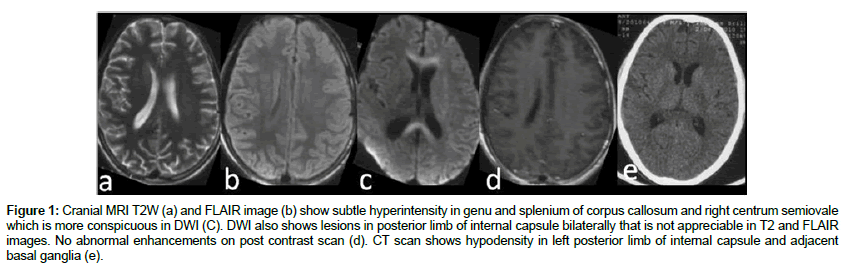
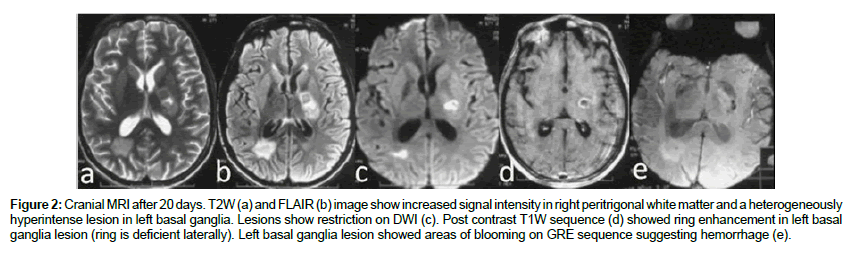
A 16-year-old village boy presented with moderate fever, for 12 days associated with chills and rigor. This was associated with holocranial non-throbbing headache, vomiting and muscle pain. On day 5 of illness, he developed erythematous maculopapular as well as petechial non purpuric rashes over the whole body with progressive altered sensorium for which he was referred to our institute. There was no history of seizure, otorrhoea, rhinorrhoea or recent vaccination. There is no family history of any chronic illness. On examination, his temperature was 101.4°F, pulse rate 110/min, regular, blood pressure 130/80 mm Hg, respiratory rate of 28/min. Petechial blanching rashes were present predominantly over the acral region and purpuric rashes over the chest and trunk. The patient was puffy with normal urinary output. He had sub-conjunctival hemorrhage and gastric aspirate revealed altered blood. He had 3 cm hepatomegaly. The patient was drowsy but arousable and had neck stiffness. He had quadriparesis with grade 4 muscle power and deep tendon reflexes were brisk with flexor plantar response. The investigations (Table 1) revealed hemoglobin of 11.9 gm/dl, platelet count 28000/mm3, total leukocyte count 5900/mm3, with polymorphs 65%, blood urea 79 mg/dL and serum creatinine 1.6 mg/dL, glutamate oxaloacetic transaminase (SGOT) 465 IU/L, glutamate pyruvic transaminase (SGPT) 313 IU/L and creatinine kinase for muscle was 1239 IU/L. Coagulation profile was deranged with International normalized ratio (INR) of 1.41 and activated partial thromboplastin time (aPTT) was prolonged by 9 seconds. Serum IgM antibody to dengue virus serum was positive. Antibody for leptospira and peripheral smear for malaria parasite were negative. Cranial magnetic resonance imaging (MRI) was obtained on day 3 of admission using 3 T Signa GE medical system and revealed subtle hyperintensity in genu and splenium of corpus callosum and right centrum semiovale in T2 and FLAIR sequences. The lesions were showing restricted diffusion on DWI. DWI also picked up lesions in posterior limb of internal capsule bilaterally that was not appreciable in T2 and FLAIR sequences. There was no contrast enhancement (Figure 1). Cerebrospinal fluid showed elevated protein 109 mg/dL with 30 cells (lymphocyte 60%) and normal sugar 81 mg/dL. The clinical diagnosis at this stage was dengue shock syndrome with encephalitis and muscle dysfunction. Patient was managed with intravenous fluid and because of gastrointestinal bleeding he was given platelet transfusion and fresh frozen plasma. During the hospital stay he developed pneumonia and septicaemia with severe hypoxemia for which he was mechanically ventilated and antibiotics were prescribed. On day 7 of ventilation when his ventilatory parameters and arterial blood gas (ABG) were maintained he developed increased weakness on the right side (grade 2) but his sensorium was normal. His CT scan revealed hypodensity of the left capsular and adjacent basal ganglia region (Figure 1e). The patient was gradually weaned off from the ventilator by day 14. His muscle pain subsided, coagulation parameters, liver and renal function normalized and he became afebrile. His hemiparesis persisted and a repeat MRI after 20 days revealed increased signal intensity in corona radiata bilaterally, right peritrigonal white matter and a heterogeneous hyperintense lesion in the left basal ganglia on T2 and FLAIR sequences. Restricted diffusion was seen in the basal ganglia lesion; however the corona radiata lesions did not show restriction of diffusion. The left basal ganglia lesion showed areas of blooming on GRE sequence suggesting hemorrhage. Post contrast T1W sequence showed ring enhancement in the left basal ganglia lesion (ring appears deficient laterally). The lesion in corpus callosum and centrum semiovale which were seen in previous MRI were not evident in this scan (Figure 2). He was diagnosed at this time as post dengue ADEM. The patient was discharged after 33 days of hospitalization with improving right hemiparesis and he was, able to walk with minimal support.
| Parameters | Values |
|---|---|
| Haemoglobin | 11.9 gm% |
| Leucocyte/mm3 | 5900 |
| Platelet count/mm3 | 28000 |
| Hematocrit | 37.9% |
| Serum Na+ | 156 meq/L |
| Serum K+ | 5.0 meq/L |
| Serum creatinine | 1.6 mg/dl |
| Serum albumin | 2.9 gm/dl |
| Prothrombin time (in seconds) | 15.6 (control 11.8) |
| Activated partial thromboplastin time (in seconds) | 39.8 (control 30.8) |
| International normalized ratio (INR) | 1.41 |
| Total bilirubin | 1.8 mg/dl |
| SGOT | 465 IU/L |
| SGPT | 313 IU/L |
| Serum Dengue IgM ELISA | Positive (twice) |
| Leptospira IgM, Malaria parasite | Leptospira IgM, Malaria parasite |
| Cerebrospinal fluid study | |
| Total cells/mm3 | 30 (Lymphocyte 60%, Neutrophil 40%) |
| Protein (mg/dl) | 109 |
| Glucose (mg/dl) | 81 (blood sugar 137) |
| Cryptococcal antigen, bacterial culture and AFB | Negative |
| Electroencephalography | Theta delta slowing |
Table 1: Laboratory parameters at admission.
Figure 1: Cranial MRI T2W (a) and FLAIR image (b) show subtle hyperintensity in genu and splenium of corpus callosum and right centrum semiovale which is more conspicuous in DWI (C). DWI also shows lesions in posterior limb of internal capsule bilaterally that is not appreciable in T2 and FLAIR images. No abnormal enhancements on post contrast scan (d). CT scan shows hypodensity in left posterior limb of internal capsule and adjacent basal ganglia (e).
Figure 2: Cranial MRI after 20 days. T2W (a) and FLAIR (b) image show increased signal intensity in right peritrigonal white matter and a heterogeneously hyperintense lesion in left basal ganglia. Lesions show restriction on DWI (c). Post contrast T1W sequence (d) showed ring enhancement in left basal ganglia lesion (ring is deficient laterally). Left basal ganglia lesion showed areas of blooming on GRE sequence suggesting hemorrhage (e).
Discussions
This patient with dengue manifested with encephalitis and muscle dysfunction characterized by altered sensorium with cerebro spinal fluid (CSF) pleocytosis and raised CK in the acute stage. During the convalescent period he developed ADEM which was characterized by right hemiparesis and white matter lesions bilaterally. ADEM is a monophasic, immune mediated acute inflammatory demyelinating disorder of central nervous system and usually occurs following a viral infection but may also occur following vaccination, bacterial and parasitic infection or even without apparent trigger [4]. The Neurological complications of dengue virus (DENV) infection are rare but can be grouped into dengue encephalopathy, encephalitis, immune-mediated syndromes, dengue muscle dysfunction, and neuroophthalmic disorders. CNS involvement following DENVinfection can occur during the para-infectious and or post-infectious period. Encephalopathy, encephalitis and transient muscle dysfunction have been reported [5]. There is controversy about the direct dengue viral injury of CNS [1]. Muscle dysfunction due to interstitial edema and hemorrhage has been reported recently [6]. Post infectious neurological manifestations are immune mediated usually begins 1-3 weeks after infections, but direct viral invasion is not the pathogenic mechanism for para infectious ADEM. In the present case the new neurological deficit in the form of right hemiparesis and new MRI lesion during convalescence is consistent with T cell mediated autoimmune response to myelin basic protein. MRI has become the most important test in the early diagnosis of ADEM [7]. Three distinct categories of disease can be classified using MRI criteria. (1) multifocal lesions in the white matter with or without basal ganglia involvement, (2) single or multifocal lesions only in the gray matter and (3) localized lesions in the brain stem, basal ganglia, or cerebellum. Cranial MRI findings in our patient fall into the first category involving the white matter and left basal ganglia. Corticosteroid has been used in ADEM but there is no class I evidence [8]. Our patient improved without corticosteroid. This patient highlights the wide spectrum of neurological manifestations following dengue virus infection, encephalitis and muscle dysfunction in acute stage and ADEM in the convalescent period.
References
- Solomon T, Dung NM, Vaughn DW, Kneen R, Thao LT, et al. (2000) Neurological manifestations of dengue infection. Lancet 355: 1053-1059.
- Cam BV, Fonsmark L, Hue NB, Phuong NT, Poulsen A, et al. (2001) Prospective case-control study of encephalopathy in children with dengue hemorrhagic fever. Am J Trop Med Hyg 65: 848-851.
- Misra UK, Kalita J, Syam UK, Dhole TN (2006) Neurological manifestations of dengue virus infection. J Neurol Sci 244: 117-122.
- Misra UK, Kalita (2011) In Diagnosis and management of Neurological disorders. J. Neurological manifestations of dengue virus infections. 21-32
- Carod-Artal FJ, Wichmann O, Farrar J, Gascón J (2013) Neurological complications of dengue virus infection. Lancet Neurol 12: 906-919.
- Misra UK, Kalita J, Maurya PK, Kumar P, Shankar SK, et al. (2012) Dengue-associated transient muscle dysfunction: clinical, electromyography and histopathological changes. Infection 40: 125-130.
- http://www.medscape.com/viewarticle/410603_3
- Alexander M, Murthy JM (2011) Acute disseminated encephalomyelitis: Treatment guidelines. Ann Indian Acad Neurol 14: S60-64.
Relevant Topics
- Bacteria Induced Neuropathies
- Blood-brain barrier
- Brain Infection
- Cerebral Spinal Fluid
- Encephalitis
- Fungal Infection
- Infectious Disease in Children
- Neuro-HIV and Bacterial Infection
- Neuro-Infections Induced Autoimmune Disorders
- Neurocystercercosis
- Neurocysticercosis
- Neuroepidemiology
- Neuroinfectious Agents
- Neuroinflammation
- Neurosyphilis
- Neurotropic viruses
- Neurovirology
- Rare Infectious Disease
- Toxoplasmosis
- Viral Infection
Recommended Journals
Article Tools
Article Usage
- Total views: 14810
- [From(publication date):
March-2014 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 10186
- PDF downloads : 4624