Research Article Open Access
Epidural versus Intravenous Patient Controlled Analgesia after Laparoscopic Gastric Bypass Surgery
| Stefan Neuwersch1, Michael Zink2, Vanessa Stadlbauer-Köllner3 and Karl Mrak4* | |
| 1Department of Anaesthesiology and Intensive Care Medicine, General Hospital Klagenfurt, Austria | |
| 2Department of Anaesthesiology and Intensive Care Medicine, General public Hospital of the Brothers of St. John of God St.Veit/Glan, Austria | |
| 3Department of Internal Medicine, Division of Gastroenterology and Hepatology, Medical University of Graz, Austria | |
| 4Department of Surgery, Division of General surgery, Medical University of Graz, Austria | |
| *Corresponding Author : | Karl Mrak Department of Surgery, Division of General surgery Medical University of Graz,Auenbruggerplatz 29, 8036-Graz, Austria Tel: 43-316-385-81548 E-mail: karl.mrak@klinikum-graz.at |
| Received: March 25, 2016 Accepted: April 19, 2016 Published: April 22, 2016 | |
| Citation: Neuwersch S, Zink M, Stadlbauer-Köllner V, Mrak K (2016) Epidural versus Intravenous Patient Controlled Analgesia after Laparoscopic Gastric Bypass Surgery. J Obes Weight Loss Ther 6:307. doi:10.4172/2165-7904.1000307 | |
| Copyright: © 2016 Neuwersch S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Obesity & Weight Loss Therapy
Abstract
Introduction: There is no clear consensus about the optimal postoperative pain management in morbidly obese patients undergoing bariatric surgery. The aim of this study was to evaluate the effects of Patient-Controlled- Epidural-Analgesia (PCEA) compared to Intravenous-Patient-Controlled-Analgesia (IV-PCA) in patients undergoing laparoscopic gastric bypass surgery. Methods: Between January 2013 and December 2014, 154 obese patients underwent laparoscopic gastric bypass surgery. Included in our analyses were all patients receiving either IV-PCA or PCEA in their postoperative course. Group comparison with respect to patient demographics, co-morbidities, details of surgical procedure, details of postoperative course, NRS-scores at rest, and medical follow-ups were performed. Results: Overall 63 (44.4%) patients were treated by PCEA, 79 (55.6%) patients by IV-PCA. We observed no differences across the groups with respect to sex, age, ASA-score, co-morbidities, postoperative BMI, body height, pre- and postoperative weight, ideal weight, weight loss, duration of surgery and postoperative ward. Patient´s BMI (p=0.025) and excess weight before surgery (p=0.029) were significantly higher in the IV-PCA-group. Surgical complications occurred significantly more often in the IV-PCA group (p=0.045). Concerning the postoperative pain management there was no statistically significant difference between different NRS-scores throughout the study period. However, individuals in the IV-PCA-group received significantly more paracetamol (p<0.0001) and diclofenac combined with orphenadrine (p=0.003). Duration of PCA was longer in the PCEA-group compared to patients treated with IV-PCA (p<0.01). Conclusions: Particularly for obese patients, PCEA is more beneficial than IV-PCA, which is borne out by a significantly lower incidence of surgical complications observed in patients receiving PCEA.
| Keywords |
| Patient-controlled-epidural-analgesia (PCEA); Intravenous-patient-controlled-analgesia (IV-PCA); gastric bypass surgery |
| Introduction |
| Obesity, the disease of the 21st century [1], affects more than 400 million people worldwide [2] and is recognized as one of the main preventable leading causes of death [3]. Co-morbidities like hypertension and type 2 diabetes mellitus are often associated with obesity and may lead to ischemic complications in terms of ischemic heart disease or peripheral arterial disease. Life expectancy of morbidly obese adults decreases by up to 7 years compared with normal-weight individuals [4]. To manage the chronic disease of obesity and its comorbidities, sustained weight loss is necessary. Individualized diet and lifestyle changes, physical activity and a change in behaviors may lead to weight loss, which reduces metabolic and cardiovascular risks [5]. Often weight regain occurs and additional pharmacotherapy with anti-obesity agents like orlistat is a possible choice of treatment. Unfortunately gastrointestinal adverse effects of pharmacotherapy often lead to decreased patient compliance [6]. |
| Bariatric surgery is an effective and safe approach in the treatment of obesity to maintain permanent weight loss and decrease obesity related co-morbidities [7,8]. Various trials have emphasized these benefits of surgical procedures [9], like minimally invasive laparoscopic Roux-en-Y gastric bypass or gastric sleeve resection, which show very low peri-operative morbidity and mortality rates [10]. Depending on patients BMI and related co-morbidities, surgery often represents the treatment of choice in morbidly obese patients. An optimal surgical treatment includes optimal pain management and there is an ongoing discussion in literature about the best post-operative pain management following bariatric surgery. |
| Beneficial effects of peri-operative use of thoracic epidural analgesia (TEA) on pulmonary, cardiovascular, and gastrointestinal functions have been shown [11]. Also health-related quality of life [12] as well as morbidity and mortality after abdominal surgery are affected by epidural analgesia. Moreover, thoracic epidural analgesia is considered the standard of postoperative pain management after major gastrointestinal surgery [13]. It has been proven that patient-controlled epidural analgesia (PCEA) is an effective pain management strategy after different surgical procedures [14] and leads to superior pain control compared to opioid based patient-controlled-analgesia (PCA) [15]. It seems obvious, that obese patients undergoing upper abdominal surgery will benefit from the convincing advantages of TEA, but due to technical difficulties, regional anaesthesia techniques are limited in obese individuals: surface anatomical landmarks may often be difficult to identify [16]. In addition, different studies have shown comparable pain control of PCEA and IV-PCA in obese patients undergoing bariatric surgery [17,18]. |
| What encouraged us to investigate the postoperative course of obese patients undergoing laparoscopic gastric bypass surgery, was the lack of studies evaluating the effects of epidural and intravenous opioid-based PCA. The finding that IV-PCA is a simple and effective alternative approach for postoperative pain management in obese patients is in contrast to convincing advantages of TEA on morbidity and mortality after abdominal surgery, and particularly obese patients may benefit from peri-operative use of thoracic epidural analgesia. The aim of this study was to evaluate the influence of pain management strategies after laparoscopic gastric bypass surgery on postoperative NRS-scores and postoperative course. |
| Methods |
| Between January 2013 and December 2014, a total of 154 obese patients underwent gastric bypass surgery at the General public Hospital of the Brothers of St. John of God St.Veit/Glan, Austria. According to the Interdisciplinary European Guidelines on Metabolic and Bariatric Surgery [19], a BMI ≥ 40 kg/m2 or BMI 35-40 kg/m2 associated with co-morbidities in which surgically induced weight loss is expected to improve the disorder were indications for surgical treatment. All patients were operated on by two senior surgeons under general anaesthesia using a standardized technique including the following major surgical steps: creation of a 10-15 ml gastric pouch, creation of a stapled sideside jejuno-jejunostomy followed by an antecolic, antegastric Rouxlimb and creation of a stapled end-side gastrojejunostomy. Depending on individual patient circumstances (e.g., patients wish after informed consent, needle phobia, failed epidural catheter placement) either IVPCA or PCEA was performed for postoperative pain management. Included in our analyses were all obese patients undergoing laparoscopic gastric bypass surgery who received either IV-PCA or PCEA for their postoperative pain management. Patients with a conversion to an open surgical technique or contraindications for a standardized pain therapy were excluded from analysis (study flow chart). |
| Postoperative pain management |
| Patients treated by IV-PCA received piritramide as repetitive bolus dose from 1.5-3 mg at the post-anaesthesia-care-unit (PACU) after surgery to achieve initial pain control defined as a pain value <4 on the numerical rating scale for pain (NRS). Further pain management was performed using a PCA-pump which allowed intravenous boluses of 1.2 mg of piritramide with a lockout interval of 10 minutes. No continuous infusion of ptritramid was given. Additionally, patients received 1 g of metamizole intravenously every six hours according to our standardized pain protocol. Alternatively 1 g of paracetamol every six hours, 40 mg of parecoxib every twelve hours or 75 mg of diclofenac combined with 30 mg of orphenadrine every twelve hours were administered. |
| Patients treated by PCEA were instrumented preoperatively with a thoracic epidural catheter placed at T7/T8 or T8/T9 vertebral interspace using a loss of resistance technique. 3 ml of lidocaine 1% with epinephrine 1:200.000 was administered through the catheter to identify accidental intravascular or intraspinal position. If needed during surgery, patients received intermittent bolus (5 ml) of ropivacaine 3.75 mg/ml combined with sufentanil 1 μg/ml. Postoperative pain management was performed using a PCEA-pump with ropivacaine 2 mg/ml and sufentanil 1 μg/ml. A continuous infusion rate of 4-8 ml/h and a patient controlled bolus of 4-6 ml depending on patients characteristics with a lockout time of 60 minutes were programmed. |
| Postoperative pain protocol |
| Postoperative pain was documented for each patient using the NRS. The postoperative pain protocol started immediately after surgery when the patient was adequate responsible and fully awake. All patients were visited two times a day by the medical pain service and the collected NSR-values were documented in an electronic database. In case of inadequate pain control or complications due to the postoperative pain management the patient was visited immediately by the pain service to improve pain therapy or resolve complications. Each event was documented additionally to the standardized pain protocol. |
| Data collection and statistical analyses |
| Patient demographics (age, gender, body height, current weight, excess weight, ideal weight, body-mass-index), co-morbidities, ASAclassification, details of surgical procedure (duration of surgery, surgical complications), details of postoperative course (pain protocol, concomitant medication, postoperative ward, duration of PCA), complications due to pain therapy (pruritus, epidural bleeding, infections or neurological complications), NRS-scores at rest (from surgery until fourth postoperative day), and medical follow-ups (percentage of excess weight loss, postoperative BMI) were documented electronically in a computer assisted database. Statistical analysis of the collected data was performed using IBM® SPSS® Statistics Version 21. Kolmogorov-Smirnov-test was used to test for normality of distribution. For parametric variables the Student‘s t-test was used, for non-parametric variables the Mann-Whitney U test was performed. Pearson‘s chi-square-test was performed to compare categorical variables. A p-value <0.05 was considered statistically significant. Mean values are reported +/- standard deviation. |
| Results |
| Patient characteristics |
| Overall 154 obese patients underwent bariatric surgery between January 2013 and December 2014. 12 (7.8%) patients were excluded from analysis due to the following reasons: in 3 (2.0%) cases surgical conversion to an open technique due to adhaesions was necessary, 9 (5.8%) patients received alternative pain management due to multiple allergies or refusal of IV-PCA or PCEA (study flow chart). Some 142 (92.2%) patients undergoing laparoscopic gastric bypass surgery were included in our statistical analyses. Of these 142 patients, 33 (23.2%) were male, 109 (76.8%) were female, median age was 39 years (range 16-70). According to the ASA-classification, 120 (84.5%) patients were classified as ASA II, 21 (14.8%) patients as ASA III, and 1 (0.7%) patient as ASA IV. GERD was diagnosed in 53 (37.3%) patients, type 2 diabetes mellitus in 18 (12.7%) patients, hypertension in 48 (33.8%) patients and coronary heart disease in 7 (4.9%) patients. Overall 39 (27.5%) patients were smokers. 9 (6.3%) patients suffered from COPD, asthma was diagnosed in 9 (6.3%) patients and obstructive sleep apnea in 5 (3.5%) patients. Postoperatively, 119 (83.8%) patients were monitored at the intensive-care-unit (ICU), 23 (16.2%) patients at the PACU. Median duration of PCEA was 3 days (range 1-9 days), median duration of IVPCA was 2 days (range 1-13 days). Patient characteristics and group distribution are shown in Tables 1 and 2. |
| Group comparison |
| Overall 63 (44.4%) patients were treated by PCEA, 79 (55.6%) patients by IV-PCA. We observed no differences across the groups with respect to sex, age, ASA-score, co-morbidities, postoperative BMI, body height, pre- and postoperative weight, ideal weight, weight loss, %EWL, duration of surgery and postoperative ward. Patient´s BMI (p = 0.025) and excess weight before surgery (p = 0.029) were significantly higher in the IV-PCA-group. Surgical complications occurred significantly more often in the IV-PCA group (p = 0.045). Data are shown in Table 2. |
| To investigate a possible selection bias for the type of pain management and postoperative complications in patients with BMI over 40 kg/m2 and 45 kg/m2 before surgery, additional analyses were performed. Group comparison showed no statistically significant differences in the number of patients in each group with BMI over 40 kg/m2 (p = 0.338) and 45 kg/m2 (p = 0.237) respectively. |
| Postoperative complications |
| Surgical complications were seen in 15 (10.6%) out of 142 patients. Minor complications (Clavien II) were identified in 2 (1.4%) patients, 13 (9.2%) patients had major complications (Clavien III), according to the Clavien classification for surgical complications [20,21]. No patient died within the first 30 days. In the PCEA-group 3 out of 63 (4.8%) patients suffered from surgical complications whereas in the IV-PCAgroup 12 out of 79 (15.2%) patients did. Statistical analyses showed that complications occurred significantly more often in the IV-PCA group (p = 0.045). Results are shown in Table 3. |
| 142 (100%) patients received metamizole, 60 (42.3%) patients received paracetamol, 12 (8.5%) patients parecoxib and 21 (14.8%) patients diclofenac combined with orphenadrine as additional pain therapy according to standardized post-operative pain management protocols. Overall 16 (11.3%) out of 142 patients had pruritus due to pain therapy. No other complications due to pain therapy were found. |
| Outcome of postoperative pain management |
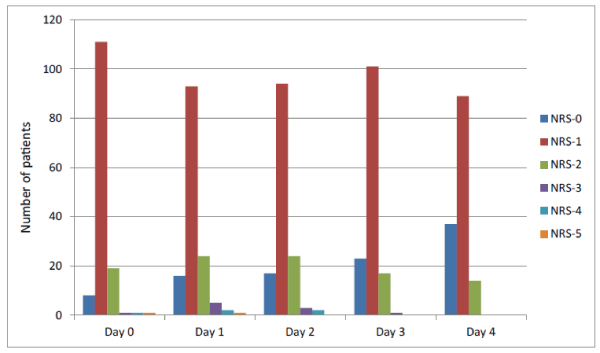
| Overall the majority of patients suffered from mild pain in their postoperative course with an increasing number of NRS-score 0 and decreasing numbers of NRS-score 1 or higher. On the second postoperative day there was no patient suffering from severe pain (NRS-score 5). The further postoperative course showed decreasing scores; no patient with NRS-score 4 or 5 on the third day, no patient with NRS-score 3 or higher on the fourth postoperative day respectively (Figure 1). |
| Concerning the postoperative pain management there was no statistically significant difference between different NRS-scores throughout the study period. However, individuals in the IV-PCAgroup received significantly more paracetamol (p < 0.0001) and diclofenac combined with orphenadrine (p = 0.003). Duration of PCA was longer in the PCEA-group compared to patients treated with IVPCA (p < 0.01). There was no statistically significant difference in pruritus due to pain therapy between the two groups. |
| Discussion |
| The present study demonstrates that both PCEA and IV-PCA are safe and effective methods for pain management after laparoscopic gastric bypass surgery with differences neither in pain NRS-scores at rest nor side effects due to pain therapy. These findings are in agreement with data obtained by Charghi et al.[17] who retrospectively compared the postoperative periods of 86 patients undergoing open Roux-en-Y gastric bypass (ORYGB). They have shown that the quality of pain control at rest after ORYGB is not affected by the method of analgesia: in all patients, VAS scores at rest after surgery were similar, independent of whether PCEA or IV-PCA were used. However, different studies and meta-analyses have shown, that PCEA is superior to opioid-based IVPCA in postoperative pain management: according to Pöpping et al. [22]. and Wu et al. [23]. pain management with PCEA is superior to IV-PCA with piritramide or morphine in patients undergoing surgery: better pain relief at rest and during activity compared to IV-PCA have been shown, regardless of continuous epidural infusion, PCEA, and respective analgesic drug. Similar to these findings a meta-analysis performed by Block et al. [24] has shown, that epidural analgesia achieves significantly reduced post-operative pain scores at rest and on movement on each post-operative day compared to pain scores in patients with parenteral opioids. These results were irrespective of analgesic solution, location of catheter placement or different forms of epidural analgesia. Moreover Werawatganon et al. [15]. performed a meta-analysis of nine studies involving 711 participants to compare pain relief after intra-abdominal surgery: individuals in the epiduralanalgesia- group had lower visual analogue pain scales than the IV-PCA group for up to 72 hours postoperatively. |
| In our study population patients in the IV-PCA group received significantly more paracetamol and diclofenac combined with orphenadrine, as compared to the PCEA group, which may proove analgesic superiority of thoracic epidural analgesia. On the other hand the duration of PCA was significantly shorter in the IV-PCA group which potentially can be explained by the fact, that patients in the IVPCA group received significantly more paracetamol and diclofenac combined with orphenadrine. |
| Carli et al. [12]. have demonstrated, that patients with TEA undergoing elective major gastrointestinal surgery had lower postoperative pain scores compared to patients with IV-PCA with morphine. There were also benefits in early mobilization, intake of food, functional exercise capacity and vitality in the epidural group. Patients with TEA were out of bed for a longer period of time and were ready to be discharged sooner. We agree with their opinion that an optimal postoperative pain management has major advantages in terms of earlier mobilization, functional exercise capacity or food intake. We think that except from better pain control, these conditions may be the main reasons for reduced post-operative complications and morbidity. In our study we found that individuals in the IV-PCA group had a significantly increased risk for surgical complications compared to patients with PCEA. Minor (Clavien II) and major (Clavien III) surgical complications were more likely in the IV-PCA group, which highlights the positive influence of peri-operative epidural analgesia on morbidity and mortality after gastrointestinal surgery. Anastomotic leakage, anastomotic fistula and anastomotic bleeding occurred more often in the IV-PCA group. Although the influence of epidural analgesia on anastomotic leakage, anastomotic healing and intestinal perfusion is discussed controversially [13,25,26] our data show that TEA may improve anastomotic healing after laparoscopic gastric bypass surgery. |
| Von Ungern-Sternberg et al. [27] and Hendolin et al. [28]. have demonstrated the positive effect of TEA on spirometric tests and lung function after gastrointestinal surgery. In this present study, postoperative pneumonia occurred only in patients receiving opioid-based PCA. This result highlights the influence of PCEA on peri-operative pulmonary morbidity and shows that pain-free ventilation, painfree coughing, and earlier mobilization in patients receiving epidural infusion may reduce peri-operative pulmonary complications. Although abdominal pain, sympathetic hyperactivity, and systemic opioid therapy may lead to gastrointestinal hypo-motility and post-operative ileus [29], we did not observe any difference in gastrointestinal dysfunction across both study groups. Bowel obstruction occurred only in one patient in the IV-PCA-group and was caused by mechanic obstruction of the jejuno-jejunostomy. In our opinion an optimal pulmonary function after gastrointestinal surgery improves arterial oxygenation which can prevent anastomotic ischemia and reduced perfusion near staple lines. |
| In spite of these advantages of TEA, epidural needle placement is sometimes a technical challenge in obese patients. Although surface anatomical landmarks [16] may often be difficult to identify and epidural needle placement is more difficult than in normal weight patients, we observed no epidural-related side effects, such as epidural bleeding, infections, or neurological complications. |
| The incidence of pruritus due to pain therapy was similar in both study groups. On average, individuals in the PCEA-group received epidural infusion for three days. This confirms the finding of Fotiadis et al. [30], that in clinical routine epidural analgesia usually continues for 72 hours after gastrointestinal surgery. |
| Other strategies in pain management include pre-incisional infiltrations. Schumann et al. [18] highlighted a multimodal approach of pain management after ORYGB: they have shown that pre-incision infiltration and supplemental field infiltration after surgery with bupivacaine in combination with an opioid-based IV-PCA provide equivalent pain relief compared to post-operative PCEA and superior pain relief compared to opioid-based IV-PCA alone. This seems to be an interesting uprating of postoperative IV-PCA, if epidural catheter placement fails or patients decline TEA. Possibly, additional intravenous pain therapy with paracetamol or non-steroidal anti-inflammatory drugs could be reduced with this approach. In addition, Choi et al. [5] have shown that an opioid-based IV-PCA is a safe and effective postoperative pain control strategy in obese individuals after RYGB. |
| Hypertension and type 2 diabetes mellitus are often associated with morbid obesity. In our study population we observed no differences across both groups with respect to overall co-morbidities. Hypertension was diagnosed in 33.8% of patients, which is in agreement with recent literature [31]. Type 2 diabetes mellitus was diagnosed in 12.7% of our patients and obstructive sleep apnea was diagnosed only in 3.5% of our patients, which is in contrast to recent literature [32,33]and seemed to be under-diagnosed. |
| There are several limitations of our present study that deserve mention. Due to the retrospective design of this study, typical biases may have affected our findings. In our department, NRS-scores were routinely assessed at rest. Pain while moving was not documented. Several studies [22-24] have shown that PCEA leads to significantly reduced post-operative pain scores on movement compared to opioidbased IV-PCA, and positive effects of TEA on early mobilization, function exercise capacity, patient vitality, and hospital stay were demonstrated [12]. The influence of pain management on early mobilization and length of hospital stay were not assessed in this study. We observed no differences across the groups with respect to sex, age, ASA-score, co-morbidities, post-operative BMI, body height, pre- and post-operative weight, ideal weight, weight loss, %EWL, duration of surgery, and post-operative ward, except that BMI (45 vs. 43 kg/m2) and excess weight before surgery (53 vs 60 kg) were higher in the IVPCA- group. We have to acknowledge a possible selection bias for the type of pain management. Our data do not show how many patients received IV-PCA because of failed epidural catheter placement. It seems possible that a higher BMI may lead to an increased failure rate in epidural catheter placement and may increase the incidence of surgical complications in the IV-PCA-group. To investigate this possible selection bias we performed additional analyses in patients with BMI over 40 kg/m2 and 45 kg/m2 respectively. These patients were stratified equally in each group and there was no statistically significant difference. Despite these limitations, we feel that our large study population, standardized surgical technique performed by two surgeons only and standardized postoperative pain management allow us to make valid conclusions. |
| In summary, PCEA and opioid-based IV-PCA are both safe and effective methods for pain relief after elective laparoscopic gastric bypass surgery. However, this present study provides evidence that, particularly for obese patients, PCEA is more beneficial than IVPCA, which is borne out by a significantly lower incidence of surgical complications observed in patients receiving PCEA. Additional prospective randomized clinical trials are required in order to confirm PCEA as gold standard for postoperative pain management after elective laparoscopic gastric bypass surgery. |
References
- Haslam DW, James WP (2005) Obesity. Lancet 366: 1197-1209.
- (2000) Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 894: i-xii, 1-253.
- Pischon T, Boeing H, Hoffmann K, Bergmann M, Schulze MB, et al. (2008) General and abdominal adiposity and risk of death in Europe. N Engl J Med 359: 2105-2120.
- Peeters A, Barendregt JJ, Willekens F, Mackenbach JP, Al Mamun A, et al. (2003) Obesity in adulthood and its consequences for life expectancy: a life-table analysis. Ann Intern Med 138: 24-32.
- Monkhouse SJ, Morgan JD, Bates SE, Norton SA (2009) An overview of the management of morbid obesity. Postgrad Med J 85: 678-681.
- Carter R, Mouralidarane A, Ray S, Soeda J, Oben J (2012) Recent advancements in drug treatment of obesity. Clin Med (Lond) 12: 456-460.
- Colquitt JL, Picot J, Loveman E, Clegg AJ (2009) Surgery for obesity. Cochrane Database Syst Rev CD003641.
- Mingrone G, Panunzi S, De Gaetano A, Guidone C, Iaconelli A, et al. (2012) Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med 366: 1577-1585.
- Buchwald H, Avidor Y, Braunwald E, Jensen MD, Pories W, et al. (2004) Bariatric surgery: a systematic review and meta-analysis. JAMA 292: 1724-1737.
- Consortium LAoBS, Flum DR, Belle SH, King WC, Wahed AS, et al. (2009) Perioperative safety in the longitudinal assessment of bariatric surgery. N Engl J Med 361: 445-454.
- Clemente A, Carli F (2008) The physiological effects of thoracic epidural anesthesia and analgesia on the cardiovascular, respiratory and gastrointestinal systems. Minerva Anestesiol 74: 549-563.
- Carli F, Mayo N, Klubien K, Schricker T, Trudel J, et al. (2002) Epidural analgesia enhances functional exercise capacity and health-related quality of life after colonic surgery: results of a randomized trial. Anesthesiology97: 540-549.
- Freise H, Van Aken HK (2011) Risks and benefits of thoracic epidural anaesthesia. Br J Anaesth 107: 859-868.
- Liu SS, Allen HW, Olsson GL (1998) Patient-controlled epidural analgesia with bupivacaine and fentanyl on hospital wards: prospective experience with 1,030 surgical patients. Anesthesiology 88: 688-695.
- Werawatganon T, Charuluxanun S (2005) Patient controlled intravenous opioid analgesia versus continuous epidural analgesia for pain after intra-abdominal surgery. Cochrane Database Syst Rev: CD004088.
- Teoh DA, Santosham KL, Lydell CC, Smith DF, Beriault MT (2009) Surface anatomy as a guide to vertebral level for thoracic epidural placement. AnesthAnalg 108: 1705-1707.
- Charghi R, Backman S, Christou N, Rouah F, Schricker T (2003) Patient controlled i.v. analgesia is an acceptable pain management strategy in morbidly obese patients undergoing gastric bypass surgery. A retrospective comparison with epidural analgesia. Can J Anaesth50: 672-678
- Schumann R, Shikora S, Weiss JM, Wurm H, Strassels S, et al. (2003) A comparison of multimodal perioperative analgesia to epidural pain management after gastric bypass surgery. AnesthAnalg 96: 469-474.
- Fried M, Yumuk V, Oppert JM, Scopinaro N, Torres AJ, et al. (2013) European Association for the Study of O, International Federation for the Surgery of Obesity - European C. Interdisciplinary European Guidelines on metabolic and bariatric surgery. Obes Facts 6: 449-468.
- Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240: 205-213.
- Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, et al. (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250: 187-196.
- Pöpping DM, Zahn PK, Van Aken HK, Dasch B, Boche R, et al. (2008) Effectiveness and safety of postoperative pain management: a survey of 18 925 consecutive patients between 1998 and 2006 (2nd revision): a database analysis of prospectively raised data. Br J Anaesth101: 832-840
- Wu CL, Cohen SR, Richman JM, Rowlingson AJ, Courpas GE, et al. (2005) Efficacy of postoperative patient-controlled and continuous infusion epidural analgesia versus intravenous patient-controlled analgesia with opioids: a meta-analysis. Anesthesiology 103: 1079-1088.
- Block BM, Liu SS, Rowlingson AJ, Cowan AR, Cowan JA Jr, et al. (2003) Efficacy of postoperative epidural analgesia: a meta-analysis. JAMA 290: 2455-2463.
- Johansson K, Ahn H, Lindhagen J, Tryselius U (1988) Effect of epidural anaesthesia on intestinal blood flow. Br J Surg 75: 73-76.
- Sala C, García-Granero E, Molina MJ, García JV, Lledo S (1997) Effect of epidural anesthesia on colorectal anastomosis: a tonometric assessment. Dis Colon Rectum 40: 958-961.
- Ungern-Sternberg BSV, Regli A, Reber A, Schneider MC (2005) Effect of obesity and thoracic epidural analgesia on perioperative spirometry. Br J Anaesth 94: 121-127.
- Hendolin H, Lahtinen J, Länsimies E, Tuppurainen T, Partanen K (1987) The effect of thoracic epidural analgesia on respiratory function after cholecystectomy. ActaAnaesthesiolScand 31: 645-651.
- Marret E, Remy C, Bonnet F; Postoperative Pain Forum Group (2007) Meta-analysis of epidural analgesia versus parenteral opioid analgesia after colorectal surgery. Br J Surg 94: 665-673.
- Fotiadis RJ, Badvie S, Weston MD, Allen-Mersh TG (2004) Epidural analgesia in gastrointestinal surgery. Br J Surg 91: 828-841.
- Choi YK, Brolin RE, Wagner BK, Chou S, Etesham S, et al. (2000) Efficacy and safety of patient-controlled analgesia for morbidly obese patients following gastric bypass surgery. ObesSurg 10: 154-159.
- Ford ES, Li C, Zhao G (2010) Prevalence and correlates of metabolic syndrome based on a harmonious definition among adults in the US. J Diabetes 2: 180-193.
- Poulain M, Doucet M, Major GC, Drapeau V, Sériès F, et al. (2006) The effect of obesity on chronic respiratory diseases: pathophysiology and therapeutic strategies. CMAJ 174: 1293-1299.
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
| Figure 1 |
Relevant Topics
- Android Obesity
- Anti Obesity Medication
- Bariatric Surgery
- Best Ways to Lose Weight
- Body Mass Index (BMI)
- Child Obesity Statistics
- Comorbidities of Obesity
- Diabetes and Obesity
- Diabetic Diet
- Diet
- Etiology of Obesity
- Exogenous Obesity
- Fat Burning Foods
- Gastric By-pass Surgery
- Genetics of Obesity
- Global Obesity Statistics
- Gynoid Obesity
- Junk Food and Childhood Obesity
- Obesity
- Obesity and Cancer
- Obesity and Nutrition
- Obesity and Sleep Apnea
- Obesity Complications
- Obesity in Pregnancy
- Obesity in United States
- Visceral Obesity
- Weight Loss
- Weight Loss Clinics
- Weight Loss Supplements
- Weight Management Programs
Recommended Journals
Article Tools
Article Usage
- Total views: 11292
- [From(publication date):
April-2016 - Aug 18, 2025] - Breakdown by view type
- HTML page views : 10307
- PDF downloads : 985
