Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
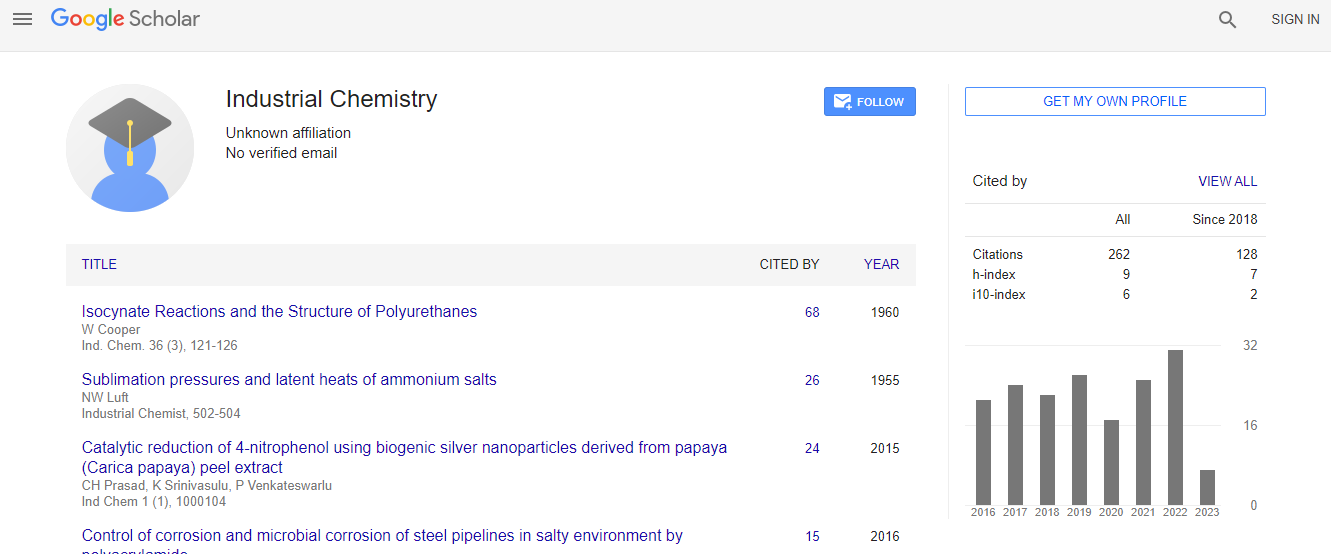
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
Chirality induction to polymers and oligomers using circularly polarized light through enantiomerselective photo excitation
17th International Conference on Industrial Chemistry and Water Treatment
Tamaki Nakano
Hokkaido University, Japan
Keynote: Ind Chem
Abstract
Interactions between substances and light play important roles in life. One of the most important aspects is photosynthesis by plants and other organisms. In photosynthesis, sugars and molecular oxygen are produced from carbon dioxide and water where photon energy drives the reaction. Moreover, in artificial chemical synthesis, light promotes various reactions that are not driven by thermal energy. Further, in reactions where chiral compounds are produced, non-racemic products can be obtained using circularly polarized light (CPL). We recently reported that a preferred-handed helical conformation is induced for a linear polyfluorene derivative (poly (9,9-dioctylfluorene-2,7-diyl [PDOF]) in the solid state when the polymer is irradiated with CPL. The mechanism of this chirality induction involves a twisted-coplanar transition (TCT) of an aromatic��? aromatic junction in the polymer where one of the enantiomeric, right- and left-handed twists is preferentially excited into the coplanar conformation. TCT through photo excitation was first predicted for biphenyl through theoretical calculations. In the CPL-driven helix formation of the linear polyfluorene derivative, strong inter-chain interactions were important in effectively induce chirality, which in turn implicates that polymers with weak inter-chain interactions cannot be subjected to the CPL method. This point of the CPL method was overcome using aid molecules which reinforce or simulate inter-chain interaction; a star-shaped fluorene oligomer having only weak inter-chain interactions was successfully made optically active. In addition, photo-transformation of 1,10-bi(2-naphthol) (BINOL) was studied.Biography
Tamaki Nakano received his BSc (1986), MSc (1988), and PhD (1991) degrees from Osaka University, Japan. He has joined Nagoya University as an Assistant Professor (Prof. Yoshio Okamoto’s group) in 1990 and was promoted as Associate Professor in 1998. During his appointment at Nagoya University, he also served as a Visiting Scientist at Cornell University (host: Prof. Dotsevi Y Sogah, 1993–1994). In 1999, he moved to Nara Institute of Science and Technology (NAIST) as an Associate Professor. He was appointed as a Professor of Hokkaido University in 2006 and is currently supervising his research group in Institute for Catalysis.
Email:tamaki.nakano@cat.hokudai.ac.jp

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi