Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
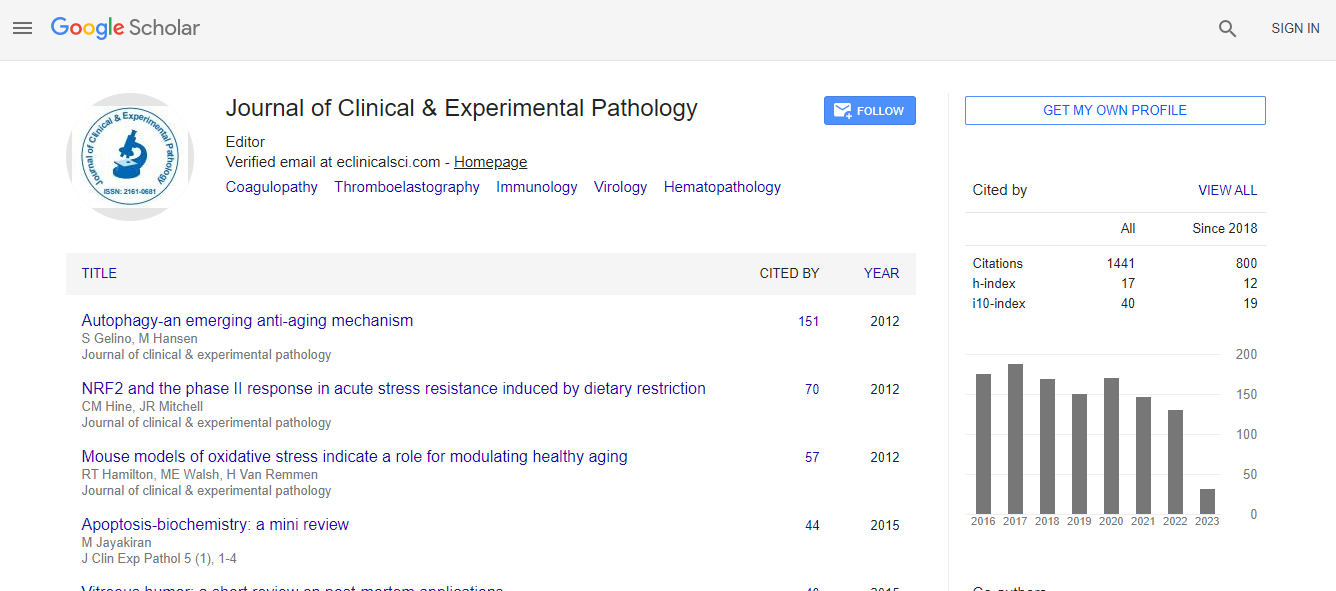
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Alveolar destruction and its therapeutic targeting
International Conference on Pathology
Vera P. Krymskaya
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
Pulmonary lymphangioleiomyomatosis (LAM), a rare lung disease affecting predominantly women of childbearing age, manifests by neoplastic growth of atypical smooth muscle-like cells, lung cyst formation, obstruction of lymphatics, and spontaneous pneumothoraces. LAM is associated with mutational inactivation of the tumor suppressor genes tuberous sclerosis complex 1 (TSC1) and TSC2. The mechanisms of cystic lung destruction, however, are not understood and potential treatment options to target destruction of lung parenchyma in LAM remain to be established. Here, we report that growth of TSC2-null lesions promotes destruction of alveoli. Lungs with TSC2-null lesions show elevated elastic fiber degradation and increased levels of MMP-2, MMP-3 and MMP-9 in the bronchoalveolar lavage suggesting that alveolar destruction and airspace enlargement are due to MMP expression caused by TSC2 loss. Further, TSC2-null lesions expressing VEGF-D show increased lymphangiogenesis detected by LYVE-1 immunostaining. Importantly, the combination of rapamycin and simvastatin treatment prevented lung TSC2-null lesion growth, alveolar space enlargement, and attenuated MMP-2, MMP-3 and MMP-9 expression. Further, although both simvastatin and rapamycin inhibited tumor growth, only simvastatin attenuated alveolar space enlargement. Collectively, our data demonstrate that TSC2-null tumor growth in mouse lungs promote alveolar destruction and airspace enlargement associated with elevated MMP expression, VEGF-D protein levels, and increased lymphangiogenesis. We also show that simvastatin and rapamycin treatment prevented both tumor growth and airspace enlargement in TSC2-null murine model of LAM, thus demonstrating beneficial effect of combined treatment and suggesting a potential promise for combinational therapeutic intervention in diseases with TSC2 dysfunction such as LAM.Biography
Vera P. Krymskaya, Ph.D., Associate Professor of Medicine, has dedicated the last several years of her career to combating LAM. Krymskaya?s lab was responsible for the breakthrough step of discovering the function of the TSC2 gene. Dr. Krymskaya linked mutational inactivation of TSC2 in human LAM cells to the constitutive activation of mTORC1 and abnormal LAM cell growth. Dr. Krymskaya has also advanced translational LAM research by demonstrating that rapamycin inhibits LAM cell growth. This discovery identified rapamycin as a promising therapeutic strategy for LAM patients, and paved the way for rapamycin clinical trials worldwide.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi