Short Communication Open Access
Microscopic Colitis: An Overview
Ashraf MU*, Aslam M, Zaheer MS, Rabbani MU, Khan SA and Ashraf J
J N Medical College, AMU, Aligarh, Uttar Pradesh, India
- *Corresponding Author:
- Dr. Muhammad Uwais Ashraf
J N Medical College, AMU, Aligarh
Uttar Pradesh, India,
Tel: +6096275587
E-mail: uwaisashraf@gmail.com
Received date: June 2, 2014; Accepted date: September 23, 2014; Published date: September 25, 2014
Citation: Ashraf MU, Aslam M, Zaheer MS, Rabbani MU, Khan SA, et al. (2014) Microscopic Colitis: An Overview. Microinflammation 1:108. doi: 10.4172/2381-8727.1000108
Copyright: © 2014 Ashraf MU, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Visit for more related articles at International Journal of Inflammation, Cancer and Integrative Therapy
Abstract
Microscopic colitis may be defined as a clinical syndrome, of unknown etiology, consisting of chronic watery diarrhea, with no or minimal changes seen on colonoscopy. Therefore, a definitive diagnosis is only possible by histology. The epidemiological characteristics of this disease have become more elaborate over the past few years, and most of the data has come from Western countries. Microscopic colitis includes two histological subtypes [collagenous colitis (CC) and lymphocytic colitis (LC)] with no differences in clinical presentation and management. Collagenous colitis is characterized by a thickening of the sub epithelial collagen layer which is not seen in LC. The main feature of LC is an increase in the density of intra-epithelial lymphocytes in the surface epithelium. A number of theories have been proposed over the years regarding the pathogenesis of this entity, including the role of luminal agents, autoimmunity, eosinophils, genetics (human leukocyte antigen), biliary acids, infections, alterations in fibroblasts, and drug intake; drugs like ticlopidine, carbamazepine or ranitidine are especially associated with the development of LC, while CC is more frequently linked to cimetidine, non-steroidal anti-inflammatory drugs and lansoprazole. Recent evidence has added new pharmacological options for the treatment of microscopic colitis: the role of steroidal therapy, especially oral budesonide, has gained popularity, as well as immunosuppressive agents such as azathioprine and 6-mercaptopurine. The use of anti-tumor necrosis factor-α agents, infliximab and adalimumab, constitutes a new, interesting tool for the treatment of microscopic colitis.
Introduction
Microscopic colitis (MC) is an intestinal inflammatory disorder which is characterized by specific histopathological findings that include an increased number of lymphocytes in the colonic epithelium. The characteristic feature is chronic watery diarrhea in the absence of macroscopic changes in the large bowel.
It was once considered a rare diagnosis, but now its prevalence is increasing [1]. There are two main variants of this disease – Lymphocytic Colitis and Collagenous Colitis. These are more or less similar conditions and are commonly referred to with a single term- ‘Microscopic Colitis’. Lymphocytic Colitis is characterized by an increased infiltration of the colonic epithelium and lamina propria by lymphocytes. Collagenous Colitis, in addition to the inflammatory changes also shows a markedly thickened sub-epithelial collagen band adjacent to the basal membrane. There is a weak association between Lymphocytic Colitis and inflammatory bowel disease (IBD).
Knowledge about microscopic colitis has improved over the last few years, with landmark reports such as the recent reports of endoscopic findings and the use of anti-tumor necrosis factor (TNF)-a agents (infliximab and adalimumab) in the management of refractory forms of this disease, adding to literature. Radiographic and endoscopic features are characteristically normal. The main symptom is chronic watery diarrhea without bleeding. This disease is more common among older individuals and in females. Although a genetic association has not yet been proven, familial occurrence has been reported. Smoking is a risk factor and bile acid malabsorption is often found [2].
Symptoms of Microscopic Colitis are different from those of Ulcerative Colitis (UC) and Crohn’s Disease. In UC and Crohn’s disease the lining of the bowel is often inflamed and ulcerated on colonoscopy. In Microscopic Colitis, the appearance of the bowel lining is usually normal on colonoscopy. However, the colonic biopsies show characteristic changes - hence the name Microscopic Colitis. Also, a typical symptom of UC and Crohn’s disease is bloody diarrhea whereas, in Microscopic Colitis the diarrhea is watery and does not contain blood.
Types of Microscopic Colitis
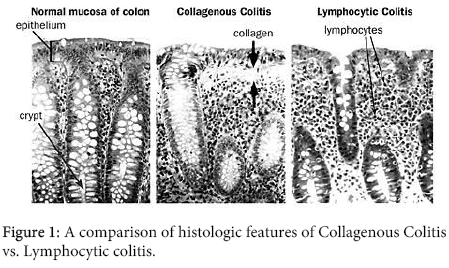
Microscopic colitis has been subdivided into two forms: collagenous and lymphocytic colitis. The biopsy of the colonic mucosa in collagenous colitis (CC) shows a thickened sub epithelial collagen band along with chronic inflammatory changes in the lamina propria and intraepithelial lymphocytosis. However, in lymphocytic colitis (LC) the histologic findings are similar but there are no thickened collagen bands. A third variant of microscopic colitis with a thickened collagen band but no lymphocytic infiltrate has also been described [3]. It has been shown that CC and LC differ slightly in their clinical presentations and disease course. Both the conditions present with watery, non-bloody diarrhea and may be associated with other conditions including connective tissue disorders, autoimmune diseases, and celiac sprue.
CC was initially reported to affect predominantly the older women in the sixth and seventh decades, whereas LC was shown to occur equally in both genders and approximately 10 years earlier. However some recent reports have questioned this dichotomy [4]. Several patients have been observed to convert from one form to the other [5], and subsequent studies have failed to confirm differences in gender distribution and age at presentation [6]. Also, recent long-term follow-up studies suggest that about 50% of patients who were initially diagnosed to have CC subsequently converted to LC [7], and this finding may reflect the patchy distribution of the thickened collagen band [8]. Close examination of biopsy specimens suggests that the morphology of the microscopic colitides lies along a continuous spectrum and that there is substantial overlap between the CC and LC [9]. Figure 1 outlines the histologic differences between CC and LC.
Epidemiology and risk factors
Microscopic colitis was first described in the early 1980s, and at that time it was felt to be a rare disease [10]. However, over the last few decades, it has been proved that microscopic colitis is a common disease with high incidence and prevalence rates. Early epidemiologic data mainly from retrospective studies that were conducted in Sweden showed rising incidences from the years 1983–1998 [11].
Most recent north American studies show incidence rates of 7.1 per 100,000 person-years for collagenous colitis and 12.6 per 100,000 person-years for lymphocytic colitis, respectively [12]. The overall prevalence for microscopic colitis was reported to be 103.0 per 100,000 persons (39.3 per 100,000 persons for collagenous colitis and 63.7 per 100,000 persons for lymphocytic colitis). Both incidence and prevalence rates of microscopic colitis seem to approach those of the inflammatory bowel diseases like ulcerative colitis and Crohn’s disease [13].
Newer epidemiological studies confirm this high incidence and prevalence also in the European population and in the Asian population [14]. Established risk factors for microscopic colitis are female gender [15], higher age [15], concomitant autoimmune diseases such as thyroid disease or celiac disease [16], a past or current diagnosis of malignancy [17], and a history of solid organ transplant [18]. In population-based studies the female to male ratio ranges from 4.4–7.9:1 for collagenous colitis and from 1.8–5:1 for lymphocytic colitis [12,15]. Various studies have established that the incidence of microscopic colitis increases substantially as the age advances [15]. 30–50% of patients with microscopic colitis have at least one concomitant autoimmune disease with as many as 10–20% having thyroid disease [16,17].
Organ transplantation patients have 50-fold higher risk of microscopic colitis than the general population [18]. Strong associations have been demonstrated between microscopic colitis and certain drugs like NSAIDs, PPIs, SSRIs, beta blockers, statins, and bisphosphonates [19,20]. A very recent study identified that PPI use increases the number of intraepithelial lymphocytes even without causing diarrhea. Whether this finding hints at the fact that intraepithelial lymphocytes are increased before symptom onset or that increased lymphocytes do not cause symptoms in some patients is not clear yet and a waits to be clarified in prospective studies [21].
Clinical features
Microscopic colitis characteristically presents with chronic or intermittent diarrhea. A large number of patients also complain of nocturnal diarrhea. The frequency of diarrhea is usually more than three times per day and it is not uncommon for patients to have 10-15 episodes per day. These stools are watery but do not contain blood. A minority of patients may also complain of fecal incontinence [22]. The diarrhea may stop for weeks or months and may restart to continue for weeks again.
In addition to diarrhea, some patients also complain of cramping abdominal pain and weight loss. An important aspect of the diagnosis of microscopic colitis is to differentiate this disease from closely resembling clinical entities like irritable bowel syndrome, inflammatory bowel disease and infectious colitis. Although the symptoms of microscopic colitis have a considerable overlap with those of irritable bowel disease, yet there are clinically discernible differences between the two conditions which should be borne in mind. The most prominent feature of microscopic colitis is diarrhea, whereas in irritable bowel syndrome, diarrhea is one of the components of bowel irregularities which include alternating stages of constipation and diarrhea along with abdominal pain, flatulence, bloating and the sensation of incomplete evacuation. Microscopic colitis characteristically presents with night time bouts of diarrhea and weight loss, both of which are not seen in irritable bowel syndrome.
Ulcerative colitis and Crohn’s disease also need to be differentiated from microscopic colitis. Although fecal leukocytes may be present in microscopic colitis, but fever, vomiting, hematochezia, etc are characteristically absent. Concomitant steatorrhea should also suggest an alternative diagnosis like celiac sprue. Unlike ulcerative colitis, microscopic colitis does not does not appear to be associated with an increased risk of colon cancer.
Arthralgias may be present in patients with microscopic colitis and they are due to the association of microscopic colitis with autoimmune disorders [23]. An elevated ESR and CRP have also been documented in some patients of microscopic colitis, even in the absence of clinical evidence of autoimmune disorders. A significant correlation has been found between microscopic colitis and celiac sprue. 2-10% patients of microscopic colitis have small intestinal changes consistent with celiac sprue, whereas up to one-third of patients with celiac sprue have findings suggestive of microscopic colitis on colonic biopsy [22].
The etiology of microscopic colitis is multifactorial. Though some familial clustering of microscopic colitis cases has been reported, there is only minimal evidence to suggest a genetic basis of microscopic colitis. The association between HLA-DQ2, DQ1, DQ3, and microscopic colitis and the high prevalence of a TNF gene polymorphism in patients with microscopic colitis deserves further attention as it may lead to a discovery of a hereditary component of microscopic colitis of presently unknown penetration [23]. Also, metalloproteinase- 9 gene variations have been reported in association with collagenous colitis [24].
Very strong evidence exists for an autoimmune basis of this disorder. The association of microscopic colitis with autoimmune disorders such as celiac sprue, thyroid disease, and rheumatoid arthritis, supports that microscopic colitis may have a strong association with autoimmune diseases. But no specific autoantibody has been identified as yet in connection with microscopic colitis [16].
Certain luminal factors also play an important role in the pathogenesis of microscopic colitis. Various drugs have been reported to increase the incidence of microscopic colitis. Other luminal factors such as infectious or toxic agents have also been shown to increase the incidence of microscopic colitis [19]. Yesinia species [25], Clostridium difficile [26], and Campylobacter species [27] were suggested in some case reports as the causative agents of microscopic colitis. In some studies, bile acid malabsorption has been found (in up to 60% of patients with lymphocytic colitis and up to 44% of patients with collagenous colitis) to increase the incidence of microscopic colitis [28]. A recent study using sigmoid tissues from patients with collagenous colitis and lymphocytic colitis has demonstrated that sodium transport and epithelial barrier functions are disturbed in patients with microscopic colitis [29].
It has further been reported that in patients with microscopic colitis increased IFN-, TNF-, and IL-1 levels are seen, which suggest that a Th1 cytokine profile is involved in the pathogenesis of microscopic colitis [29]. Cigarette smoking is strongly suggested to be a risk factor for microscopic colitis and preliminary reports suggest that lung cancer is also associated with microscopic colitis [30]. The odds ratio for lymphocytic colitis and smoking (OR, 3.8) is higher than for collagenous colitis and smoking (OR, 2.4), though this difference was presently calculated on a small cohort of 120 patients with collagenous colitis, 70 patients with lymphocytic colitis, and 128 controls and thus has to be verified in larger subsets of patients [31]. Apart from the inflammatory processes contributing to the pathogenesis of this disorder, certain neuronal mechanisms have also been implicated. A recent study identified increased chromogranin A, chromogranin B, and secretoneurin levels in feces of patients with collagenous colitis compared to controls. These observations point towards a neurogenic process in the pathogenesis of microscopic colitis and these stool markers may be helpful in differentiating microscopic colitis from irritable bowel syndrome and inflammatory bowel disease [32].
Diagnosis
The diagnosis of microscopic colitis is largely based on normal or minimally nonspecific endoscopic findings with biopsies showing histopathologic findings consistent with a diagnosis of either collagenous colitis or lymphocytic colitis. When making a diagnosis of microscopic colitis, the first step is a thorough history with particular attention to the differential diagnoses like IBS, IBD, and infectious Colitis. Radiographic and laboratory tests may be helpful to rule out these conditions but are typically unremarkable with microscopic colitis. There is no increased risk in patients with microscopic colitis undergoing diagnostic procedures and complicated cases like spontaneous perforations are rarely reported [33].
The next step includes a colonoscopy that usually shows a macroscopically normal mucosa. Recent publications list non-specific changes such as abnormal vascular markings, erythema, or mucosal thickening. It is important to take biopsies from areas throughout the colon, as they are needed to make a diagnosis of microscopic colitis. Both collagenous colitis and lymphocytic colitis show a lymphocytic infiltration of the lamina propria and the colonic epithelium [34]. Collagenous colitis can be distinguished from lymphocytic colitis by the presence of a marked thickening of the sub epithelial collagen layer [34]. The typical histopathological features of collagenous colitis and lymphocytic colitis are listed in the Table 1.
| COLLAGENOUS COLITIS | LYMPHOCYTIC COLITIS |
|---|---|
| 1.Thickening of subepithelial collagen layer of >10μm | 1. Intraepithelial lymphocytosis (≥20 intraepithelial lymphocytes/100 superficial epithelial cells |
| 2.Inflammation in the lamina propria consisting of mainly lymphocytes and plasma cells | 2. Inflammation in the lamina propria consisting of mainly lymphocytes and plasma cells |
| 3.Epithelial damage such as flattening and detachment | 3.Epithelial damage such as flattening and detachment |
| 4.Intraepithelial lymphocytosis could be present but is not necessary for diagnosis | 4.Subepithelial collagen layer not present or <10 μm |
Table 1: Table showing histological features of Collagenous Colitis and Lymphocytic Colitis.
Numerous studies have shown that the microscopic lesions can be skipped, and therefore random colonic biopsies should be taken [35]. Most recent studies show that biopsies of the rectosigmoid colon alone are insufficient to rule out microscopic colitis [36]. Since up to 40% of cases will be missed with biopsies taken from the rectosigmoid region alone, a full colonoscopy with biopsies taken from every region is recommended [37]. Novel tools may help in making the diagnosis of microscopic colitis in future and a recent report on confocal laser endomicroscopy guiding the diagnosis of microscopic colitis is an indication of the use of newer methods in the diagnosis of microscopic colitis [38].
Treatment
Prior to any drug treatment a medication history should be taken and potentially precipitating medications should be stopped. Also, the associated conditions like celiac disease should be appropriately sought and managed. In some patients dietary restrictions (e.g., avoiding caffeine or lactose) might be helpful, especially if symptoms are triggered by these food items. As studies provide evidence that microscopic colitis is associated with cigarette smoking it is worth advising to stop smoking prior to any drug treatment. Randomized controlled trials have shown that the steroid budesonide is an effective treatment for moderate to severe collagenous and lymphocytic colitis. This was confirmed by a recent meta-analysis [39]. Antidiarrheal agents such as loperamide may be used in patients with mild or transient symptoms.
Other steroids like prednisolone were investigated in a double-blind, placebo-controlled randomized trial. Due to the small number of patients included and the short duration of follow up, the results of this trial were inconclusive and of limited value. Presently, steroids other than budesonide are not supported by evidence from clinical trials in the treatment of microscopic colitis [41]. For patients with mild to moderate symptoms antidiarrheal drugs can be tried although no trials have supported this treatment. Retrospective trials found some clinical benefit for loperamide with doses ranging from 2 to 16 mg per day [19].
Due to the safety of this agent and the possibility of spontaneous remission of microscopic colitis, loperamide is frequently used as first-line therapy for patients with mild symptoms. Prior to budesonide availability, aminosalicylates were the mainstay for the treatment of microscopic colitis. Treatment with aminosalicylates is presently not supported by randomized, placebo controlled clinical trials. Retrospective case series have suggested symptomatic improvement in up to 50% of patients treated with mesalamine (5-aminosalicylic acid, 5-ASA) [19]. Other therapies were either tested in small uncontrolled trials or suggested from retrospective analyses and individual case reports. These approaches include immunosuppressants like azathioprine and methotrexate [42], biological anti- TNF treatments [43], Boswellia serrata extract, probiotics, pentoxifylline [44], verapamil [45], octreotide, and empirical antibiotic treatment [46]. For probiotics, E. coli Nissle 1917 improved stool consistency and stool frequency in an open label study on 14 patients with collagenous colitis, E. coli Nissle 1917 was not tested in patients with lymphocytic colitis [47].
Other probiotics like Lactobacillus acidophilus LA-5 and Bifidobacterium animalis subspp. can be tried. Lactis B12 failed to be superior to placebo treatment, in a 12- week clinical trial in patients with collagenous colitis [48]. A placebo-controlled, randomized clinical trial testing bismuth subsalicylate in patients with microscopic colitis reported a 100% clinical and histological remission rate in the treatment group, compared to 0% in the placebo group [49].
For refractory microscopic colitis, azathioprine was used in an uncontrolled fashion in 9 patients. 7 of these patients went into a remission of symptoms and 1 additional patient had at least a significant improvement of symptoms [50]. More recently anti-TNF drugs were tested in patients refractory to budesonide treatment [43]. One study tested the anti-TNF drug adalimumab in 3 patients who developed side effects or were refractory to methotrexate and budesonide. 2 patients tolerated adalimumab treatment well and were in remission after 6 weeks whereas 1 patient had to stop the medication due to severe side effects [50]. Historical evidence also suggests that surgical interventions may be resorted to in a small subset of patients not responding to all the available medical therapies [51]. Subtotal colectomy, sigmoidostomy, or diversion ileostomy was reported to alleviate symptoms. However, with the use of budesonide, such procedures have largely gone into oblivion.
Conclusion
Microscopic colitis is a common cause of chronic watery diarrhea. The epidemiological burden has been increasing over the past few years. Colon biopsies are required for the definitive diagnosis, as the colonoscopy is largely normal. Over the last few years, a series of new pharmacological agents for the treatment of MC have come up. The role of steroidal therapy, especially regarding the use of oral budesonide, has gained popularity. Immunosuppressive agents as azathioprine and 6-mercaptopurine have also been tried. Few and very recent evidences have demonstrated the usefulness of anti-TNF-a agents (infliximab and adalimumab) as the possible treatment of MC, but larger studies are required to confirm these data.
References
- Pardi DS, Smyrk TC, Tremaine WJ, Sandborn WJ (2002) Microscopic colitis: a review.Am J Gastroenterol 97: 794-802.
- Chetty R, Govender D (2012) Lymphocytic and collagenous colitis: an overview of so-called microscopic colitis.Nat Rev GastroenterolHepatol 9: 209-218.
- Veress B, Löfberg R, Bergman L (1995) Microscopic colitis syndrome.Gut 36: 880-886.
- Jessurun J, Yardley JH, Lee EL, Vendrell DD, Schiller LR, et al. (1986) Microscopic and collagenous colitis: different names for the same condition?Gastroenterology 91: 1583-1584.
- Bowling TE, Price AB, al-Adnani M, Fairclough PD, Menzies-Gow N, et al. (1996) Interchange between collagenous and lymphocytic colitis in severe disease with autoimmune associations requiring colectomy: a case report.Gut 38: 788-791.
- Fernandez-Banares F, Forne M, Esteve M (1997). Collage nous colitis and lymphocytic colitis in Terrassa, Spain: an epidemiological study 1993-1996. Gastroenterology 112: 15.
- Mantzaris GJ, Archavlis E, Kourtessas D (1997). The natural history of microscopic (lymphocytic) colitis and collagenous colitis after long-term treatment with 5-aminosalicylic acid. Gastroenterology 112: 1032.
- Wang KK, Perrault J, Carpenter HA, Schroeder KW, Tremaine WJ (1987) Collagenous colitis: a clinicopathologic correlation.Mayo ClinProc 62: 665-671.
- Jawhari A, Talbot IC (1996) Microscopic, lymphocytic and collagenous colitis.Histopathology 29: 101-110.
- Giardiello FM, Lazenby AJ, Bayless TM (1995) The new colitides, Collagenous, lymphocytic, and diversion colitis.GastroenterolClin North Am 24: 717-729.
- Olesen M, Eriksson S, Bohr J, Järnerot G, Tysk C (2004) Lymphocytic colitis: a retrospective clinical study of 199 Swedish patients.Gut 53: 536-541.
- Pardi DS, Loftus EV Jr, Smyrk TC, Kammer PP, Tremaine WJ, et al. (2007) The epidemiology of microscopic colitis: a population based study in Olmsted County, Minnesota.Gut 56: 504-508.
- Sonnenberg A, Genta RM (2012) Geographic distributions of microscopic colitis and inflammatory bowel disease in the United States.Inflamm Bowel Dis 18: 2288-2293.
- Stewart M, Andrews CN, Urbanski S, Beck PL, Storr M (2011) The association of coeliac disease and microscopic colitis: a large population-based study.Aliment PharmacolTher 33: 1340-1349.
- Agnarsdottir M, Gunnlaugsson O, Orvar KB, Cariglia N, Birgisson S, et al. (2002) Collagenous and lymphocytic colitis in Iceland.Dig Dis Sci 47: 1122-1128.
- Cindoruk M, Tuncer C, Dursun A, Yetkin I, Karakan T, et al. (2002) Increased colonic intraepithelial lymphocytes in patients with Hashimoto's thyroiditis.J ClinGastroenterol 34: 237-239.
- Williams JJ, Kaplan GG, Makhija S, Urbanski SJ, Dupre M, et al. (2008) Microscopic colitis-defining incidence rates and risk factors: a population-based study.ClinGastroenterolHepatol 6: 35-40.
- Kaplan GG, Seminowich S, Williams J, Muruve D, Dupre M, et al. (2008) The risk of microscopic colitis in solid-organ transplantation patients: a population-based study.Transplantation 85: 48-54.
- Bohr J, Tysk C, Eriksson S, Abrahamsson H, Järnerot G (1996) Collagenous colitis: a retrospective study of clinical presentation and treatment in 163 patients.Gut 39: 846-851.
- Hilmer SN, Heap TR, Eckstein RP, Lauer CS, Shenfield GM (2006) Microscopic colitis associated with exposure to lansoprazole.Med J Aust 184: 185-186.
- Yu YH, Han DS, Choi EY, Park HS, Jeong JY, et al. (2012) Is use of PPIs related to increased intraepithelial lymphocytes in the colon?Dig Dis Sci 57: 2669-2674.
- Hjortswang H, Tysk C, Bohr J, Benoni C, Kilander A, et al. (2009) Defining clinical criteria for clinical remission and disease activity in collagenous colitis.Inflamm Bowel Dis 15: 1875-1881.
- Koskela RM, Karttunen TJ, Niemelä SE, Lehtola JK, Ilonen J, et al. (2008) Human leucocyte antigen and TNFalpha polymorphism association in microscopic colitis.Eur J GastroenterolHepatol 20: 276-282.
- Beaugerie L, Pardi DS (2005) Review article: drug-induced microscopic colitis - proposal for a scoring system and review of the literature.Aliment PharmacolTher 22: 277-284.
- Mäkinen M, Niemelä S, Lehtola J, Karttunen TJ (1998) Collagenous colitis and Yersinia enterocolitica infection.Dig Dis Sci 43: 1341-1346.
- Perk G, Ackerman Z, Cohen P, Eliakim R (1999) Lymphocytic colitis: a clue to an infectious trigger.Scand J Gastroenterol 34: 110-112.
- Erim T, Alazmi WM, O'Loughlin CJ, Barkin JS (2003) Collagenous colitis associated with Clostridium difficile: a cause effect?Dig Dis Sci 48: 1374-1375.
- Ung KA, Gillberg R, Kilander A, Abrahamsson H (2000) Role of bile acids and bile acid binding agents in patients with collagenous colitis.Gut 46: 170-175.
- Barmeyer C, Erko I, Fromm A, Bojarski C, Allers K, et al. (2012) Ion transport and barrier function are disturbed in microscopic colitis.Ann N Y AcadSci 1258: 143-148.
- Yen EF, Pokhrel B, Du H, Nwe S, Bianchi L, et al. (2012) Current and past cigarette smoking significantly increase risk for microscopic colitis.Inflamm Bowel Dis 18: 1835-1841.
- Fernández-Bañares F, de Sousa MR, Salas A, Beltrán B, Piqueras M, et al. (2013) Epidemiological risk factors in microscopic colitis: a prospective case-control study.Inflamm Bowel Dis 19: 411-417.
- Wagner M, Stridsberg M, Peterson CG, Sangfelt P, Lampinen M, et al. (2013) Increased fecal levels of chromogranin A, chromogranin B, and secretoneurin in collagenous colitis.Inflammation 36: 855-861.
- Akamoto S, Fujiwara M, Okano K, Suzuki Y (2014) Spontaneous perforation in collagenous colitis.Surgery 155: 198-199.
- Jessurun J, Yardley JH, Giardiello FM, Hamilton SR, Bayless TM (1987) Chronic colitis with thickening of the subepithelial collagen layer (collagenous colitis): histopathologic findings in 15 patients.Hum Pathol 18: 839-848.
- Eisen GM, Dominitz JA, Faigel DO, Goldstein JA, Kalloo AN, et al. (2001) Use of endoscopy in diarrheal illnesses.GastrointestEndosc 54: 821-823.
- Tanaka M, Mazzoleni G, Riddell RH (1992) Distribution of collagenous colitis: utility of flexible sigmoidoscopy.Gut 33: 65-70.
- Chang F, Deere H, Vu C (2005) Atypical forms of microscopic colitis: morphological features and review of the literature.AdvAnatPathol 12: 203-211.
- Neumann H, Grauer M, Vieth M, Neurath MF (2013) In vivo diagnosis of lymphocytic colitis by confocal laser endomicroscopy.Gut 62: 333-334.
- Stewart MJ, Seow CH, Storr MA (2011) Prednisolone and budesonide for short- and long-term treatment of microscopic colitis: systematic review and meta-analysis.ClinGastroenterolHepatol 9: 881-890.
- Baert F, Schmit A, D'Haens G, Dedeurwaerdere F, Louis E, et al. (2002) Budesonide in collagenous colitis: a double-blind placebo-controlled trial with histologic follow-up.Gastroenterology 122: 20-25.
- Fine KD, Lee EL (1998) Efficacy of open-label bismuth subsalicylate for the treatment of microscopic colitis.Gastroenterology 114: 29-36.
- Munck LK, Kjeldsen J, Philipsen E, Fischer Hansen B (2003) Incomplete remission with short-term prednisolone treatment in collagenous colitis: a randomized study.Scand J Gastroenterol 38: 606-610.
- Esteve M, Mahadevan U, Sainz E, Rodriguez E, Salas A, et al. (2011) Efficacy of anti-TNF therapies in refractory severe microscopic colitis.J Crohns Colitis 5: 612-618.
- Peterson TC, Tanton RT (1996). “Effect of pentoxifylline in collagenous colitis: a new therapy?” Gastroenterology 990.
- Scheidler MD, Meiselman M (2001) Use of verapamil for the symptomatic treatment of microscopic colitis.J ClinGastroenterol 32: 351-352.
- Fisher NC, Tutt A, Sim E, Scarpello JH, Green JR (1996) Collagenous colitis responsive to octreotide therapy.J ClinGastroenterol 23: 300-301.
- Tromm A, Niewerth U, Khoury M, Baestlein E, Wilhelms G, et al. (2004) The probiotic E. coli strain Nissle 1917 for the treatment of collagenous colitis: first results of an open-label trial.Z Gastroenterol 42: 365-369.
- Wildt S, Munck LK, Vinter-Jensen L (2006). “Probiotic treatment of collagenous colitis: a randomized, double-blind, placebo-controlled trial with Lactobacillus acidophilus and Bifidobacteriumanimalis subsp. Lactis,” Inflammatory Bowel Diseases 395 401.
- Pardi DS, Loftus EV Jr, Tremaine WJ, Sandborn WJ (2001) Treatment of refractory microscopic colitis with azathioprine and 6-mercaptopurine.Gastroenterology 120: 1483-1484.
- Münch A, Ignatova S, Ström M (2012) Adalimumab in budesonide and methotrexate refractory collagenous colitis.Scand J Gastroenterol 47: 59-63.
- Riaz AA, Pitt J, Stirling RW, Madaan S, Dawson PM (2000) Restorative proctocolectomy for collagenous colitis.J R Soc Med 93: 261.
Relevant Topics
Recommended Journals
- Journal of Lung Cancer Diagnosis & Treatment
- Advances in Cancer Prevention
- Breast Cancer: Current Research
- Cancer Surgery
- Immunology: Current Research
- Current Trend in Gynecologic Oncology
- Journal of Cancer Diagnosis
- Journal of Gastrointestinal Cancer and Stromal Tumors
- Cervical Cancer: Open Access
- Journal of Mucosal Immunology Research
- Journal of Oncology Research and Treatment
- Journal of Orthopedic Oncology
- Journal of Prostate Cancer
- Research and Reviews on Pathogens
Article Tools
Article Usage
- Total views: 18566
- [From(publication date):
September-2014 - Aug 20, 2025] - Breakdown by view type
- HTML page views : 13864
- PDF downloads : 4702