Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
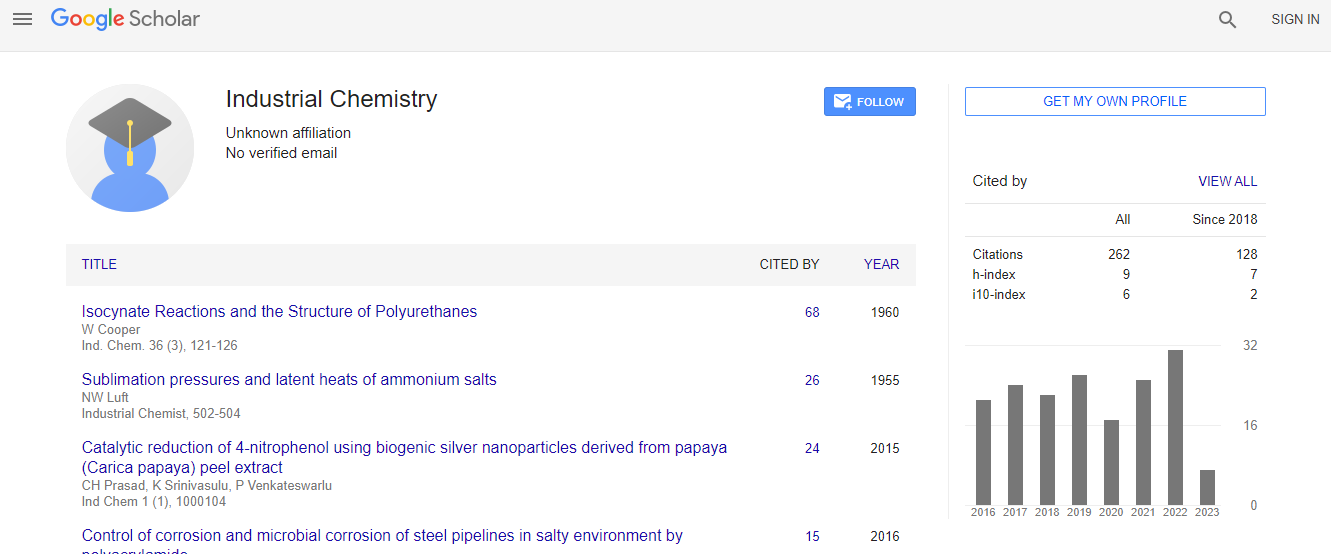
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
Acoustic and 1H NMR responses of an acidic nuclear extractant with some polar liquids
International Conference on Industrial Chemistry
Biswajit Dalai
Eastern Academy of Science and Technology, India
Posters & Accepted Abstracts: Ind Chem
Abstract
The ultrasonic velocity, density and viscosity of an acidic nuclear extractant namely di (2-ethylhexyl) phosphoric acid (D2EHPA) and its binary mixtures with three monocarboxylic acids viz., acetic, propionic and n-butyric were measured at temperature, T = 303.15 K and at pressure, P = 0.1MPa. The experimental data were utilized to compute relaxation time, molecular association constant, deviations in intermolecular free length, acoustic impedance, free volume and change in entropy, excess Gibb��?s energy of activation of viscous flow, excess enthalpy in the entire mole fraction range of D2EHPA. The excess/deviation functions were fitted to Redlich-Kister type polynomial equation to estimate binary coefficients and standard errors between experimental and calculated data. The nature of these functions was utilized to speculate the nature of molecular interaction between component molecules of all studied binary mixtures. Furthermore, 1H NMR spectra of pure D2EHPA and its binary mixtures with the monocarboxylic acids were used to assess molecular interactions between components of liquid mixtures at microscopic level and to corroborate with the results obtained from bulk properties.Biography
Biswajit Dalai has completed his PhD from Utkal University, India. He is working as Assistant Professor in Department of Physics, Eastern Academy of Science and Technology, Bhubaneswar, India. He has published more than 12 papers in reputed journals.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi