Research Article Open Access
Unexpected Increase in Deaths from Alzheimers, Dementia and Other Neurological Disorders in England and Wales during 2012 and 2013
Rodney P Jones*
Healthcare Analysis and Forecasting, Camberley, UK
- *Corresponding Author:
- Rodney P Jones
Healthcare Analysis and Forecasting Camberley, UK
Tel: +44 (0)1276 21061
E-mail: hcaf_rod@yahoo.co.uk
Received January 14, 2015; Accepted March 28, 2015; Published March 31, 2015
Citation: Jones RP (2015) Unexpected Increase in Deaths from Alzheimer’s, Dementia and Other Neurological Disorders in England and Wales during 2012 and 2013. J Neuroinfect Dis 6:172. doi: 10.4172/2314-7326.1000172
Copyright: © 2015 Jones RP. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Neuroinfectious Diseases
Abstract
Background: There was a large and unexpected increase in deaths due to Alzheimer’s, Dementia and certain other neurological diseases in 2012 and 2013 in England and Wales. The Office for National Statistics has suggested that the elevation of dementia to the leading cause of death in women during 2013 was simply due to changes in coding. This claim is investigated against a background of a suspected outbreak of a new type of immune modulating disease.
Methods: Cause of death statistics for England and Wales were analyzed in 2011, 2012 and 2013 using five year age bands. Change in deaths was adjusted for expected population growth.
Results: All-cause mortality in England and Wales increased relative to the expected downward trajectory in both 2012 and 2013. However deaths due to Alzheimer’s and Dementia were particularly affected in an age-specific manner, which discounts explanations based on better coding of the cause of death. Certain other neurological conditions were also affected, especially in those aged over 70 years. Deaths in the oldest old (age 95+) generally declined. The effects upon late-onset Alzheimer’s was far more pronounced in 2013 than 2012, especially in females.
Conclusion: An infectious event appears to have occurred which led to a general increase in deaths, however, the percentage increase was far greater for those suffering from neurological disorders and over the age of 70, but generally not in the oldest old. The nature and mode of action of a presumed infectious agent requires urgent research.
Keywords
Cause of death; Age; Neurological conditions; Emerging infectious diseases; Cytomegalovirus; Alzheimer’s; Parkinson’s; Dementia
Introduction
Since the early 1990’s, both deaths and age-standardized mortality rates in England and Wales have been steadily declining due to improvements in health care, immunization, housing (mainly central heating) and policies to reduce smoking, salt consumption, etc. [1,2]. In 2008 the Office for National Statistics (ONS) forecast that deaths (allcause mortality) would continue to decline until around 2016, when sheer weight of population growth and the impact of the World War II baby boom would lead to increased deaths. Compared to 2011 deaths were expected to be 0.7% lower in 2012 and 1.3% lower in 2013 [3].
However, contrary to expectation, deaths showed a sudden and large increase in early 2012 which continued through to late 2013 [4-6]. Recent research has demonstrated that this sudden onset of increased deaths showed spatial spread throughout England and Wales, between February 2011 and July 2013 (although 75% of initiation occurred beyond November 2011) [6], and is part of a longer time series of infectious-like events going back to the 1950’s and perhaps earlier. In each location deaths jump to a higher level for around 12 to 18 months before reverting back to the expected level [7-10], hence at a national level deaths appear to peak in both 2012 and 2013 calendar years. Furthermore the increased deaths were accompanied by increased medical admission to hospital, which also shows spatial spread [8-12].
However, in opposition to adults, both neonatal, perinatal and infant deaths continued to decline in 2012 and 2013 along the established time trajectory [13]. An earlier outbreak leading to a peak in deaths during 2008 provoked a large increase in GP referral for an outpatient first attendance which also showed spatial spread across the UK [14]. A similar event also led to a peak in deaths and medical admissions during 2003 [15,16]. A further outbreak in 2010 was highly granular, and the pattern of spread was such that only small increases in deaths were observed for England and Wales as a spatial unit [6].
The increase in deaths is specific to a set of diagnoses/conditions which could be broadly described as being immune-sensitive, i.e. subject to the consequences of immune imbalance which can occur in the process of inflammation, efficacy of immunization, susceptibility to infection, autoimmunity and depression, and seems to be matched by a similar set of conditions seen in increased medical admissions [17-22]. Whatever happened in 2012 appears to be best described as an infectious outbreak since it shows clear evidence of spatial spread and small area granularity [8-12]. Both these and other studies have been recently reviewed with a view to understanding how these outbreaks have gone unnoticed for so long – although prior to 2000 these outbreaks were probably incorrectly attributed to the effects of influenza [2,5].
In 2013 dementia became the highest cause of death in females, which the ONS suggested could be due to better coding [23]. This suggestion is unlikely for several reasons. Firstly, changes to the allocation of cause of death were implemented in 2011 and this resulted in a large step increase in deaths attributed to dementia [24], and there is no specific reason that coding of dementia and Alzheimer’s should improve markedly in 2012 and 2013. Secondly the increase in 2012 and 2013 was part of wider increases in all-cause mortality [1,24-27]. Finally, the changes have been shown to be single-year-of-age age specific [26] and improved coding should not be so highly age specific.
This article will investigate the gap between deaths in 2012 and 2013 versus 2011 by age and gender to investigate if changes in death due to neurological disorders are due to changes in coding or reflect a potential new type of immune-based infectious etiology.
Materials and Methods
Mortality statistics
Cause of death statistics (Series DR) for England and Wales from 2002 to 2013 were obtained from the ONS website. Cause of death was coded using the International Classification of Diseases (ICD) version 2001.2 between 2002 and 2010, and version 2010 from 2011 onward [23].
Changes in population by age band
The latest census in the UK took place in 2011, and as a result population estimates by age band in 2011, 2012 and 2013 should be reasonably accurate (within the limitations that the increased deaths in 2012 and 2013 were unanticipated). Data by age band was obtained from the ONS and the expected change in population for each band was calculated relative to 2011.
Change in deaths relative to 2011
Since all-cause mortality jumps in 2012 and stays high during 2013, the number of deaths in 2012 or 2013 has been compared to the number of deaths in 2011 plus the square root of the number of deaths in 2011, i.e. (2012 or 2013)/(2011 + √2011). This calculation was performed for all age bands, and the expected change in the population was then subtracted to give a growth-adjusted increase.
The square root of the number of deaths in 2011 is an adjustment for the possibility that the number of deaths in this year may be low due to Poisson-based statistical randomness. By definition, the standard deviation of a Poisson distribution is equal to the square root of the average, and hence, this adjustment adds one standard deviation to the total deaths in 2011. This adjustment guards against over-estimation but can lead to under-estimation of the percentage increase. However, the effect either way is only significant when the numbers (in total or per age band) are less than 1,000 and especially so for less than 300.
All data is available in the supplementary material Table S1.
Results
Adjustment for the ageing population
Growth in the number of elderly persons relative to 2011 could potentially skew the calculation of percentage change in deaths, and the expected change in the number of living persons by age band relative to 2011 is given in Table 1. Changes relative to 2011 are generally modest, however, the World War II baby boom accounts for the higher percentage increases seen for ages 65-69, and a combination of the World War I baby boom plus the generally ageing population accounts for the large increases seen in the 90+ age group. These changes have been used to adjust all subsequent calculated increases (called the ‘growth-adjusted change’).
| Age Group | Male | Female | ||
|---|---|---|---|---|
| 2012 | 2013 | 2012 | 2013 | |
| 50-54 | 2.9% | 5.5% | 2.9% | 5.8% |
| 55-59 | 1.6% | 3.6% | 1.6% | 3.6% |
| 60-64 | -4.9% | -7.2% | -4.6% | -6.9% |
| 65-69 | 9.2% | 14.1% | 8.9% | 13.8% |
| 70-74 | 1.0% | 3.7% | 0.9% | 3.4% |
| 75-79 | 2.1% | 4.9% | 1.1% | 2.9% |
| 80-84 | 2.7% | 4.7% | 1.0% | 1.3% |
| 85-89 | 2.6% | 4.9% | -0.8% | -1.1% |
| 90+ | 8.5% | 13.8% | 4.7% | 6.7% |
Table 1: Forecast changes in population relative to 2011.
Increase in all-cause mortality
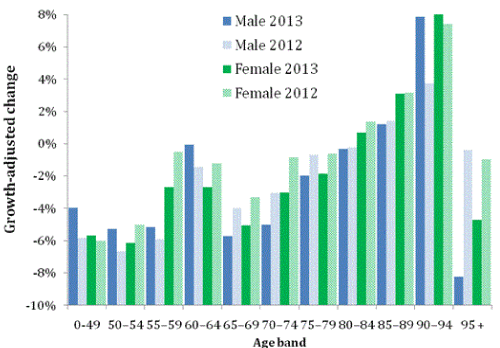
To provide a benchmark to assess whether deaths have risen in dementia, Alzheimer’s and other neurological disorders, Figure 1 presents the situation for the change in all-cause mortality between 2012 or 2013 versus 2011. Deaths exceed 6,000 per annum for all agegender combinations above age 54, and changes are therefore highly statistically significant at well beyond the 95% confidence interval.
As is expected, Figure 1 shows that most age groups show a reduction due to the expected decline in deaths relative to 2011 as forecast by the ONS [3]. Deaths do, however, show an age dependent increase especially above the age of 70 and also a significant increase specific to males aged 60-64. There is a decline in deaths for the oldest old (age 95+). Maximum increase of around +8% occurs for both males and females aged 90-94 in 2013.
Changes in coding
The coding of death in England and Wales has undergone two changes in recent times with the switch from ICD-9 to ICD-10 occurring on the 1st of January 2001, and a further change from 1st January 2011 with a switch from ICD-10 v2001.2 to v2010 [23]. This resulted in a 32% increase in the number of deaths with an underlying cause coded to ‘Mental and Behavioral Disorders’. A large proportion of this increase was caused by correction to the coding of vascular dementia which was previously assigned to the underlying cause of cerebrovascular disease (I67.9) in ICD–10 v2001.2, but was corrected to vascular dementia (F01) in ICD–10 v2010. A number of dementia deaths were also previously coded as N39.0 (urinary tract infection, site not specified). Diseases of the Nervous System also experienced a 7% increase due to a range of changes covering other chapters, with the biggest contribution from recoding of respiratory deaths which were considered secondary to the primary cause of Alzheimer’s, etc.
Long-term trends
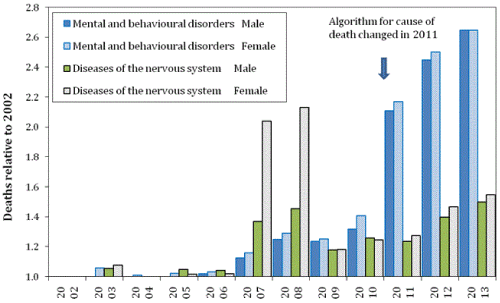
To evaluate the hypothesis that the increase in deaths due to dementia were due to changes in coding, the trend in deaths for ICD Chapters F (Mental and behavioral disorders, F00-F99, with Dementia as the leading cause of death) and Chapter G (Diseases of the nervous system, G00-G99, with Alzheimer’s and Parkinson’s as the leading cause of death) between 2002 and 2013 are displayed in Figure 2 Specific changes can be noted during the years when the infectious outbreaks have been proposed to occur. Hence a relatively modest increase in 2003, a larger increase in 2007/2008 (especially for women in Diseases of the Nervous System), and further increases in 2012/2013 for both ICD chapters. A small increase in 2010 seems to be associated with a lesser and more spatially granular outbreak of the proposed agent in this year [6].
Specific conditions
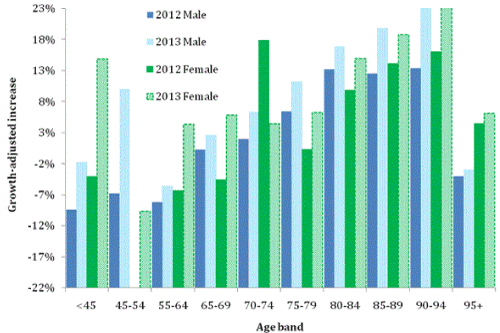
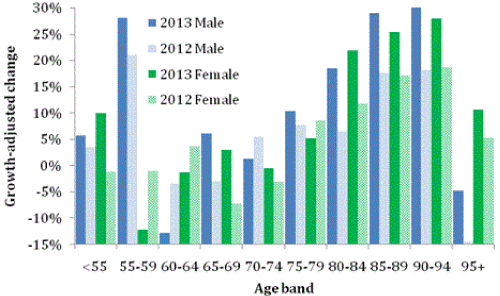
Figure 3 shows deaths in ICD Chapter F (mental and behavioral disorders, F00-F99). In Chapter F, dementia accounts for more than 99.8% of deaths, however, the non-dementia deaths (mostly due to the use of psychoactive substances), are almost exclusively below age 55 where both males and females undergo the greatest increase in 2013. Hence we see a very large increase in deaths relative to Figure 1 which is associated with the dementia age bands. Maximum increase is around +20% for both males and females aged 90-94 compared to +8% for allcause mortality. An effect specific to dementia has therefore occurred.
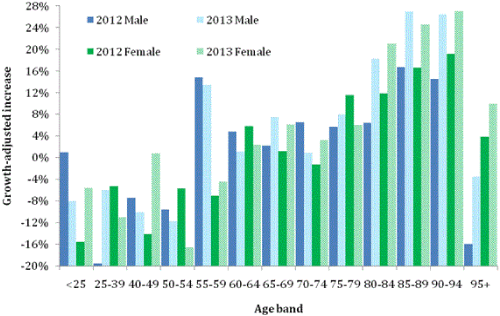
Figure 4 shows deaths for ICD Chapter G (diseases of the nervous system, G00-G99). In Chapter G, Parkinson’s plus Alzheimer’s accounts for 58% of male and 68% of female deaths. However this is less than 5% of deaths for ages under 55, but rising to over 80% for those aged over 85 years [28]. Hence in Figure 3 there are general reductions for the other nervous system disorders in those aged under 55, but large increases for ages specific to Alzheimer’s and Parkinson’s. Largest increases occur in 2013 in the age range 85-94 in both males and females.
Finally Table 2 investigates the relationship between disorders where a large proportion of deaths are for those aged 80+ and a high percentage increase in 2012 and 2013. Two measures of the percentage increase are given, namely, the raw change in deaths relative to 2011 (without adjustment for population change) and the weighted increase (change across all age bands weighted by deaths in each age band). Table 2 also gives the total number of female deaths in 2012 as an indication of those diagnoses which have the highest number of deaths. Table 2 contains all diagnoses with more than 400 deaths per annum, and also illustrates the relative difficulty of demonstrating statistically significant changes for the majority of diagnoses where the numbers per age band are far too small, i.e. even at 400 deaths the largest age band will only contain 200 deaths giving ± 14% as the minimum for the 95% confidence interval for Poisson-based uncertainty in the percentage change. Table S2 (Supplement) also shows that the only diagnosis experiencing a large increase in the 95+ age group is vascular dementia in females during both 2012 and 2013. Also that Alzheimer’s with late onset shows a distinct maximum increase in 2013 in both males and females aged 85-89, but especially in females. A time-lag may be involved in this latter observation.
| Year | ICD Code | Description | Deaths | Female | Male | |||
|---|---|---|---|---|---|---|---|---|
| All Ages | 80+ | All | Weighted | All | Weighted | |||
| 2012 | F01,F03 | Dementias | 23,787 | 91% | 14.4% | 11.4% | 15.5% | 10.0% |
| 2013 | F01,F03 | Dementias | 25,216 | 91% | 21.3% | 17.4% | 24.7% | 16.0% |
| 2012 | F01 | Vascular dementia | 5,390 | 90% | 17.1% | 13.5% | 20.3% | 14.4% |
| 2013 | F01 | Vascular dementia | 5,758 | 90% | 25.1% | 21.3% | 26.1% | 17.1% |
| 2012 | F03 | Unspecified dementia | 18,397 | 91% | 13.3% | 10.1% | 12.9% | 7.1% |
| 2013 | F03 | Unspecified dementia | 19,458 | 91% | 19.8% | 15.7% | 23.5% | 14.1% |
| 2012 | G10-G13 | Affecting the central nervous system | 1,124 | 26% | 5.3% | -0.7% | 10.8% | 3.5% |
| 2013 | G10-G13 | Affecting the central nervous system | 1,098 | 27% | 2.9% | -4.9% | 15.2% | 5.6% |
| 2012 | G12 | Spinal muscular atrophy and syndromes | 976 | 27% | 3.5% | -3.0% | 11.5% | 4.1% |
| 2013 | G12 | Spinal muscular atrophy and syndromes | 943 | 28% | 0.0% | -7.9% | 16.3% | 6.4% |
| 2012 | G20-G26 | Extrapyramidal and movement disorders | 1,802 | 74% | 12.5% | 8.6% | 15.6% | 9.8% |
| 2013 | G20-G26 | Extrapyramidal and movement disorders | 1,813 | 76% | 13.2% | 9.2% | 23.0% | 14.7% |
| 2012 | G20 | Parkinson's disease | 1,778 | 75% | 12.4% | 8.4% | 15.7% | 10.0% |
| 2013 | G20 | Parkinson's disease | 1,792 | 77% | 13.3% | 9.3% | 23.2% | 14.9% |
| 2012 | G30-G32 | Diseases of nervous system | 6,678 | 83% | 16.9% | 10.4% | 13.0% | 7.2% |
| 2013 | G30-G32 | Diseases of nervous system | 7,329 | 82% | 28.3% | 18.0% | 27.1% | 19.1% |
| 2012 | G30 | Alzheimer's disease | 6,086 | 85% | 17.2% | 13.4% | 14.0% | 7.9% |
| 2013 | G30 | Alzheimer's disease | 6,634 | 85% | 27.7% | 22.6% | 29.7% | 20.8% |
| 2012 | G30.1 | Alzheimer's disease with late onset | 1,170 | 97% | 13.1% | 7.7% | -2.3% | -11.9% |
| 2013 | G30.1 | Alzheimer's disease with late onset | 1,267 | 98% | 22.5% | 16.3% | 19.1% | 5.7% |
| 2012 | G30.9 | Alzheimer's disease, unspecified | 4,901 | 82% | 17.4% | 13.6% | 15.5% | 9.5% |
| 2013 | G30.9 | Alzheimer's disease, unspecified | 5,358 | 82% | 28.3% | 23.0% | 30.3% | 21.6% |
| 2012 | G31 | Other nervous system | 592 | 57% | 9.9% | 1.4% | 6.3% | 0.1% |
| 2013 | G31 | Other nervous system | 695 | 61% | 29.0% | 17.7% | 14.9% | 5.5% |
| 2012 | G35-G37 | Demyelinating diseases of the CNS | 788 | 16% | 9.9% | 1.8% | 9.1% | -0.2% |
| 2013 | G35-G37 | Demyelinating diseases of the CNS | 747 | 15% | 4.1% | -4.9% | -7.9% | -14.3% |
| 2012 | G35 | Multiple sclerosis | 779 | 16% | 11.3% | 3.2% | 7.7% | -1.3% |
| 2013 | G35 | Multiple sclerosis | 727 | 15% | 3.9% | -5.3% | -8.5% | -14.6% |
| 2012 | G40-G47 | Episodic and paroxysmal disorders | 560 | 39% | 5.6% | -1.1% | -1.4% | -6.0% |
| 2013 | G40-G47 | Episodic and paroxysmal disorders | 557 | 39% | 5.0% | -5.4% | -2.0% | -9.2% |
| 2012 | G40 | Epilepsy | 450 | 33% | 6.4% | 0.1% | -3.8% | -7.7% |
| 2013 | G40 | Epilepsy | 437 | 30% | 3.3% | -6.3% | -3.8% | -9.1% |
Table 2: Percentage increase in deaths relative to 2011 for a range of neurological conditions. The count of female deaths and the proportion of deaths for those aged 80+ are provided as a reference point. The weighted increase is the average of the change in deaths in each age band weighted according to the number of deaths in that age band. See Supplementary material for full details of change by age band.
Discussion
With over 1,400 known human pathogens (of which over 220 are viruses) [29], the possibility of new disease types is an area of increasing interest. Recent research has explored the evidence for a new type of immune-based disease and how its effects upon death and increased medical admissions for particular conditions have been repeatedly misdiagnosed [2,29-31].
Outbreaks of this disease have gone unnoticed for two key reasons, Firstly relatively slow spread within a country dilutes the effect observed at whole-country level, and secondly the effect upon deaths or hospital admissions occurs across a 12 to 18 month period. Hence the outbreaks evade all current disease surveillance methodologies which tend to look for influenza-type large and sudden spikes in deaths or admissions [5]. Preliminary evidence suggests that an outbreak of this disease prior to a seasonal influenza epidemic acts to potentiate the effect of influenza, and in this scenario the larger influenza peak is wrongly attributed to influenza alone [2].
Re-analysis of monthly deaths during the last three outbreaks of this disease across Europe, which occurred in the interval 2002 to 2013, reveals an estimated 476,000 deaths during each outbreak [2]. Due to the relatively slow spread within each country this is assumed to be an underestimate.
An effect of these outbreaks upon Alzheimer’s, Parkinson’s and dementia during 2012 has already been demonstrated [20], however this study makes use of the fact that the increased deaths remained high in both 2012 and 2013, due to slower spread of the agent throughout the entire UK during both of these years [6]. Preliminary evidence suggests that a further outbreak may also have occurred in 2014 [32], but it will take several years for all the relevant data to become available to fully investigate this possibility. A similar series of short-spaced multiple outbreaks occurred in 1993, 1996, 1999 and 2002 [2,6].
The key point is that a large infectious event has occurred which has all the characteristics of an outbreak of a previously unrecognized persistent agent.
The reduction in deaths forecast by the ONS
As has been demonstrated in Figure 1, for most age bands below age 75 (except for females age 5-15), the number of deaths did indeed decline in 2012 and 2013 relative to 2011. However specific increases occurred in the older age groups. In 2013 over 50% of deaths in England & Wales occurred above age 75 with the numerical peaks between ages 76 to 89 (male) and 83 to 93 (female) accounting for 45% of deaths. Any agent specifically targeting these older age groups will have the potential to create the large changes in all-cause mortality seen to occur in 2012 and 2013.
However all other figures and Table S2 (in the Supplementary material) show far higher age-dependent percentage increases in deaths than that observed in Figure 1, and are therefore indicative of condition-specificity with Alzheimer’s and Parkinson’s in particular
Statistical Significance
Statistical significance in this study has been assured in two ways. Firstly, all changes have been adjusted for estimated changes in population, under the assumption that more persons in an age band implies greater opportunity for death. In a time of generally increasing life expectancy this adjustment is an over-estimate of the potential impact on deaths, hence, the calculated percentage change in the various figures is likely to be conservative. This conservative nature has been further reinforced by adding one-standard deviation to deaths in the base year to avoid any possibility that this year was low due to statistical randomness.
Strictly speaking the one standard deviation should have been calculated based on the total deaths for each condition and this should then have been apportioned to each age band. However calculation based on the deaths in each age band only accentuates the conservative nature of all calculations.
In addition, this study has avoided the use of the usual measures of statistical confidence for the simple reason that the actual effect of these outbreaks is known to be highly single-year-of-age specific, and consequently the use of five year age bands can give inconclusive results depending on the disposition of peaks and troughs within an age band [7-10,26]. Further research needs to extend the analysis to explore these single-year-of-age effects more fully.
Evidence for different strains of the same agent
This single-year-of-age specificity has been suggested to be due to ‘original antigenic sin’ [26], the phenomena by which the immune system is primed to respond (optimally or sub optimally) to new antigens via past exposure to antigens resident on different strains of the same infectious agent [33]. The potential impact of different strains of the same agent is strongly implied in Figure 2 where the 2003 outbreak provokes a modest increase in death among those suffering from neurological disorders. The 2007/2008 outbreak provokes a very strong effect, especially upon females, while the 2010 outbreak has a modest effect, and is then followed by a strong effect against both males and females in the 2012/2013 outbreak. The analysis presented here is therefore designed to point researchers to the existence of a very large infectious-like event which requires further study, and to the fact that individuals within certain age bands are affected more than others and that gender can also moderate the effect of each outbreak.
Figure 2 is of particular interest because it covers three outbreaks of the proposed agent. Each outbreak exhibits a high degree of singleyear- of-age specificity for both deaths and admissions, and this has been interpreted as evidence for immune priming by a series of infections with different strains of the same agent [7-10,23]. Infection by different strains of the same agent would explain why the outbreaks commencing in 2002, 2007 and 2012 show such a variable response between the two ICD chapters and gender.
Evidence for time-lags
Most of the figures and Table S2 (supplementary material) show evidence for time lags specific to gender and condition, and this observation only confirms previous studies showing that theseds outbreaks have time-based effects [2,8], presumably via idmmune manipulation, and the exact balance of inflammatory andddd other forces required for ultimate exacerbation of existing conditions to the point where the person dies.
Time-lags are also likely due to the complex patterns of spatial spread exhibited by the agent. Spread across the entire UK takes around two years when measured at Local Authority Level [6,12], while spread within a Local Authority itself takes around 18 months [7-10]. Spread across Europe occurs across similar time spans [2]. Hence the observed pattern of effect very much depends on the geographic unit (in this case England and Wales) and the time frame (monthly versus calendar year) over which the effect is documented.
Roles for cytomegalovirus
The issue which requires resolution is the exact involvement of the infectious agent. In this respect, it has been suggested that the ubiquitous herpes virus, cytomegalovirus (CMV), may be implicated in these outbreaks, either via the introduction of a new strain or reactivation in the presence of another agent [15,19,21-22,27,31].
The evidence for CMV rests mainly on its ability to substantially increase all-cause mortality [15,19,21,27] and that the spectrum of conditions associated with both the increase in deaths and medical admissions mirror the known clinical conditions where CMV is most active [25,29,31,34,35]. Future research will undoubtedly clarify these issues. Infections involving the nervous system are one of the more common manifestations of CMV [25,32]. If CMV is the agent, the question remains as to whether the increase in death is potentiated by a primary infection (perhaps by exposure to a new CMV strain), or was CMV merely taking opportunistic advantage of infection with another agent?
It has been noted that those suffering from neurological diseases will already have weakened immune function due to general immobility and lack of exposure to sunlight (and hence low levels of vitamin D) [22], which will render them more susceptible to infection in general. The smaller peak in 2010 (Figure 2), which involved the tail end of the swine-flu influenza epidemic, indicates that other infectious agents will also be circulating at the time of these outbreaks and may also take opportunistic advantage of whatever immune impairments are afforded by the outbreaks.
Gender specific effects
Differences between the two genders are difficult to interpret given the very high single-year-of-age specificity of these outbreaks [7-10,23]. This age specificity can obscure the exact effects when using standard 5 year age bands, although on this occasion the national data is only presented in this manner. Figure 2 has, however, demonstrated far higher gender specificity during the 2007/2008 outbreak. Further detailed research is required regarding these effects, especially in other countries. One suggestion has been to align the start date for each outbreak across Local Authority areas in order to amplify the effect of the outbreak [7], and thereby tease out the hidden gender, single-yearof- age, and time-lag issues specific to each condition.
Insight from other studies
A fascinating development has been the observation that natural killer (NK) cells are activated in the asymptomatic stages of Alzheimer’s, which suggests an active immune response against a presently undefined ‘aggression’ [34]. In this respect the suggested involvement of CMV is somewhat apposite.
Infants and the oldest old
Some comments regarding the situation with infants and the oldest old are warranted. While mortality in infants appears to be unaffected by these outbreaks, it is already known that there is a significant increase in non-fatal admission of infants to hospital due to infectious conditions during these outbreaks, i.e. immune-mediated resistance to certain infectious agents appears to be targeted during the outbreaks [31]. A specific CD4 T cell impairment toward CMV exists in young children [31].
A potential explanation based on CMV is also possible for the oldest old who are known to have a higher incidence of strict immune control of CMV infection [36]. Premature death in those members of the population who are CMV seropositive appears to enrich the oldest old with individuals who may be resistant to CMV infection or CMV-reactivation. In the elderly (aged 60-80), those who are CMV seropositive have double the level of C-reactive protein (CRP) to their non-CMV infected counterparts indicating a high background inflammatory state [37]. Factors such as these are likely to explain the general decline in deaths in 2012 and 2013 observed in the 95+ age group in Figure 3-5. CMV-mediated inflammation is also known to be associated with both cognitive and functional decline in the elderly [38]. CMV therefore remains a pathogen of interest in these outbreaks, although complex interaction with different CMV strains, other pathogens and human genetic variants are likely [21-22,27].
Conclusion
While changes in the coding of the cause of death introduced in 2011 resulted in a step-increase in deaths for Alzheimer’s and dementia, it cannot explain the unexpected surge in deaths in 2012 and 2013. This surge in deaths was accompanied by the spatial spread of a presumed unidentified infectious agent. The elevation of dementia to the leading cause of deaths in females in 2013 was the outworking of this infectious-like event, and the explanation offered by the ONS lacks credible evidence.
There is now a significant body of evidence to suggest that a new type of disease outbreak is occurring which leads to very large increases in deaths among those already suffering from certain neurological disorders. The exact agent responsible needs to be urgently identified. Given the higher susceptibility of those suffering from neurological disorders, especially in the age range 75 to 94, it is suggested that these represent a defined group where research efforts can be focused.
Indeed the possibility needs to be explored that outbreaks of this agent may also lead to the initiation of neurological disorders in susceptible individuals [31].
References
- Jones R (2015) A new type of infectious outbreak? SMU Medical Journal 2: 19-25.
- Jones R (2015) Recurring outbreaks of an infection apparently targeting immune function, and consequent unprecedented growth in medical admission and costs in the United Kingdom: A review. Brit J Med Medical Res 6: 735-770.
- Office for National Statistics (2009) 2008-based national population projections.
- Jones R (2013) An unexplained increase in deaths during 2012. Brit J Healthcare Manage 19: 248-253.
- Jones R (2013) Analysing excess winter mortality: 2012/13. Brit J Healthcare Manage 19: 601-605.
- Jones R (2015) A previously uncharacterized infectious-like event leading to spatial spread of deaths across England and Wales: Characteristics of the most recent event and a time series for past events. Brit J Med Medical Res 5: 1361-1380.
- Jones R, Beauchant S (2015) Spread of a new type of infectious condition across Berkshire in England between June 2011 and March 2013: Effect on medical emergency admissions. Brit J Med Medical Res 6: 126-148.
- Jones R (2015) Unexpected and disruptive changes in admissions associated with an infectious-like event experienced at a hospital in Berkshire, England around May of 2012. Brit J Med Medical Res 6: 56-76.
- Jones R (2014) Infectious-like spread of an agent leading to increased medical hospital admission in the North East Essex area of the East of England. Biomedicine International 5: in press
- Jones R (2014) Infectious-like Spread of an Agent Leading to Increased Medical Admissions and Deaths in Wigan (England), during 2011 and 2012. Brit J Med Medical Res 4: 4723-4741.
- Jones R (2015) Unexplained infectious events leading to deaths and medical admissions. Brit J Healthcare Manage 21: 46-47.
- Jones R (2013) A recurring series of infectious-like events leading to excess deaths, emergency department attendances and medical admissions in Scotland. Biomedicine International 4: 72-86.
- Office for National Statistics (2014) Statistical bulletin: Deaths Registered in England and Wales, 2013.
- Jones R (2012) Increasing GP referrals: collective jump or infectious push? Brit J Healthcare Manage 18: 487-495.
- Jones R (2012) Diagnoses, deaths and infectious outbreaks. Brit J Healthcare Manage 18: 539-548.
- Jones RP (2010) The case for recurring outbreaks of a new type of infectious disease across all parts of the United Kingdom. Med Hypotheses 75: 452-457.
- Jones RP (2010) Can time-related patterns in diagnosis for hospital admission help identify common root causes for disease expression? Med Hypotheses 75: 148-154.
- Jones R (2013) Trends in elderly diagnoses: links with multi-morbidity. Brit J Healthcare Manage 19: 553-558.
- Jones R (2015) Roles for cytomegalovirus in infection, inflammation and autoimmunity, Infection and Autoimmunity (2ndedn), Elsevier, Amsterdam.
- Jones RP, Goldeck D (2014) Unexpected and unexplained increase in death due to neurological disorders in 2012 in England and Wales: is cytomegalovirus implicated? Med Hypotheses 83: 25-31.
- Jones R (2014) A Study of an Unexplained and Large Increase in Respiratory Deaths in England and Wales: Is the Pattern of Diagnoses Consistent with the Potential Involvement of Cytomegalovirus? Brit J Med Medical Res 4: 5179-5192.
- Jones R (2014) An unexpected increase in adult appendicitis in England (2000/01 to 2012/13): Could cytomegalovirus (CMV) be a risk factor? Brit J Med Medical Res 5: 579-603.
- Office for National Statistics (2014) Statistical bulletin: Deaths Registered in England and Wales (Series DR), 2013.
- Office for National Statistics (2011) Statistical Bulletin. Results of the ICD-10 v2010 bridge coding study, England and Wales, 2009. London: ONS, 2011.
- Jones R (2014) Increased deaths in 2012: which conditions? Brit J Healthcare Manage 20: 45-47.
- Jones R (2014) Unexpected single-year-of-age changes in the elderly mortality rate in 2012 in England and Wales. Brit J Med Medical Res 4: 3196-3207.
- Jones R (2013) Recurring outbreaks of a subtle condition leading to hospitalization and death. Epidemiology: Open access 4: 137.
- Public Health Wales (2013) Influenza.
- Woolhouse ME, Gowtage-Sequeria S (2005) Host range and emerging and reemerging pathogens. See comment in PubMed Commons below Emerg Infect Dis 11: 1842-1847.
- Jones RP (2010) Unexpected, periodic and permanent increase in medical inpatient care: man-made or new disease? Med Hypotheses 74: 978-983.
- Jones R (2013) Could cytomegalovirus be causing widespread outbreaks of chronic poor health?, Hypotheses in Clinical Medicine, Nova Science Publishers Inc
- Jones R (2015) Has the UK experienced a four-in-a-row epidemic? Brit J Healthcare Manage 21: in press.
- Pan K (2011) Understanding original antigenic sin in influenza with a dynamical system. PLoS One 6: e23910.
- Le Page A, Bourgade K, Lamoureux J, Frost E, Pawelec G, et al. (2015) NK Cells are Activated in Amnestic Mild Cognitive Impairment but not in Mild Alzheimer's Disease Patients. J Alzheimers Dis.
- Rafailidis PI, Mourtzoukou EG, Varbobitis IC, Falagas ME (2008) Severe cytomegalovirus infection in apparently immunocompetent patients: a systematic review. Virol J 5: 47.
- Derhovanessian E, Maier AB, Hähnel K, Zelba H, de Craen AJ, et al. (2013) Lower proportion of naïve peripheral CD8+ T cells and an unopposed pro-inflammatory response to human Cytomegalovirus proteins in vitro are associated with longer survival in very elderly people. (Dordr) 35: 1387-1399.
- Frasca D, Diaz A, Romero M, Landin AM, Blomberg BB (2015) Cytomegalovirus (CMV) seropositivity decreases B cell responses to the influenza vaccine. Vaccine 33: 1433-1439.
- Vescovini R, Biasini C, Telera AR, Basaglia M, Stella A, et al. (2010) Intense antiextracellular adaptive immune response to human cytomegalovirus in very old subjects with impaired health and cognitive and functional status. J Immunol 184: 3242-3249.
Relevant Topics
- Bacteria Induced Neuropathies
- Blood-brain barrier
- Brain Infection
- Cerebral Spinal Fluid
- Encephalitis
- Fungal Infection
- Infectious Disease in Children
- Neuro-HIV and Bacterial Infection
- Neuro-Infections Induced Autoimmune Disorders
- Neurocystercercosis
- Neurocysticercosis
- Neuroepidemiology
- Neuroinfectious Agents
- Neuroinflammation
- Neurosyphilis
- Neurotropic viruses
- Neurovirology
- Rare Infectious Disease
- Toxoplasmosis
- Viral Infection
Recommended Journals
Article Tools
Article Usage
- Total views: 15739
- [From(publication date):
May-2015 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 11064
- PDF downloads : 4675