Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
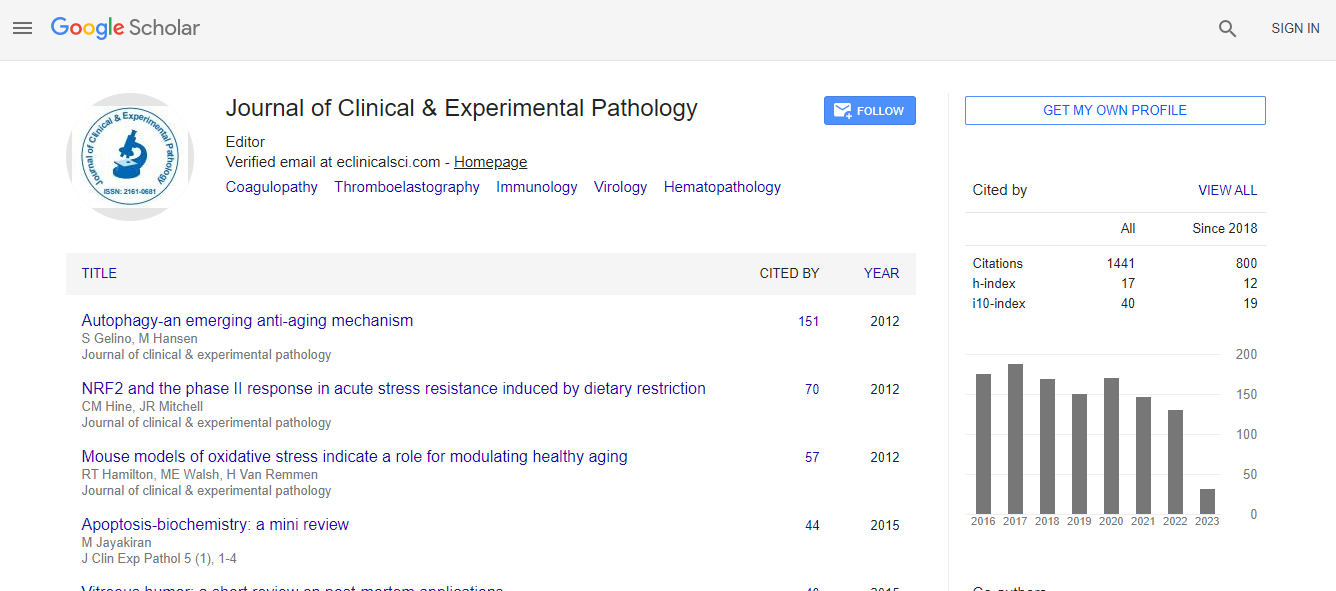
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Contemporary genomic studies in hematologic pathology: Utility of next generation sequencing in clinical evaluation of myeloid and lymphoid malignancies
13th International conference on Pathology and Molecular Diagnosis
Bevan Tandon
Molecular Pathology Laboratory Network, Inc., USA
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
Next generation sequencing (NGS) methodologies are emerging as an extremely valuable adjunct in the clinical diagnostic evaluation of hematologic cancers. Simultaneous assessment for prognostic or therapeutically predictive mutations across numerous disease relevant genes can be easily accommodated by clinical targeted NGS panels, facilitating significant reductions in labor, cost, and turnaround time for clinical reporting. Multiplex targeted NGS panels also eliminate reliance upon cascaded mutation testing algorithms often found to be highly complex and cumbersome for ordering physicians. Thus, extended mutational profiling using NGS may show significant utility in the evaluation of acute myeloid leukemias and myeloproliferative neoplasms. Targeted NGS gene panels have also been reported to show potential, emerging significance in evaluation of myelodysplastic syndromes, one of the most common clinical indications for bone marrow biopsy. In the setting of acute leukemias and mantle cell lymphoma, minimal residual disease (MRD) testing by NGS has also been reported to show significant improvements in sensitivity and specificity compared to the standard reference methodologies including flow cytometry and PCR. Lastly, Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL) is the most common leukemia diagnosed among adults in western countries and is associated with heterogeneous clinical outcomes. Somatic hyper mutation status of the IGH gene is one of the most important prognostic biomarkers for risk stratification and guidance of therapy in this setting, and NGS confers significant practical and technical advantages over the current gold standard Sanger Sequencing based approach.Biography
Bevan Tandon, MD is board certified by the American Board of Pathology in both Hematologic Pathology and Molecular Genetic Pathology. His Hematopathology training was completed at the University of Pittsburgh under the guidance of WHO lead Author, Steven Swerdlow. His Molecular Pathology training at Washington University in St. Louis was focused on next generation sequencing for clinical testing in Oncology. He has multiple publications in the peer reviewed literature including the International Journal of Laboratory Hematology and Modern Pathology. He currently serves as the Director of Clinical Molecular Diagnostics at Molecular Pathology Laboratory Network, Inc., USA.
Email: btandon@mplnet.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi